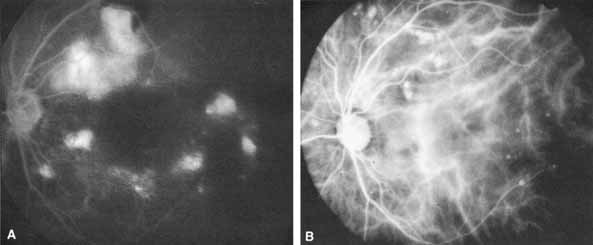
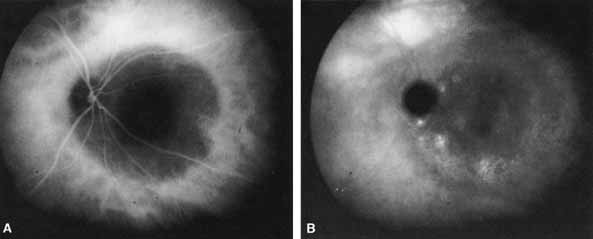
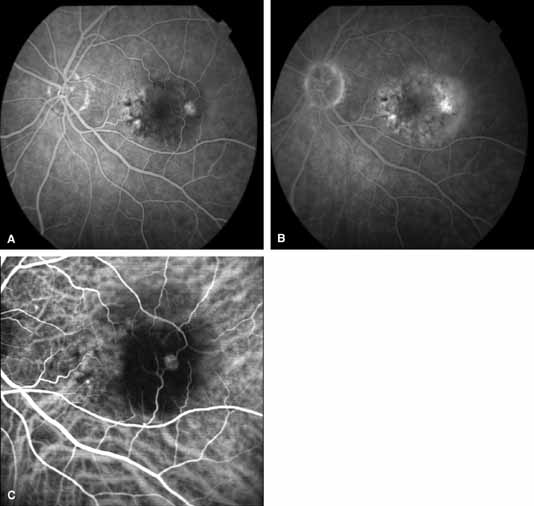
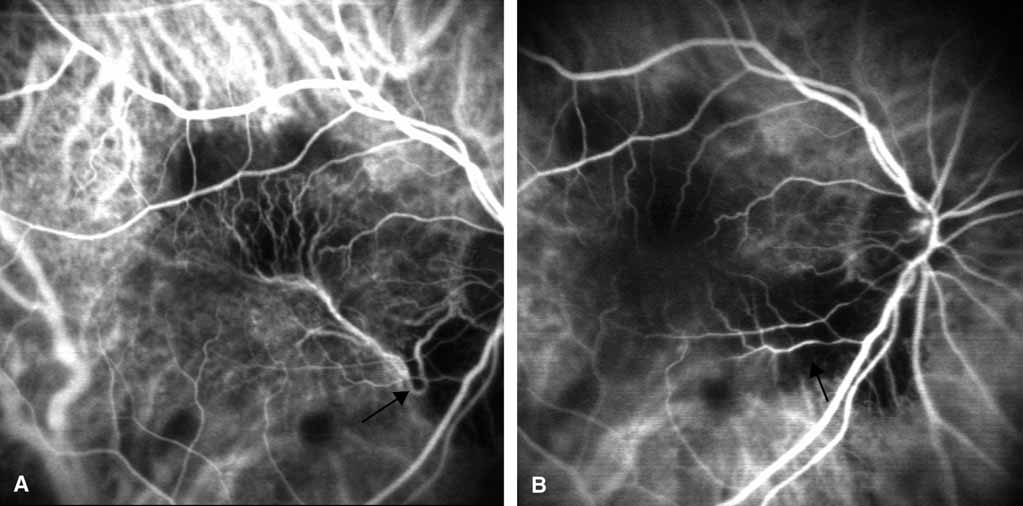
1. Destro M, Puliafito CA: Indocyanine green videoangiography of choroidal neovascularization. Ophthalmology 96:846–853, 1989 2. Scheider A, Schroedel C: High resolution indocyanine green angiography with a scanning laser ophthalmoscope. Am J Ophthalmol 108:458–459, 1989 3. Guyer DR, Puliafito CA, Monés JM, et al: Digital indocyanine-green angiography in chorioretinal disorders. Ophthalmology 99:287–291, 1992 4. Yannuzzi LA, Slakter JS, Sorenson JA, et al: Digital indocyanine green videoangiography and choroidal neovascularization. Retina 12:191–223, 1992 5. Kogure K, David NJ, Yamanouchi U, et al: Infrared absorption angiography of the fundus circulation. Arch Ophthalmol 83:209–214, 1970 6. Hochheimer BF: Angiography of the retina with indocyanine green. Arch Ophthalmol 86:564–565, 1971 7. Flower RW, Hochheimer BF: A clinical technique and apparatus for simultaneous angiography of the
separate retinal and choroidal circulations. Invest Ophthalmol 12:248–261, 1973 8. Bischoff PM, Flower RW: Ten years experience with choroidal angiography using indocyanine green
dye: A new routine examination or an epilogue? Doc Ophthalmol 60:235–291, 1985 9. Hayashi K, Hasegawa Y, Tokoro T: Indocyanine green angiography of central serous chorioretinopathy. Int Ophthalmol 9:37–41, 1986 10. Hayashi K, Hasegawa Y, Tazawa T, et al: Clinical application of indocyanine green angiography to choroidal neovascularization. Jpn J Ophthalmol 33(1):57–65, 1989 11. Regillo CD, Benson WE, Maguire JI, et al: Indocyanine green angiography and occult choroidal neovascularization. Ophthalmology 101:280–288, 1994 12. Scheider A, Kaboth A, Neuhauser L: Detection of subretinal neovascular membranes with indocyanine green and
an infrared scanning laser ophthalmoscope. Am J Ophthalmol 113:45–51, 1992 13. Kuck H, Inhoffen W, Schneider U, et al: Diagnosis of occult subretinal neovascularization in age-related
macular degeneration by infrared scanning laser videoangiography. Retina 13:36–39, 1993 14. Fox IJ, Wood EH: Indocyanine green: Physical and physiologic properties. Mayo Clin Proc 35:732–744, 1960 15. Cherrick GR, Stein SW, Leevy CM, Davidson CS: Indocyanine green: Observations on its physical properties, plasma decay, and
hepatic extraction. J Clin Invest 39:592–600, 1960 16. Paumgartner G: The handling of indocyanine green by the liver. Schweiz Med Wochenschr 105(17 suppl):1–30, 1975 17. Flower RW, Hochheimer BF: Indocyanine green dye fluorescence and infrared absorption choroidal angiography
performed simultaneously with fluorescein angiography. Johns Hopkins Med J 138:33–42, 1976 18. Benson RC, Kues HA: Fluorescence properties of indocyanine green as related to angiography. Phys Med Biol 23:159–163, 1978 19. Baker KJ: Binding of sulfobromophthalein (BSP) sodium and indocyanine green
by plasma alpha 1 lipoproteins. Proc Soc Exp Biol Med 122(4):957–963, 1966 20. Brown N, Strong R: Infrared fundus angiography. Br J Ophthalmol 57(10):797–802, 1973 21. Rapaport E, Ketterer SG, Wiegand BD: Hepatic clearance of indocyanine green. Clin Res 7:289–290, 1959 22. Probst P, Paumgartner G, Caucig H, et al: Studies on clearance and placental transfer of indocyanine green during
labor. Clin Chim Acta 29:157–160, 1970 23. Benya R, Quintana J, Brundage B: Adverse reactions to indocyanine green: A case report and a review of the
literature. Cathet Cardiovasc Diagn 17:231–233, 1989 24. Hope-Ross M, Yannuzzi LA, Gragoudas ES, et al: Adverse reactions to indocyanine green. Ophthalmology 101:529–533, 1994 25. Olsen TW, Lim JI, Capone A, et al: Anaphylactic shock following indocyanine green angiography. Arch Ophthalmol 114:97, 1996 26. Wolf S, Arend O, Schulte K, et al: Severe anaphylactic reaction after indocyanine green fluorescein angiography. Am J Ophthalmol 114:638–639, 1992 27. Yannuzzi LA, Rohrer KT, Tindel LJ, et al: Fluorescein angiography complication survey. Ophthalmology 93:611–617, 1986 28. Fineman MS, Maguire JI, Fineman SW, et al: Safety of indocyanine green angiography during pregnancy: A survey of the
retina, macula, and vitreous societies. Arch Ophthalmol 119(3): 352–355, 2001 29. Maberley DA, Cruess AF: Indocyanine green angiography: An evaluation of image enhancement for the
identification of occult choroidal neovascular membranes. Retina 19:37–44, 1999 30. Flower RW, Csaky KG, Murphy RP: Disparity between fundus camera and scanning laser ophthalmoscope indocyanine
green imaging of retinal pigment epithelium detachments. Retina 18:260–268, 1998 31. Bratsch D, Weinreb RN, Zinser G, et al: Confocal scanning infrared laser ophthalmoscopy for indocyanine green angiography. Am J Ophthalmol 120:642–651, 1995 32. Gelisken F, Inhoffen W, Schneider U, et al: Indocyanine green videoangiography of occult choroidal neovascularization: A
comparison of scanning laser ophthalmoscope with high-resolution
digital fundus camera. Retina 18:37–43, 1998 33. Brucker AJ, Brant A, Nyberg W: Landmark injection for localization of choroidal lesions using indocyanine
green angiography. Retina 13:169–171, 1993 34. Scheider A, Nasemann JE, Lund OE: Fluorescein and indocyanine green angiographies of central serous choroidopathy
by scanning laser ophthalmoscope. Am J Ophthalmol 115:50–56, 1993 35. Shields CL, Shields JA, DePotter P: Patterns of indocyanine green videoangiography of choroidal tumours. Br J Ophthalmol 79(3):237–245, 1995 36. Guyer DR, Yannuzzi LA, Slakter JS, et al: Digital indocyanine green videoangiography of central serous chorioretinopathy. Arch Ophthalmol 112:1057–1062, 1994 37. Dhaliwal RS, Maguire AM, Flower RW, et al: Acute posterior multifocal placoid pigment epitheliopathy: An indocyanine
green angiographic study. Retina 13:317–325, 1993 38. Ie D, Glaser BM, Murphy RP, et al: Indocyanine green angiography in multiple evanescent white-dot syndrome. Am J Ophthalmol 117:7–12, 1994 39. Prunte C, Niesel P: Quantification of choroidal blood-flow parameters using indocyanine
green video-fluorescence angiography and statistical picture
analysis. Graefes Arch Clin Exp Ophthalmol 226:55–58, 1988 40. Klein GJ, Baumgartner RH, Flower RW: An image processing approach to characterizing choroidal blood flow. Invest Ophthalmol Vis Sci 31:629–637, 1990 41. Flower RW, Klein GJ: Pulsatile flow in the choroidal circulation: A preliminary investigation. Eye 4:310–318, 1990 42. Flower RW: Extraction of choriocapillaris hemodynamic data from ICG fluorescence angiograms. Invest Ophthalmol Vis Sci 18:2720–2729, 1993 43. MacCumber MW, Flower RW, Langham ME: Ischemic hypertensive choroidopathy: Fluorescein angiography, indocyanine
green videoangiography, and measurement of pulsatile flow. Arch Ophthalmol 111:704–705, 1993 44. Regillo CD: The present role of indocyanine green angiography in ophthalmology. Curr Opin Ophthalmol 10:189–196, 1999 45. Ross RD, Barofsky JM, Cohen G, et al: Presumed macular choroidal watershed vascular filling, choroidal neovascularization, and
systemic vascular disease in patients with age-related
macular degeneration. Am J Ophthalmol 125:71–80, 1998 46. Goldberg MF, Dhaliwal RS, Olk RJ: Indocyanine green angiography patterns of zones of relative decreased choroidal
blood flow in patients with exudative age-related macular
degeneration. Ophthalmic Surg Lasers 29:385–390, 1998 47. Arnold JJ, Quaranta M, Soubrane G: Indocyanine green angiography of drusen. Am J Ophthalmol 124:344–356, 1997 48. Gass JDM: Stereoscopic Atlas of Macular Diseases: Diagnosis and Treatment, 4th ed, vol 1. St. Louis: CV Mosby, 1997:70–105 49. Lim JI, Sternberg P, Capone A, et al: Selective use of indocyanine green angiography for occult choroidal neovascularization. Am J Ophthalmol 120:75–82, 1995 50. Guyer DR, Yannuzzi LA, Slakter JS, et al: Classification of choroidal neovascularization by digital indocyanine green
videoangiography. Ophthalmology 103:2054–2060, 1996 51. Reichel E, Duker JS, Puliafito CA: Indocyanine green angiography and choroidal neovascularization obscured
by hemorrhage. Ophthalmology 102:1871–1876, 1995 52. Guyer DR, Yannuzzi LA, Slakter JS, et al: Digital indocyanine-green videoangiography of occult choroidal neovascularization. Ophthalmology 101:1727–1737, 1994 53. Sorenson JA, Yannuzzi LA, Slakter JS, et al: A pilot study of digital indocyanine green videoangiography for recurrent
occult choroidal neovascularization in age-related macular degeneration. Arch Ophthalmol 112:473, 1994 54. Guyer DR, Yannuzzi LA, Ladas I, et al: Indocyanine green-guided laser photocoagulation of focal spots at
the edge of plaques of choroidal neovascularization. Arch Ophthalmol 114:693–697, 1996 55. Bressler NM, Frost LA, Bressler SB, et al: Natural course of poorly defined choroidal neovascularization associated
with macular degeneration. Arch Ophthalmol 106:1537–1542, 1988 56. Soubrane G, Coscas G, Français C, et al: Occult subretinal new vessels in age-related macular degeneration: Natural
history and early laser treatment. Ophthalmology 97:649–657, 1990 57. Baumal CR, Reichel E, Duker JS, et al: Indocyanine green hyperfluorescence associated with serous retinal pigment
epithelial detachment in age-related macular degeneration. Ophthalmology 104:761–769, 1997 58. Lim JI, Aaberg TM, Capone A, et al: Indocyanine green angiography-guided photocoagulation of choroidal
neovascularization associated with retinal pigment epithelial detachment. Am J Ophthalmol 123:524–532, 1997 59. Chang TS, Freund KB, De La Cruz Z, et al: Clinicopathologic correlation of choroidal neovascularization demonstrated
by indocyanine green angiography in a patient with retention of good
visions for almost four years. Retina 14:114–124, 1994 60. Regillo CD, Blade KA, Custis PH, O'Connell SR: Evaluating persistent and recurrent choroidal neovascularization: The role
of indocyanine green angiography. Ophthalmology 105:1821–1826, 1998 61. Phillips WB, Regillo CD, Maguire JI: Indocyanine green angiography of idiopathic polypoidal choroidal vasculopathy. Ophthalmic Surg Lasers 27:467–470, 1996 62. Ahuja RM, Stanga PE, Vingerling JR, et al: Polypoidal choroidal vasculopathy in exudative and haemorrhagic pigment
epithelial detachements. Br J Ophthalmol 84:479–484, 2000 63. Moorthy RS, Lyon AT, Rabb MF, et al: Idiopathic polypoidal choroidal vasculopathy of the macula. Ophthalmology 105:1380–1385, 1998 64. Yannuzzi LA, Sorenson J, Spaide RF, Lipson B: Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina 10:1–8, 1990 65. Kleiner RC, Brucker AJ, Johnston RL: The posterior uveal bleeding syndrome. Retina 10:9–17, 1990 66. Ciardella AP, Donsoff IM, Yannuzzi LA: Polypoidal choroidal vasculopathy. Ophthalmol Clin N Am 15:537–554, 2002 67. Yanuzzi LA, Ciardella AP, Spaide RF, et al: The expanding clinical spectrum of idiopathic polypoidal choroidal vasculopathy. Arch Ophthalmol 115:478–485, 1999 68. Spaide RF, Yanuzzi LA, Slakter JS, et al: Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina 15:100–110, 1995 69. Labaut BA, Aisenbrey S, van den Broecke C, et al: Polypoidal choroidal vasculopathy pattern in age-related macular
degeneration. Retina 20:650–654, 2000 70. Yanuzzi LA, Wong DW, Sforzoline BS, et al: Polypoidal choroidal vasculopathy and neovascularized age-related
macular degeneration. Arch Ophthalmol 17:1503–1510, 1999 71. Spaide RF, Donsoff I, Lam DL, et al: Treatment of polypoidal choroidal vasculopathy with photodynamic therapy. Retina 22:529–535, 2002 72. Yannuzzi LA, Negrão S, Iida T, et al: Retinal angiomatous proliferation in age-related macular degeneration. Retina 21:416–434, 2001 73. Slatker JS, Yanuzzi LA, Scheider U, et al: Retinal choroidal anastomosis and occult choroidal neovascularization. Ophthalmol 107:742–753, 2000 74. Borrillo JL, Sivalingam A, Martidis A, Federman JL: Surgical ablation of retinal angiomatous proliferation. Arch Ophthalmol 12:558–561, 2003 75. Flower RW: Optimizing treatment of choroidal neovascularization feeder vessels associated
with age-related macular degeneration. Am J Ophthalmol 134:228–239, 2002 76. Melberg NS, Thomas MA: Successful feeder vessel laser treatment of recurrent neovascularization
following subfoveal surgery. Arch Ophthalmol 114:224–226, 1996 77. Shiraga F, Ojima Y, Matsuo T, et al: Feeder vessel photocoagulation of subfoveal choroidal neovascularization
secondary to age-related macular degeneration. Ophthalmology 105:662–669, 1998 78. Staurenghi G, Orzalesi N, La Capria A, et al: Laser treatment of feeder vessels in subfoveal choroidal neovascular membranes: A
revisitation using dynamic indocyanine green angiography. Ophthalmology 105:2297–2305, 1998 79. Desatnik H, Treister G, Alhalel A, et al: ICGA-guided laser photocoagulation of feeder vessels of choroidal
neovascular membranes in age-related macular degeneration. Retina 20:143–150, 2000 80. Schneider U, Gelisken F, Inhoffen W, et al: Indocyanine green angiographic findings in fellow eyes of patients with
unilateral occult neovascular age-related macular degeneration. Int Ophthalmol 21:79–85, 1997 81. Hanutsaha P, Guyer D, Yannuzzi LA, et al: Indocyanine green videoangiography of drusen as a possible predictive indicator
of exudative maculopathy. Ophthalmology 105:1632–1636, 1998 82. Piccolino FC, Borgia L: Central serous chorioretinopathy and indocyanine green angiography. Retina 14:231–242, 1994 83. Spaide RF, Hall L, Haas A, et al: Indocyanine green videoangiography of older patients with central serous
chorioretinopathy. Retina 16:203–213, 1996 84. Uyama M, Matsunaga H, Matsubara T, et al: Indocyanine green angiography and pathophysiology of multifocal posterior
pigment epitheliopathy. Retina 19:12–21, 1999 85. Shiraki K, Moriwaki M, Matsumoto M, et al: Long-term follow-up of severe central serous chorioretinopathy
using indocyanine green angiography. Int Ophthalmol 21:245–253, 1998 86. Yannuzzi LA, Slakter JS, Gross NE: Indocyanine green angiography-guided photodynamic therapy for treatment
of chronic central serous chorioretinopathy. Retina 23:288–298, 2003 87. Piccolino FC, Borgia L, Zinicola E: Indocyanine green angiography of circumscribed choroidal hemangiomas. Retina 16:19–28, 1996 88. Quaranta M, Arnold J, Coscas G, et al: Indocyanine green angiographic features of pathologic myopia. Am J Ophthalmol 122:663–671, 1996 89. Ohno-Matsui K, Morishima N, Ito M, et al: Indocyanine green angiographic findings of lacquer cracks in pathologic
myopia. Jpn J Ophthalmol 42:293–299, 1998 90. Ohno-Matsui K, Ito M, Tokoro T: Subretinal bleeding without choroidal neovascularization in pathologic
myopia: A sign of new lacquer crack formation. Retina 16:196–202, 1996 91. Obana A, Kusumi M, Miki T: Indocyanine green angiographic aspects of multiple evanescent white dot
syndrome. Retina 16:97–104, 1996 92. Howe LJ, Woon H, Graham EM, et al: Choroidal hypoperfusion in acute posterior multifocal placoid pigment epitheliopathy: An
indocyanine green angiography study. Ophthalmology 102:790–798, 1995 93. Park D, Schatz H, McDonald R, Johnson RN: Indocyanine green angiography of acute multifocal posterior placoid pigment
epitheliopathy. Ophthalmology 102:1877–1883, 1995 94. Pece A, Sadun F, Trabucchi G, et al: Indocyanine green angiography in enlarged blind spot syndrome. Am J Ophthalmol 126:604–607, 1998 95. Pece A, Avanza P, Introini U, et al: Indocyanine green angiography in angioid streaks. Acta Ophthalmol Scand 75:261–265, 1997 96. Quaranta M, Cohen S, Krott R, et al: Indocyanine green videoangiography of angioid streaks. Am J Ophthalmol 119:136–142, 1995 97. Oshima Y, Harino S, Hara Y, et al: Indocyanine green angiographic findings in Vogt-Koyanagi-Harada
disease. Am J Ophthalmol 122:58–66, 1996 98. Slakter JS, Giovannini A, Yannuzzi LA, et al: Indocyanine green angiography of multifocal choroiditis. Ophthalmology 104:1813–1819, 1997 99. Kohno T, Miki T, Hayashi K: Choroidopathy after blunt trauma to the eye: A fluorescein and indocyanine
green angiographic study. Am J Ophthalmol 126:248–260, 1998 100. Singh AD, De Potter P, Shields CL, et al: Indocyanine green angiography and ultrasonography of a varix of vortex
vein. Arch Ophthalmol 111:1283–1284, 1993 101. Piccolino FC, Borgia L, Zinicola E: Pre-injection fluorescence in indocyanine green angiography. Ophthalmology 103:1837–1845, 1996 |