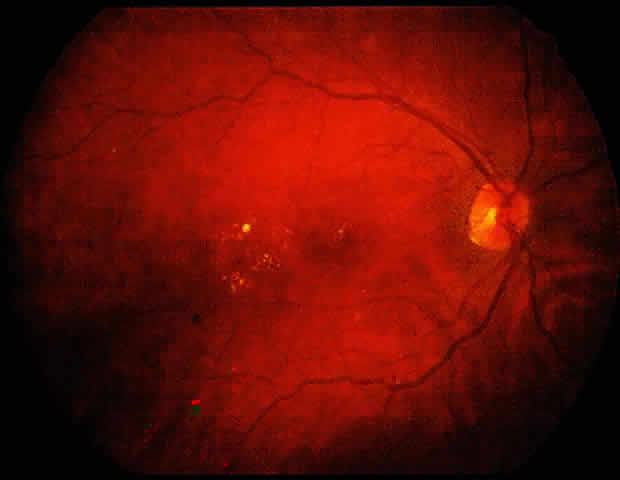
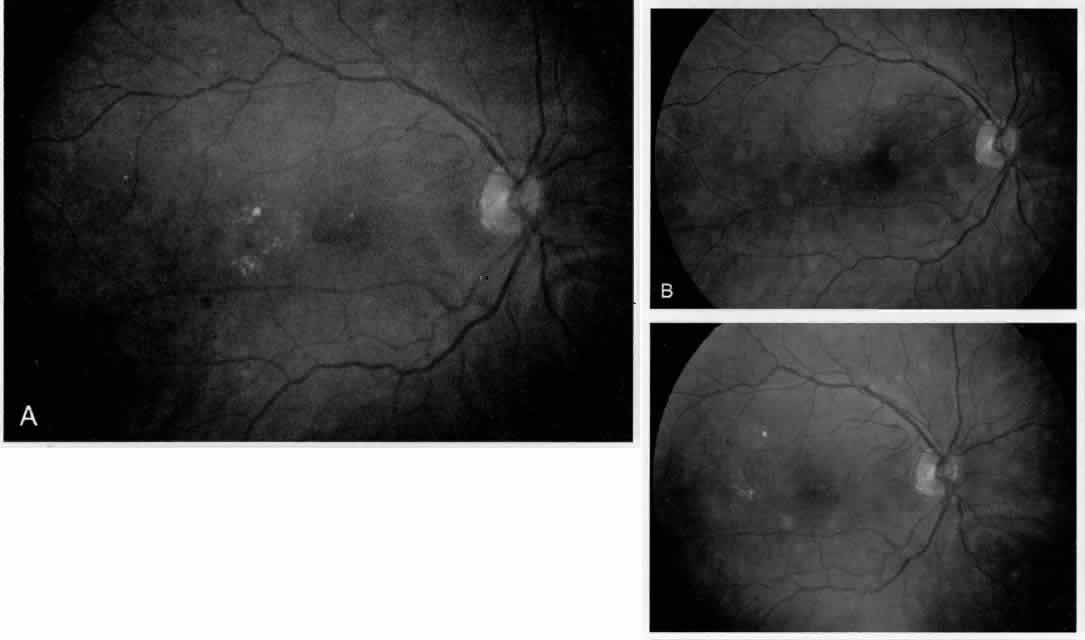
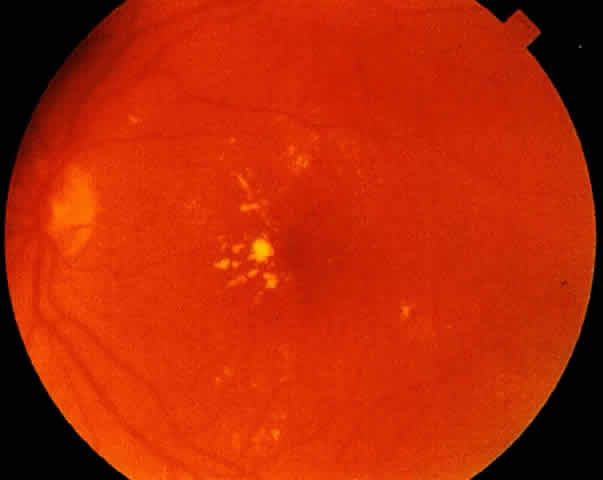
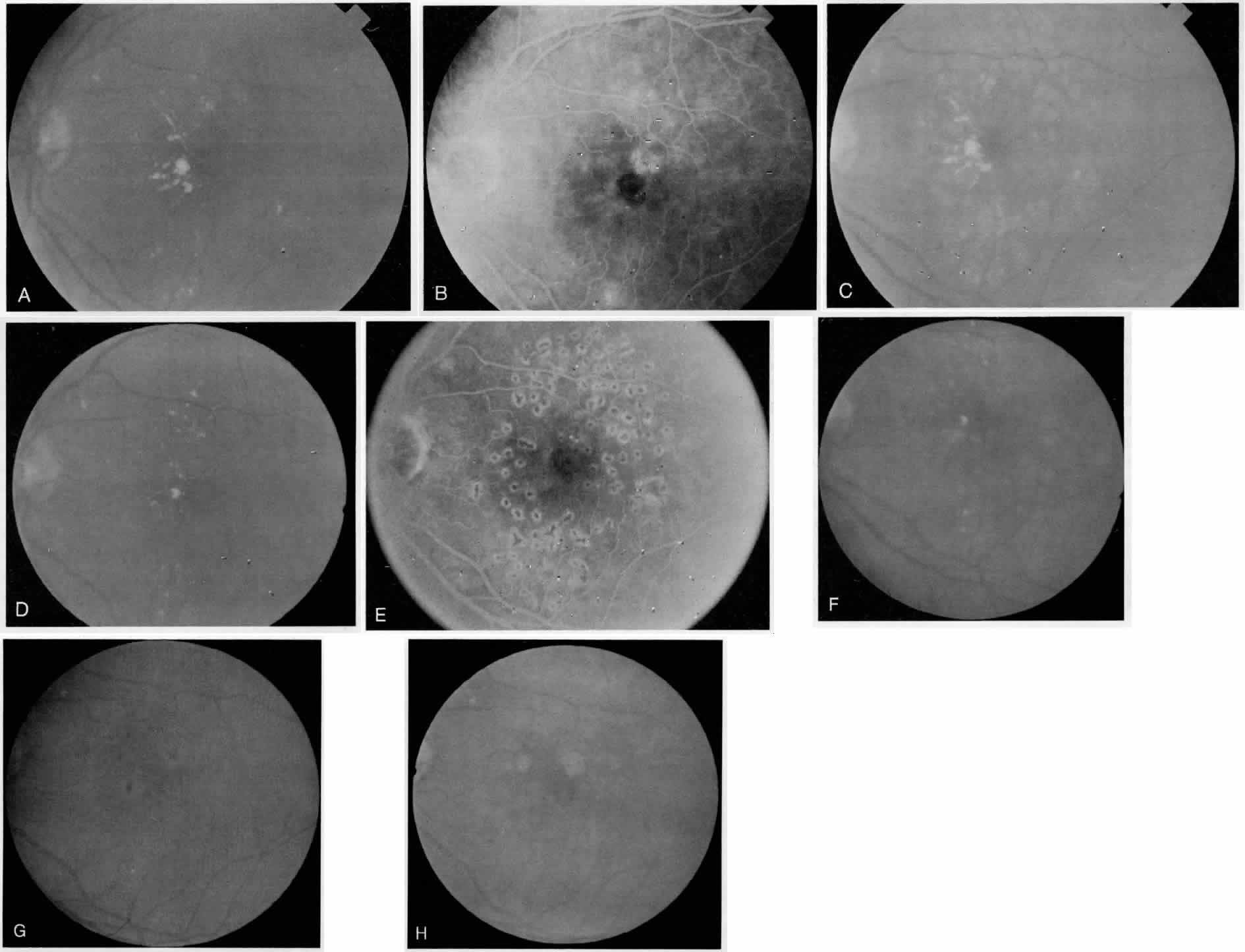
1. American Academy of Ophthalmology: Preferred Practice Pattern: Diabetic
Retinopathy. American Academy of Ophthalmology, San Francisco, CA 1989 2. Bresnick GH: Background diabetic retinopathy. In Ryan SJ (ed): Retina, pp 327–366. St. Louis, CV Mosby, 1989 3. Diabetic Retinopathy Study Research Group: A modification on the Airlie
House classification of diabetic retinopathy. Report No. 7. Invest Ophthalmol
Vis Sci 21:210, 1981 4. Frank RN: Etiologic mechanisms in diabetic retinopathy, In Ryan SJ (ed): Retina, pp 301–326. St. Louis, CV Mosby, 1989 5. Javitt JC, Canner JK, Sommer A: Cost effectiveness of current approaches to the control of retinopathy
in Type I diabetes. Ophthalmology 96:255, 1989 6. Javitt JC, Canner JK, Frank RG et al: Detecting and treating retinopathy in patients with Type I diabetes mellitus. Ophthalmology 97:483, 1990 7. Kohner EM, Sleightholm M, Kroc Collaborative Study Group (appended): Does
microaneurysm count reflect severity of early diabetic retinopathy? Ophthalmology 93:586, 1986 8. Wilkinson CP: The clinical examination: Limitation and over-utilization of angiographic
services. Ophthalmology 93:410, 1986 9. Moss SE, Klein R, Kessler SD et al: Comparison between ophthalmoscopy and fundus photography in determining
severity of diabetic retinopathy. Ophthalmology 92:62, 1985 10. Sussman EJ, Tsiaras WG, Soper KA: Diagnosis of diabetic eye disease. JAMA 247:3231, 1982 11. Klein R, Davis MD, Segal P et al: Diabetic retinopathy: Assessment of severity and progression. Ophthalmology 91:10, 1984 12. Klein R, Klein BEK, Neider NW et al: Diabetic retinopathy as detected using ophthalmoscopy, a nonmydriate camera
and standard fundus camera. Ophthalmology 92:485, 1985 13. Bresnick GH, Condit R, Syrjala S et al: Abnormalities of the foveal avascular zone in diabetic retinopathy. Arch Ophthalmol 102: 1286, 1984 14. Bresnick GH, Engerman R, Davis MD et al: Patterns of ischemia in diabetic retinopathy. Trans Am Acad Ophthalmol Otolaryngol 81:694, 1976 15. Patz A, Schatz H, Berkow JE et al: Macular edema: An overlooked complication of diabetic retinopathy. Trans Am Acad Ophthalmol Otolaryngol 77:34, 1973 16. Multicentered Controlled Study: Photocoagulation in treatment of diabetic
maculopathy. Lancet 2:1110, 1975 17. Blankenship GW: Diabetic macular edema and argon laser photocoagulation: A prospective
randomized study. Ophthalmology 86:69, 1979 18. Townsend C, Bailey J, Kohner E: Xenon arc photocoagulation for the treatment of diabetic maculopathy: Interim
report of a multicenter controlled clinical study. Br J Ophthalmol 64:385, 1980 19. Early Treatment Diabetic Retinopathy Study Research Group: Photocoagulation
for diabetic macular edema. ETDRS Report No. 1. Arch Ophthalmol 103: 1796, 1986 20. Olk RJ: Modified grid argon (blue-green) laser photocoagulation for diffuse diabetic
macular edema. Ophthalmology 93:938, 1986 21. Olk RJ: Argon green (514 nm) versus krypton red (647 nm) modified grid laser photocoagulation
for diffuse diabetic macular edema. Ophthalmology 97:1101, 1990 22. Early Treatment Diabetic Retinopathy Study Research Group: Treatment techniques
and clinical guidelines for photocoagulation of diabetic macular
edema. ETDRS Report No. 2. Ophthalmology 94:761, 1987 23. Early Treatment Diabetic Retinopathy Study Research Group: Photocoagulation
for diabetic macular edema: Early Treatment Diabetic Retinopathy
Study Report No. 4. Int Ophthalmol 27:265, 1987 24. Early Treatment Diabetic Retinopathy Study Research Group: Detection of
diabetic macular edema: Ophthalmoscopy vs. photography. ETDRS Report
No. 5. Ophthalmology 96:746, 1989 25. Bresnick GH: Diabetic maculopathy: A critical review highlighting diffuse macular edema. Ophthalmology 90: 1301, 1983 26. Lee CM, Olk RJ: Modified grid laser photocoagulation for diffuse diabetic
macular edema: Long-term visual results. (submitted) 27. Diabetic Retinopathy Study Research Group: Photocoagulation treatment of
proliferative diabetic retinopathy: The second report of the Diabetic
Retinopathy Study findings. Ophthalmology 85:82, 1978 28. Diabetic Retinopathy Study Research Group: Four risk factors for severe
visual loss in diabetic retinopathy: The third report of Diabetic Retinopathy
Study findings. Arch Ophthalmol 97:654, 1979 29. Diabetic Retinopathy Study Research Group: Indication for photocoagulation
treatment of diabetic retinopathy: Diabetic Retinopathy Study Report
No. 14. Int Ophthalmol Clin 27:239, 1987 30. Davis MD: Proliferative diabetic retinopathy. In Ryan SJ (ed): Retina. St. Louis, CV
Mosby, 1989 31. McDonald HR, Schatz H: Macular edema following panretinal photocoagulation. Retina 5:5, 1985 32. McDonald HR, Schatz H: Visual loss following panretinal photocoagulation for proliferative diabetic
retinopathy. Ophthalmology 92:388, 1985 33. Meyers SM: Macular edema after scatter laser photocoagulation for proliferative diabetic
retinopathy. Am J Ophthalmol 90:210, 1980 34. Early Treatment Diabetic Retinopathy Study Research Group: Techniques for
scatter and local photocoagulation treatment of diabetic retinopathy: The
Early Treatment Diabetic Retinopathy Study Report No. 3. Int Ophthalmol
Clin 27:254, 1987 35. Shimizu K, Kobayashi Y, Muraoka K: Midperipheral fundus involvement in diabetic retinopathy. Ophthalmology 88:601, 1981 36. Niki T, Muraoka K, Shimizu K: Distribution of capillary nonperfusion in early stage diabetic retinopathy. Ophthalmology 91:1431, 1984 37. Gass JD: Retinal capillary diseases. In Gass JD (ed): Stereoscopic Atlas
of Macular Diseases, p 368. St. Louis, CV Mosby, 1987 |