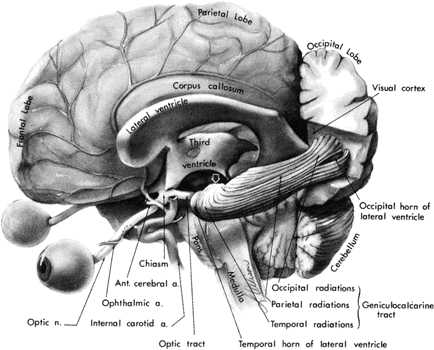
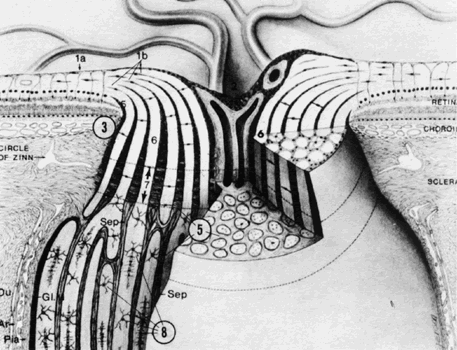
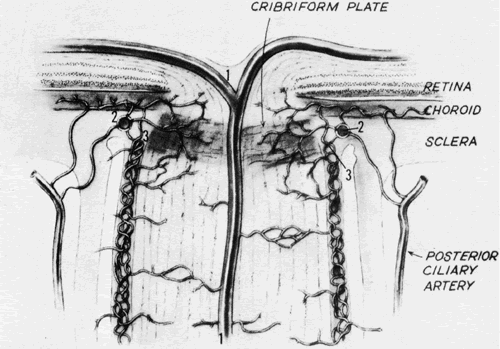
1. Buesch SR, Arey LB: The number of myelinated and unmyelinated fibers in the optic nerve of
vertebrates. J Comp Neurol 77:631, 1942 2. Lemire R, Loeser J, Leech R et al: Normal and abnormal development of the
human nervous system. In: The Optic System, pp. 196–205. New
York, Harper & Row, 1975 3. Provis JM, van Driel D, Billson FA et al: Human fetal optic nerve overproduction and elimination of retinal axons
during development. J Comp Neurol 238:92, 1985 4. Frost DO: Orderly anomalous retinal projections to the medical geniculate, vetrobasal
and lateral posterior nucleus of the hamster. J Comp Neurol 203:227, 1981 5. Rakic P: Prenatal development of the visual system in the rhesus monkey. Philos Trans R Soc Lond [Biol] 278:245, 1977 6. Godement P: Development of retinal projections in the mouse. In Stone J, Dreher
B, Rapaport DH, eds: Development of Visual Pathways in Mammals. New
York, Alan R. Liss, 1984 7. Livingstone M, Hubel D: Segregation of form, color, movement, and depth: Anatomy, physiology, and
perception. Science 240:740, 1988 8. Rodieck RW: The primate retina. In Steklis HD, Erwin J, eds: Comparative
Primate Biology Vol. 4, Neurosciences, pp. 203–278. New York, Alan
R. Liss, 1988 9. Honrobia FM, Elliot JH: Efferent innervation of the retina: Morphological study of the human retina. Arch Ophthalmol 80:98, 1968 10. Osterberg G: Topography of the layer of rods and cones in the human retina. Acta Ophthalmol (Suppl) 6:1, 1935 11. Van Buren JM: The Retinal Ganglion Cell Layer, p 130. Springfield, IL, Charles
C. Thomas, 1963 12. Glaser JS: The nasal visual field. Arch Ophthalmol 77:358, 1967 13. Hendrickson AK, Floren I, Patterson R et al: Neurotransmitter localization in the Macaca monkey retina. Invest Ophthalmol Vis Sci (Suppl) 20:237, 1981 14. Frederick JM, Rayborn ME, Laties AM et al: Dopaminergic neurons in thehuman retina. J Comp Neurol 210:65, 1982 15. Ogden TE: Nerve fiber layer of the primate retina: Morphometric analysis. Invest Ophthalmol Vis Sci 25:19, 1984 16. Bishop GH: Fiber groups in the optic nerve. Am J Physiol 106:460, 1933 17. Polyak S: The Retina. Chicago, University of Chicago Press, 1941 18. Perry VH, Oehler R, Dowey A: Retinal ganglion cells that project to the dorsal lateral geniculate nucleus
in the macaque monkey. Neuroscience 12:1101, 1984 19. Enroth-Cugell C, Robson GJ: The contrast sensitivity of retinal ganglion
cells of the cat. J Physiol 1966 20. Boycott BB, Wassle H: The morphological types of ganglion cells of the domestic cat's retina. J Physiol Lond 240:397, 1974 21. Rodieck RW: The Vertebrate Retina. San Francisco, WH Freeman, 1973 22. Stone J: Parallel processing in the visual system, pp 3397. New York, Plenum
Press, 1983 23. Leventhal AG, Rodieck RW, Dreher B: Retinal ganglion cell classes in the Old World monkey: Morphology and central
projections. Science 213:1139, 1981 24. Livingstone MS, Hubel DH: Psychophysical evidence for separate channels for the perception of form, color
movement, and depth. J Neurosci 7:3416, 1987 25. Rodieck RW, Binmuoeller KF, Dineen J: Parasol and midget ganglion cells of human retina. J Comp Neurol 233:115, 1985 26. Watanabe J, Rodieck RW: Parasol and midget ganglion cells of the primate retina. J Comp Neurol 289:434, 1989 27. Leventhal AG, Ault SJ, Vitek DJ: The nasotemporal division in primate retina: The neural bases of macular
sparing and splitting. Science 240:66, 1988 28. Ogden TE: Nerve fiber layer of the owl monkey retina: Retinotopic organization. Invest Ophthalmol Vis Sci 24:265, 1983 29. Jonas JB, Gusek GC, Naumann GOH: Optic disc, cup, and neuroretinal rim size, configuration and correlations
in normal eyes. Invest Ophthalmol Vis Sci 29:1151, 1988 30. Jonas JB, Schmidt AM, Muller-Bergli JA et al: Human optic nerve fier count and optic disc size. Invest Ophthalmol Vis Sci 33:2012, 1992 31a. Quigley HA, Brown AE, Morrison JP et al: The size and shape of the optic disc in normal human eyes. Arch Ophthalmol 108:51, 1990 31b. Johnson BM, Miao M, Sadun AA: Age-related decline of human optic nerve axon population. Age 10:509, 1987 32. Hernandez MR, Igoe F. Neufeld AH et al: Extracellular matrix of the human optic nerve head. Am J Ophthalmol 102:139, 1986 33. Minckler DS: Correlations between anatomic features and axonal transport in primate
optic nerve head. Trans Am Ophthalmol Soc 84:429, 1986 34. Anderson DR, Hoyt WF: Ultrastructure of intraorbital portion of human and monkey optic nerve. Arch Ophthalmol 82:506, 1969 35. Anderson DR: Ultrastructure of meningeal sheaths: Normal human and monkey optic nerves. Arch Ophthalmol 82:659, 1969 36. Anderson DR: Ultrastructure of human and monkey lamina cribrosa and optic nerve head. Arch Ophthalmol 82:800, 1969 37. Anderson DR: Ultrastructure of the optic nerve head. Arch Ophthalmol 83:63, 1970 38. Kurosawa H, Kurosawa A: Scanning electron microscopic study of pial septa of the optic nerve in
humans. Am J Ophthalmol 99:490, 1985 39. Anderson DR: Vascular supply of the optic nerve of primates. Am J Ophthalmol 70:341, 1970 40. Hayreh SS: Anatomy and physiology of the optic nerve head. Trans Am Acad Ophthalmol Otolaryngol 78:240, 1974 41. Fujii K, Chambers SM, Rhoton AL: Neurovascular relationships of the spheroid sinus: A microsurgical study. J Neurosurg 50:31, 1979 42. Walker AK: The neurosurgical evaluation of the chiasmal syndromes. Am J Ophthalmol 54:563, 1962 43. Bergland RM, Ray BS, Torack RM: Anatomical variations in the pituitary gland and adjacent structures in 2252 human
autopsy cases. J Neurosurg 28:93, 1968 44. Rucker CW: The concept of a semidecussation of the optic nerves. Arch Ophthalmol 59:159, 1958 45. Daniels DL, Haughton VM, Williams AC et al: Computed tmography of the optic chiasm. Radiology 137: 123, 1980 46. Parravano JG, Toledo A, Kucharczyk W: Dimensions of the optic nerves, chiasm, and tracts. MR quantitative comparison
between patients with optic atrophy and controls. J Comput Assist Tomogr 17:688, 1993 47. Barber AN, Ronstrom GN, Mueeling RJ: Development of the visual pathway: Optic chiasm. Arch Ophthalmol 52:447, 1954 48. Kupfer C, Chumbley L, Downer J, De CC: Quantitative histology of optic nerve, optic tract and lateral geniculate
nucleus of man. J Anat 101:393, 1967 49. Jacobson H, Hirose G: Origin of the retina from both sides of the embryonic brain: A contribution
to the problem of crossing at the optic chiasm. Science 202:637, 1978 50. Silver J, Sapiro J: Axonal guidance during development of the optic nerve: The role of pigmented
epithelia and other extrinsic factors. J Comp Neurol 202:521, 1981 51. Strongin AC, Guillery RW: The distribution of melanin in the developing optic cup and stalk and its
relation to cellular degeneration. J Neurosci 1:1193, 1981 52. Recordon E, Griffith SO: A case of primary bilateral anophthalmia. Br J Ophthalmol 22:253, 1938 53. Rogalski T: The visual path in a case of unilateral anophthalmia with special reference
to the problem of crossed and uncrossed visual fibers. J Anat 80:153, 1946 54. Sadun AA, Schaechter JD, Smith LEH: A retinohypothalamic pathway in man: Light mediation of circadian rhythms. Brain Res 302:371, 1984 55. Sadun AA, Johnson BM, Schaechter JD: Neuroanatomy of the human visual system: III. Three retinal projections
to the hypothalamus. Neuro-ophthalmology 6:371, 1986 56. Stephan FK, Sucker I: Circadian rhythms in drinking behavior and locomotor activity are eliminated
by hypothalamic lesions. Proc Natl Acad Sci USA 69:1583, 1982 57. Schaechter JD, Sadun AA: A second hypothalamic nucleus receiving retinal input in man: The paraventricular
nucleus. Brain Res 340:243, 1985 58. Chacko LW: The lamina pattern of the lateral geniculate body in primates. J Neurol Neurosurg Psychiatry 11:211, 1948 59. Hubel DH, Wiesel TN, LeVay S: Plasticity of ocular dominance columns in monkey striate cortex. Philos Trans R Soc Lond [Biol] 278:131, 1977 60. Kupfer C: The projection of the macula in the lateral geniculate nucleus of man. Am J Ophthalmol 54:597, 1962 61. Bishop PO, Kozak W, Levick WR, Vakkur GJ: The determination of the projection of the visual field on the lateral
geniculate nucleus in the cat. J Physiol Lond 163:503, 1962 62. Kaas JH, Guillery RW, Allman JM: Some principles of organization in the dorsal lateral geniculate nucleus. Brain Behav Evol 6:253, 1972 63. Lin CS, Kass JH: Projections from cortical visual areas 17, 18, and MT onto the dorsal lateral
geniculate nucleus in owl monkeys. J Comp Neurol 173:457, 1977 64. Zehi SM: Representation of central visual fields in prestriate cortex of monkeys. Brain Res 14:271, 1969 65. Lachica EA, Casagrande VA: The morphology and collicular axons ending on small relay (W-like) cells
of the primate lateral geniculate nucleus. Vis Neurosci 10:403, 1993 66. Lund JS, Boothe RG: Intralaminar connections and pyramidal neuron organization in the visual
cortex, area 17, of the macaque monkey. J Comp Neurol 159:305, 1975 67. Szentagothi J: Neuronal and synaptic architecture of the lateral geniculate
nucleus. In Jung R, ed: Handbook of Sensory Physiology, Vol. VII/3B, pp 141–176. Berlin, Springer-Verlag, 1973 68. Freund J-H: Neuronal mechanisms of the lateral genicuIate body. In Jung
R, ed: Handbook of Sensory Physiology, Vol. VII/3B, pp 177–246. Berlin, Springer-Verlag, 1973 69. Matthews MR: Transneuronal cell degeneration in the lateral geniculate nucleus of the
macaque monkey. J Anat 94:145–169169, 1960 70. Beatty B, Sadun A, Smith L, Richardson E: Direct demonstration of transsynaptic
degeneration in the human visual system: A comparison of retrograde
and anterograde changes J Neurol Neurosurg Psychiatry 45:143, 1982 71. Sadun AA: The neuroanatomy of the human visual system: 1. Retinal projections to
the LGN and PT as demonstrated with a new stain. Neuro-ophthalmology 6:353, 1986 72. Polyak S: The Vertebrate Visual System. Chicago, University of Chicago
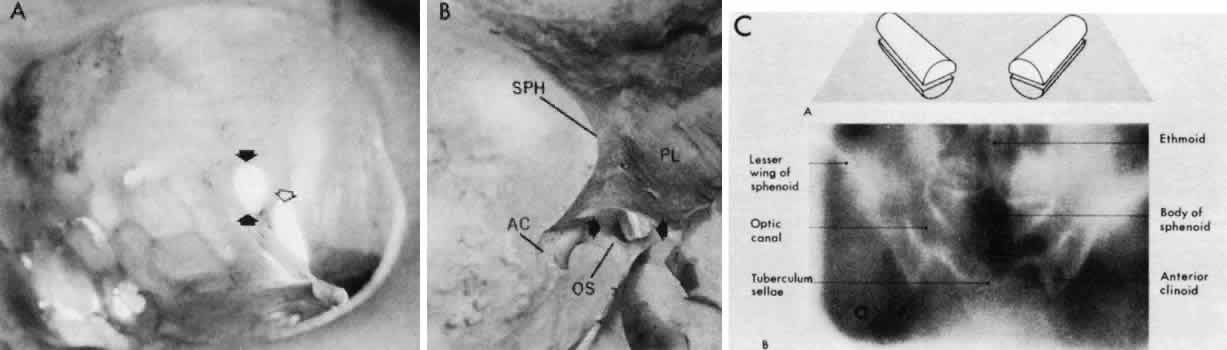
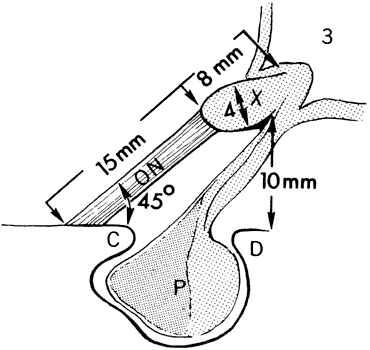
Press, 1957 73. Hoblen AL: The central visual pathways. In Davson H, ed: The Eye, Vol. 2A, Visual
Function in Man, pp 357–475. New York, Academic Press, 1976 74. Lund JS: Anatomical organization of macaque monkey striate visual cortex. Ann Rev Neurosci 11:253, 1988 75. Sadun AA: Parallel processing in the human visual system: A new perspective. Neuro-ophthalmology 6:351, 1986 76. Schiller PH: The role of the monkey superior colliculus in eye movement and vision. Invest Ophthalmol Vis Sci 11:451, 1972 77. Sadun AA, Johnson BM, Smith LEH: Neuroanatomy of the human visual system: 11. Retinal projections to the
superior colliculus and pulvinar. Neuro-ophthalmology 6:363, 1986 78. Fredericks CA, Giolli RA, Blanks RH, Sadun AA: The human accessory optic system. Brain Res 454:116, 1988 79. Adams JE, Rutkin BB: Visual responses to subcortical stimulation in the visual and limbic systems. Confin Neurol 32:158, 1970 80. Nashold BS: Phosphenes resulting from stimulation of the midbrain in man. Arch Ophthalmol 84:433, 1970 81. Brindley GS, Gautier-Smith PC, Lewin W: Cortical blindness and the functions of the non-geniculate fibers of the
optic tracts. J Neurol Neurosurg Psychiatry 32:259, 1969 82. Weiskrantz L: Blindsight: A Case Study in Implications. Oxford Psychological
Series #10. Oxford, Clarendon Press, 1986 83. Sadun AA: The afferent visual system. Anatomy and physiology. In M Yanoff, JS
Duker, eds: Ophthalmology. London, Mosby, 1999 84. Hoyt WF, Luis O: Visual fiber anatomy in the infrageniculate pathway of the primate: Uncrossed
and crossed retinal quadrant fiber projections studied with Nauta
silver stain. Arch Ophthalmol 6:94, 1962 85. Hoyt WF, Luis O: The primate chiasm: Details of visual fiber organization studies by silver
impregnation techniques. Arch Ophthalmol 70:69, 1963 86. Hoyt WF, Tudor RC: The course of parapapillary temporal retinal axons through the anterior
optic nerve: A Nauta degeneration study in the primate. Arch Ophthalmol 69:503, 1963 87. Potts AM, Hodges D, Shelman CB et al: Morphology of the primate optic nerve: I-III. Invest Ophthalmol Vis Sci 11:980, 1972 88. Spalding JMK: Wounds of the visual pathway: I. The visual radiation. J Neurol Neurosurg Psychiatry 15:99, 1952 89. Van Buren JM, Baldwin M: The architecture of the optic radiation in the temporal lobe of man. Brain 81:15, 1958 90. Holmes G: A contribution to the cortical representation of vision. Brain 54:470, 1931 91. Spalding JMK: Wounds of the visual pathway: II. The striate cortex. J Neurol Neurosurg Psychiatry 15:169, 1952 92. Stensaas MA, Eddington DK, Dobelle WH: The topography and variability of the primary visual cortex in man.J Neurosurg 40:747, 1974 93. Horton JC, Hoyt WF: The representation of the visual field in human striate cortex. Arch Ophthalmol 109:816, 1991 94. Brindley GS: Sensory effects of electrical stimulation of the visual and
paravisual cortex in man. In June R, ed: Handbook of Sensory Physiology, Vol. VII/3B, pp 583–594. Berlin, Springer-Verlag, 1973 95. Karten HJ, Keyser KT, Brecha NC: Biochemical and morphological heterogeneity
of retinal ganglion cells. In Cohen B, Bodis-Wollner I, eds: Vision
and the Brain. New York, Raven Press, 1984 96. Ehrlich D, Keyser A, Karten HJ: Distribution of substance P-like immunoreactive retinal ganglion cells
and their pattern of termination in the optic tectum of the chick (Gallus gallus). J Comp Neurol 266:220, 1987 97. Zucker CL, Dowling JE: Centrifugal fibers synapse on interplexiform cells in the teleost retina. Nature 330:166, 1987 98. Schiller PH: The on and of channels of the visual system. In Cohen B, Bodis-Wollner
I, eds: Vision and the Brain. New York, Raven Press, 1990 99. Reiter HO, Stryker MP: Neural plasticity without postsynaptic action potentials: Less active inputs
become dominant when kitten visual cortex cells are pharmacologically
inhibited. Proc Natl Acad Sci USA 85:3623, 1988 100. Livingstone M: Segregation of form, color, movement, and depth processing
in the visual system: Anatomy, physiology, art and illusion. In Cohen
B, Bodis-Wollner I, eds: Vision and the Brain. New York, Raven Press, 1990 101. Mikami A, Newsom WT, Wurtz RH: Motion selectivity in macaque visual cortex: I. Mechanisms of direction
and speed selectivity in extrastriate area MT. J Neurophysiol 55:1308, 1986 102. Sadun AA, Bassi C: Optic nerve damage in Alzheimer's disease. Ophthalmology 97:1, 1990 103. Fox PT, Fox JM, Raichle ME, Burde RM: The role of cerebral cortex in the generation of voluntary saccades: A
positron emission tomographic study. J Neurophysiol 54:348, 1985 104. Fitzke FW: Imaging the optic nerve and ganglion cell layer. Eye 14:450, 2000 105. Chen W, Kato T, Zhu XH et al: Mapping of lateral geniculate nucleus activation during visual stimulation
in human brain using fMRI. Magn Reson Med 39:89, 1998 106. Dupont P, Vogels R, Vandenberghe R et al: Regions in the human brain activated by simultaneous orientation discrimination: a
study with positron emission tomography. Eur J Neurosci 10:3689, 1998 107. Bergland RM, Ray BS: The arterial supply of the human optic chiasm. J Neurosurg 31:327, 1969 108. Smith CG, Richardson WFG: The course and distribution of the arteries supplying the visual (striate) cortex. Am J Ophthalmol 61:1391, 1966 |