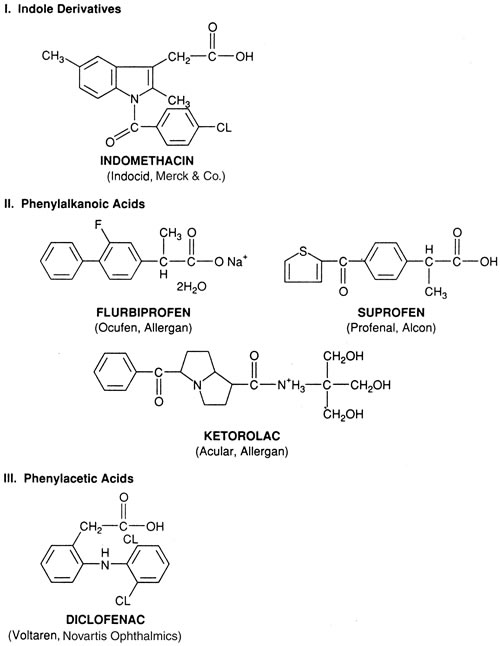
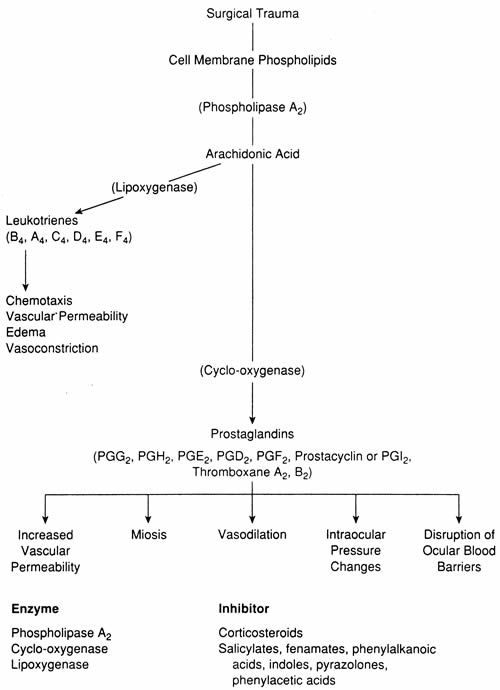
1. Goodman LS, Gilman A. Preface. In Hardman JG, Limbird LE, Gilman AG, eds. Goodman & Gilman's The Pharmacological Basis of Therapeutics, 10th
ed. New York: McGraw-Hill, 2001:xxvii 2. Spector R, Roberts R, Vesell ES: Clinical pharmacology: A new specialty. Am J Med 70:221, 1981 3. Soine TO, Willette RE: Analgesic agents. In Wilson CC, Cisvoed O, eds. Textbook of Organic Medicinal and Pharmaceutical Chemistry, 4th ed. Philadelphia: JB Lippincott, 1962:592 4. Abrishami MA, Thomas J: Aspirin intolerance: A review. Ann Allergy 39:28, 1977 5. Roberts LJ, Morrow JD: Analgesic-antipyretics and anti-inflammatory agents: Drugs
employed in the treatment of rheumatoid arthritis and gout. In Hardman JG, Limbird LE, Gilman AG, eds. Goodman & Gilman's The Pharmacological Basis of Therapeutics, 10th
ed. New York: McGraw-Hill, 2001:687–732 6. Simon LS, Mills JA: Nonsteroidal anti-inflammatory drugs. N Engl J Med 302:1237, 1980 7. Day RO, Graham GG, William KM, et al: Clinical pharmacology of non-steroidal anti-inflammatory
drugs. Pharmacol Ther 33:383, 1987 8. Magons K, Ball JP, Escale R, et al: Evidence for the existence of high affinity binding sites for indomethacin. Mol Pharmacol 29:39, 1985 9. Mochizuki M, Sawa M, Masuda K: Topical indomethacin in intracapsular extraction of senile cataract. Jpn J Ophthalmol 21:215, 1977 10. Brocks DR, Jamali F: Clinical pharmacokinetics of ketorolac tromethamine. Clin Pharmacokinet 23:415, 1992 11. Flurbiprofen—An ophthalmic NSAID. Med Lett Drugs Ther 29:58, 1987 12. Podos SM, Becker B: Comparison of ocular prostaglandin synthesis inhibitors. Invest Ophthalmol 15:841, 1976 13. Rooks WH, Maloney PJ, Shott LD, et al: The analgesic and anti-inflammatory profile of ketorolac and its
tromethamine salt. Drugs Exp Clin Res 11:479, 1985 14. Skoutakis VA, Carter GA, Mickle TR, et al: Review of diclofenac and evaluation of its place in therapy as a nonsteroidal
anti-inflammatory agent. Drug Intell Clin Pharm 22:850, 1988 15. Todd PA, Heel RC: Suprofen—A review of its pharmacodynamic and pharmacokinetic properties
and analgesic efficacy. Drug Eval 30:514, 1985 16. Todd PA, Sarkin EM: Diclofenac sodium: A reappraisal of its pharmacodynamics and pharmacokinetic
properties and therapeutic efficacy. Drugs 35:244, 1988 17. Benson MD, Aldo-Benson M, Brandt KD: Synovial fluid concentrations of diclofenac in patients with rheumatoid
arthritis and osteoarthritis. Semin Arthritis Rheum 15:65, 1985 18. Buckley MMT, Brogden RN: Ketorolac. A review of its pharmacodynamic and pharmacokinetic properties
and therapeutic potential. Drugs 39:86, 1990 19. Jallad NS, Garg DC, Martinez JJ, et al: Pharmacokinetics of single-dose oral and intramuscular ketorolac
tromethamine in the young and elderly. J Clin Pharmacol 30:76, 1990 20. Tang-Lui DD, Liu SS, Weinkam RJ, et al: Ocular and systemic bioavailability of ophthalmic flurbiprofen. J Pharmacokinet Biopharm 12:611, 1984 21. Weintraub HS: Human acute pharmacokinetics and comparative bioavailability of suprofen
from various dosage forms. Clin Res 26:595, 1978 22. Brune K, Glatt M, Graf P: Mini review: Mechanisms of action of anti-inflammatory drugs. Gen Pharmacol 7:27, 1976 23. Kaarela K, Lehtinen K, Makisara P, et al: Pharmacokinetics and tolerance of slow-released indomethacin tablets
in rheumatoid arthritis. Eur J Clin Pharmacol 23:349, 1982 24. Huckey HB, Kwan KC, Duggan DE: Pharmacokinetics and metabolism of NSAIDs. Prog Drug Metab 5:165, 1980 25. Meffin PJ: The effect of renal dysfunction on the disposition of NSAIDs forming acyl-glucuronides. Agents Actions 17(suppl):85, 1985 26. Conquet PH, Plazonnet B, LeDourarec JC: Arachidonic acid-induced elevation of intraocular pressure and anti-inflammatory
agents. Invest Ophthalmol 14:772, 1975 27. Hanna C, Sharp D: Ocular absorption of indomethacin by the rabbit. Arch Ophthalmol 88:196, 1972 28. Sanders DR, Kraff MC, Goldstick B: Aqueous penetration of oral and topical indomethacin in humans. Arch Ophthalmol 101:1614, 1983 29. Ling TL, Combs DL: Ocular bioavailability and tissue distribution of ketorolac tromethamine
in rabbits. J Pharm Sci 76:289, 1987 30. Van Haeringen NJ: A comparison of the effects of nonsteroidal compounds on the disruption
of the blood-aqueous barrier. Exp Eye Res 35:271, 1982 31. Miyake K, Sugiyama S, Noriamatsu I, et al: Prevention of cystoid macular edema after lens extraction by topical indomethacin—Radio-immunoassay measurement of prostaglandins
in the aqueous during and after lens extraction. Albrecht von Graefes Arch Klin Exp Ophthalmol 209:83, 1978 32. Flower RJ, Blackwell GJ: Anti-inflammatory steroids induce biosynthesis of a phospholipase
A2 inhibitor which prevents PG production. Nature 278:456, 1979 33. Mroszczak EJ, Ling T, Yee J, et al: Ketorolac absorption and pharmacokinetics in humans. Clin Pharmacol Ther 37:215, 1985 34. Fraunfelder FT: Extraocular fluid dynamics: How best to apply topical ocular medication. Am Ophthalmol Soc Trans 74:457, 1976 35. Zimmerman TJ, Kooner KS, Kandarakis AS: Improving the therapeutic index of topically applied ocular drugs. Arch Ophthalmol 102:551, 1984 36. Zipser RD, Laffi G: Prostaglandins, thromboxanes and leukotrienes in clinical medicine. West J Med 143:485, 1985 37. Ambache N: Irin, a smooth-muscle contracting substance present in rabbit iris. J Physiol 129:65, 1955 38. Eakins KE: Prostaglandins and prostaglandin synthetase inhibitors: Actions in ocular
disease. In Robinson HJ, Vane JR, eds. Prostaglandin Synthetase Inhibitors. New York: Raven Press, 1974:343 39. Neufeld AH, Sears ML: Prostaglandins and the eye. Prostaglandins 4:157, 1973 40. Waitzman MB: Possible new concepts relating prostaglandins to various ocular functions. Surv Ophthalmol 14:301, 1970 41. Eakins KE, Whitelocke RAF, Bennett A, et al: Prostaglandin-like activity in ocular inflammation. Br Med J 3:452, 1972 42. Masuda K, Izawa Y, Mishima S: Prostaglandins and uveitis. Jpn J Ophthalmol 17:166, 1973 43. Abelson MB, Butrus SI, Weston JH: Aspirin therapy in vernal conjunctivitis. Am J Ophthalmol 95:502, 1983 44. Vane JR: Prostaglandins as mediators of inflammation. Prostaglandin Thromboxane Res 2:791, 1976 45. Boctor AM, Eickholt M, Pugsley TA: Meclofenamate is an inhibitor of both 5-lipoxygenase and cyclooxygenase
in vitro. Prostaglandins Leukot Med 23:229, 1986 46. Siegel MI, McConnell RT, Porter NA: Aspirin-like drugs inhibit arachidonic acid metabolism via lipoxygenase
and cyclo-oxygenase in rat neutrophils from carrageenan
pleural exudates. Biochem Biophys Res Commun 92:688, 1980 47. Bonomi L, Perfetti S, Bellucci R, et al: Prevention of surgically induced sinuses by diclofenac eye drops. Ann Ophthalmol 19:142, 1987 48. Bomalaski JS, Hirata F, Clark MA: Aspirin inhibits phospholipase C. Biochem Biophys Res Commun 139:115, 1986 49. Ku EC, Lee W, Kothan HV, et al: Effect of diclofenac on rachidonic acid cascade. Am J Med 80(Suppl 4B):18, 1986 50. Abelson MB, Madrwale NA, Weston NH: The role of PGD2 in allergic ocular disease. In O'Connor GR, Chandler JM, eds. Advances in Immunology and Immunopathology of the Eye. New York: Masson, 1985:163–166 51. Abramson SB, Weissman G: The mechanisms of action of nonsteroidal anti-inflammatory drugs. Arthritis Rheum 32:1, 1989 52. Lee RF: The influence of psychotropic drugs on prostaglandin biosynthesis. Prostaglandins 5:63, 1974 53. Abramson S, Korchaktt LR, Edelson H, et al: Modes of action of aspirin-like drugs. Proc Natl Acad Sci USA 82:7227, 1985 54. Forrest M, Brooks PM: Mechanism of action of non-steroidal anti-rheumatic drugs. Baillieres Clin Rheumatol 2:275, 1988 55. Biemond P, Swaak AG, Penders JA, et al: Superoxide production by PMNs in rheumatoid arthritis and osteoarthritis. Ann Rheum Dis 45:249, 1986 56. Siegel M, McConnell R, Cuatrecasas A: Aspirin-like drugs interfere with arachidonate metabolism by inhibition
of peroxidase activity of lipoxygenase pathway. Proc Natl Acad Sci USA 76:3774, 1979 57. Kim C, Tirama LA, Mann JD, et al: Saturable accumulation of an anion herbicide by rabbit choroid plexus. J Pharmacol Exp Ther 225:699, 1983 58. Herman JH, Appel AM, Khosia RC, et al: Cytokine modulation of chondrocyte metabolism. Inflammation 8:5125, 1984 59. Simchowitz L, Mehta J, Spilberg I: Chemotactic factor–induced generation of superoxide radicals by human
neutrophils, effect of anti-inflammatory drugs. Arthritis Rheum 22:755, 1979 60. Perez HD, Elfman F, Marder S: Meclofenamate inhibits chemotactic factor–induced human PMN function. Arthritis Rheum 30:1023, 1987 61. Menta JO, Williams MD: Some NSAIDs inhibit the generation of superoxide anions by activated polymorphs
by blocking ligand-receptor interactions. J Rheumatol 12:751, 1985 62. Silverstein FE, Graham DY, Senior JR, et al: Misoprostol reduced serious complications in patients with rheumatoid arthritis
receiving nonsteroidal anti-inflammatory drugs. Ann Intern Med 123:241, 1995 63. Garcia L: Pain-free at last. Dallas Morning News. Sept 6, 1999:1C 64. Needleman P, Isakson PC: COX-1 and COX-2. J Rheumatol 24:6, 1997 65. Warner TD, Giuliano F, Vojnovic I, et al: Nonsteroid drug selectivities for cyclo-oxygenase-1 rather
than clclooxygenase-2 are associated with human gastrointestinal
toxicity. Proc Natl Acad Sci USA 96:7563, 1999. 66. Peterson WL, Cryer B: COX-1-Sparing NSAIDs-Is the enthusiasm justified? JAMA 2821961, 1999 67. Bombardier C, Laine L, Reicin A, et al: For VIGOR Study Group. Comparison of upper gastrointestinal toxicity of
rofecoxib and naproxen in patients with rheumatoid arthritis. N Engl J Med 343:1520, 2000 68. Silverstein FE, Faich G, Goldstein JF, et al: Gastrointestinal toxicity with celecoxib vs NSAIDs for osteoarthritis and
rheumatoid arthritis. The CLASS Study: A randomized controlled trial. JAMA 284:1247, 2000 69. Martinez B, Mathews AW, Lublin JS, et al: Merck pulls Vioxx from market after link to heart problems. Wall Street Journal October 1,2004:A1 70. Mukherjee D, Nissen SE, Topol EJ: Risk of cardiovascular events associated with selective COX-2 inhibitors. JAMA 286:954, 2001 71. Kloner DW: Relationship between cyclo-oxygenase-2 inhibition and thrombogenesis. J Cardiovasc Pharmacol Ther 9:51, 2004 72. Crofford LJ: Specific cyclo-oxygenase-2 inhibitors: What have we learned
since they came into widespread clinical use? Curr Opin Rheumatol 13:225, 2002 73. Chiolero A, Maillard MP, Burnier M: Cardiovascular hazard of selective COX-2 inhibitors: myth or reality? Expert Opin Drug Saf 1:45, 2002 74. Hankey GJ, Eikelboom JW: Cyclooxygenase-2 inhibitors: Are they really atherothrombotic, and
if not, why not? Stroke 34:2736, 2003 75. Proceeding of the National Academy of Science 10:1073, 2002 76. Duffin RM, Camras CB, Gardner SK: Inhibitors of surgically induced miosis. Ophthalmology 89:966, 1982 77. Camras CB, Miranda OC: The putative role of prostaglandins in surgical miosis. In Bito LZ, Stjernschantz J, eds. The Ocular Effects of Prostaglandins and Other Eicosanoids. New York: Alan R. Liss, 1989 78. Waitzman MB, King CD: Prostaglandin influences on intraocular pressure and pupil size. Am J Physiol 212:329, 1967 79. Kramer SG, Oyakawa RT, Drake M: Enhancement of pupillary dilation during intraocular surgery by prostaglandin
inhibition. Invest Ophthalmol Vis Sci 15:63, 1976 80. Bito LZ: Species differences in the response of the eye to irritation and trauma: A
hypothesis of divergence in ocular defense mechanisms, and the choice
of experimental animals for eye research. Exp Eye Res 39:807, 1984 81. Mishima S, Masuda K: Clinical complications of prostaglandins and synthesis inhibitors. In Leopold IH, Burns RP, eds. Symposium on Ocular Therapy, Vol. 10. New York: John Wiley & Sons, 1977:1–19 82. ICL in Treatment of Myopia (ITM) Study Group: Postoperative inflammation after implantation of the implantable contact
lens. Ophthalmology 110:2335, 2003 83. Dick HB, Schwenn O, Krummenauer F, et al: Inflammation after sclerocorneal versus clear corneal tunnel phacoemulsification. Ophthalmology 107:241, 2000 84. Dowler JGF, Hykin PG, Hamilton AMP: Phacoemulsification versus extracapsular cataract extraction in patients
with diabetes. Ophthalmology 107:457, 2000 85. Friedlaender MH: Allergy and Immunology of the Eye. New York: Harper & Row, 1979 86. O'Connor GR: Corticosteroids and immuno-suppressives reviewed. In Srinivasan BD, ed. Ocular Therapeutics. New York: Masson, 1980:69 87. Dunne JA, Jacobs N, Morrison A, et al: Efficacy in anterior uveitis of two unknown steroids and topical tolmetin. Br J Ophthalmol 69:12, 1985 88. Hogan MJ, Kimura SJ, Thygeson P: Signs and symptoms of uveitis. Am J Ophthalmol 47:155, 1959 89. Yourston D, Whicher J, Chambers R, et al: The acute phase response in acute anterior uveitis. Trans Ophthalmol Soc UK 104:166, 1985 90. Fearnley IR, Spalton DJ, Smith SE: Anterior segment fluorophotometry in acute anterior uveitis. Arch Ophthalmol 105:1550, 1987 91. Flach AJ, Graham J, Kruger L, et al: Quantitative assessment of postsurgical breakdown of the blood aqueous
barrier following administration of ketorolac tromethamine solution: A
double-masked, paired comparison with vehicle-placebo solution
study. Arch Ophthalmol 106:344, 1988 92. Mishima S, Tanishima T, Masuda K: Pathophysiology and pharmacology of intraocular surgery. Aust N Z J Ophthalmol 13:147, 1985 93. Podos SM. Discussion of nonsteroidal anti-inflammatory agents after argon
laser trabeculoplasty. Ophthalmology 91:97, 1984 94. Sanders DR, Kraff MC: Steroidal and nonsteroidal anti-inflammatory agents. Effects on
postsurgical inflammation and blood-aqueous barrier breakdown. Arch Ophthalmol 102:1453, 1984 95. Weinreb RN, Rohm AL, Baerveldt G, et al: Flurbiprofen pretreatment in argon laser trabeculoplasty for primary open
angle glaucoma. Arch Ophthalmol 102:1629, 1984 96. Flach AJ, Stegman RC, Graham J: Prophylaxis of aphakic cystoid macular edema without corticosteroids. Ophthalmology 97:1253, 1990 97. Kuchle M, Naumann GOH: Intraocular inflammation after cataract surgery. Ophthalmology 110:632, 2003 98. Stjernschantz J: Autacoids and neuropeptides. In Sears ML (ed): Pharmacology of the Eye. New York: Springer-Verlag, 1984:311 99. Ma uger TF, Craig EL: Havener's Ocular Pharmacology, 6th ed. St. Louis: CV Mosby, 1994:364 100. Stewart R, Horwitz B, Howes J, et al: Double-masked, placebo-controlled evaluation of loteprednol
etabonate 0.5% for postoperative inflammation. J Cataract Refract Surg 24:1480, 1998 101. The Loteprednol Etabonate Postoperative Inflammation Study Group 2: A double-masked, placebo-controlled evaluation of 0.5% loteprednol
etabonate in the treatment of postoperative inflammation. Ophthalmology 105:1780, 1998 102. Fan DSP, Yu CBO, Chiu TYH: Ocular-hypertensive and anti-inflammatory response to rimexolone
therapy in children. Arch Ophthalmol 121:1716, 2003 103. Floman N, Zor U. Mechanism of steroid action in ocular inflammation: Inhibition of prostaglandin
production. Invest Ophthalmol 16:69, 1977 104. Kazuo S, Tsurufuji S: Mechanism of anti-inflammatory action of glucocorticoids: Re-evaluation
of vascular constriction hypothesis. Br J Pharmacol 73:605, 1981 105. Stark WJ, Worthen DM, Halladay JT: The FDA report on intraocular lenses. Ophthalmology 90:311, 1983 106. Irvine AR: Cystoid maculopathy. Surv Ophthalmol 21:1, 1976 107. Jampol LM: Aphakic cystoid macular edema. A hypothesis. Arch Ophthalmol 103:1134, 1985 108. Miller D, Gruenberg P, Miller P, et al: Topical flurbiprofen or prednisolone: Effect on corneal wound healing in
rabbits. Arch Ophthalmol 99:681, 1981 109. Yannuzzi LA: Perspective on the treatment of aphakic cystoid macular edema. Surv Ophthalmol 28(May Suppl):540, 1984 110. Flach AJ: The incidence, pathogenesis and treatment of cystoid macular edema following
cataract surgery. Trans Am Ophth Soc 95:557, 1998 111. Cunha-Vaz JG, Travassos A: Breakdown of the blood-retinal barriers and cystoid macular edema. Surv Ophthalmol 28(May Suppl):485, 1984 112. Gass JDM, Norton EWD: Cystoid macular edema and papilledema following cataract extraction. Arch Ophthalmol 76:646, 1966 113. Irvine SR: A newly defined vitreous syndrome following cataract surgery: Interpreted
according to recent concepts of the structure of the vitreous. Am J Ophthalmol 36:699, 1953 114. Martin WF, Green WR, Martin LW: Retinal phlebitis in the Irvine-Gass syndrome. Am J Ophthalmol 83:377, 1977 115. Norton AL, Brown WJ, Carson M: Pathogenesis of aphakic macular edema. Am J Ophthalmol 80:86, 1975 116. Obstbaum SA, Galen MA: Cystoid macular edema and ocular inflammation. The corneo-retinal
inflammatory syndrome. Trans Ophthalmol Soc UK 99:187, 1979 117. Stark WJ, Maumenee AE, Fagadau W: Cystoid macular edema in pseudophakia. Surv Ophthalmol (May suppl) 28:442, 1984 118. Wilkinson CP: A long-term follow-up study of cystoid macular edema in aphakic
and pseudophakic eyes. Trans Am Ophthalmol Soc 79:810, 1981 119. Sears ML: Aphakic cystoid macular edema: The pharmacology of ocular trauma. Surv Ophthalmol 28(May Suppl):525, 1984 120. Bonnet M, Bievelez B: Iris fluorescein angiography and Irvine-Gass syndrome. Albrecht von Graefes Arch Klin Exp Ophthalmol 213:187, 1980 121. Kondo T, Havashi M, Miki M: The results of intraocular lens implantation. The relationship between
anterior segment fluorescein angiograph and cystoid macular edema. Acta Soc Ophthalmol Jpn 85:451, 1981 122. Kraff MC, Sanders DR, Jampol LM: Effect of an ultraviolet filtering intraocular lens on cystoid macular
edema. Ophthalmology 92:366, 1985 123. Yamaaki H, Hendriks F, Deutman AF: Iris angiography after cataract extraction and the effect of indomethacin
eye drops. Ophthalmologica 188:82, 1984 124. Kremer M, Baikoff G, Charbonnel B: The release of prostaglandins in human aqueous after intraocular surgery: Effect
of indomethacin. Prostaglandins 23:695, 1982 125. Miyake K: Prostaglandins as a causative factor of the cystoid macular edema after
lens extraction. Acta Soc Ophthalmol Jpn 81:1449, 1977 126. Worst JCF: Biotoxizitat des Kammerwassers. Klin Monatsbl Augenheilkd 167:376, 1975 127. Samuelsson B, Goldone M, Cranstrom E, et al: Prostaglandins and thromboxanes. Annu Rev Biochem 47:997, 1978 128. Eakins KE: Prostaglandin and non-prostaglandin mediated breakdown of the blood-aqueous
barrier. Exp Eye Res 80(suppl):483, 1977 129. Kottow M, Hendrickson P: Iris angiography in cystoid macular edema after cataract extraction. Arch Ophthalmol 93:487, 1975 130. Neufeld AH, Jampol LM, Sears ML: Aspirin prevents the disruption of the blood-aqueous barrier in
the rabbit eye. Nature 238:158, 1972 131. Bito LZ, Salvador EV: Intraocular fluid dynamics: The site of mechanism of prostaglandin transfer
across the blood intraocular barriers. Exp Eye Res 14:233, 1972 132. Peyman GA, Sptiznas M, Straatsma BR: Chorioretinal diffusion of peroxidase before and after photocoagulation. Invest Ophthalmol 10:489, 1971 133. Gass JDM, Norton EWD: Follow-up of aphakic cystoid macular edema. Trans Am Acad Ophthalmol 73:665, 1969 134. Kaliner M, Eggleston PA, Mathews KP: Rhinitis and asthma. JAMA 258:2851, 1987 135. Leonardi A, Busato F, Fregona I, et al: Anti-inflammatory and antiallergic effects of ketorolac in the conjunctival
provocation model. Br J Ophthalmol 84:1228, 2000 136. Okazaki T, Reisman RE, Arbesman CE: Prostaglandin E in the secretions of allergic rhinitis. Prostaglandins 13:681, 1977 137. Dhir SP, Garg SK, Sharma YR, et al: Prostaglandins in human tears. Am J Ophthalmol 87:403, 1979 138. Bhattacherjee P: The role of arachidonate metabolites in ocular inflammation. Prog Clin Biol Res 312:211, 1989 139. Ballas Z, Blumenthal M, Tinkelman D, et al: Clinical evaluation of ketorolac tromethamine 0.5% ophthalmic solution
for the treatment of seasonal allergic conjunctivitis. Surv Ophthalmol 38(suppl):141, 1993 140. Tinkelman D, Rupp G, Kaufman H, et al: Ketorolac tromethamine 0.5% ophthalmic solution in treatment of
seasonal allergic conjunctivitis. Surv Ophthalmol 38(suppl):133, 1993 141. Bazan HEP: The synthesis and effects of eicosanoids in avascular ocular tissues. In Bito LZ, Stjernschantz J, eds. Ocular Effects of Prostaglandins and Other Eicosanoids. New York: Alan R. Liss, 1989:73–84 142. Drews R, Knights K: Postulated reactive metabolites of NSAIDs. Agents Actions 17(suppl):127, 1985 143. Ferreira SH, Moncada S, Vane JR: Prostaglandins and the mechanism of analgesia, produced by aspirin-like
drugs. Br J Pharmacol 49:86, 1973 144. Capitola JR, Rosenthal ME, Dubinski B, et al: Peripheral analgesics. A review. J Clin Pharmacol 23:545, 1983 145. Frucht-Pery J, Levinger S, Zaukerman H: The effect of topical administration of indomethacin on symptoms in corneal
scars and edema. Am J Ophthalmol 112:186, 1991 146. Guzek JP, Holm M, Cotter JB, et al: Risk factors for intraoperative complications in 1000 extracapsular cataract
cases. Ophthalmology 94:461, 1987 147. Summary Basis of Approval for Ocufen (Allergan's flurbiprofen) subsequent
to New Drug Application 19-404. Washington, DC, Department
of Health & Human Services, Food and Drug Administration, 1987 148. Summary Basis of Approval for Profenal (Alcon's suprofen) subsequent
to New Drug Application 19-387. Washington, DC, Department
of Health & Human Services, Food and Drug Administration, 1989 149. Keates RH, McGowan KA: Clinical trial of flurbiprofen to maintain pupillary dilation during cataract
surgery. Ann Ophthalmol 16:919, 1984 150. Stark WJ, Fagadau WR, Stewart RH: Reduction of pupillary constriction during cataract surgery using suprofen. Arch Ophthalmol 104:364, 1986 151. Flach AJ: Cyclo-oxygenase inhibitors in ophthalmology. Surv Ophthalmol 36:259, 1992 152. Solomon KD, Turkalj JW, Whiteside SB, et al: Topical 0.5% ketorolac vs 0.03% flurbiprofen for inhibition
of miosis during cataract surgery. Arch Ophthalmol 115:1119, 1997 153. Keates RH, McGowan KA: The effect of topical indomethacin in maintaining mydriasis during cataract
surgery. Ann Ophthalmol 16:1116, 1984 154. Keulen-DeVos HC, van Rij JCG, deLavalette J, et al: Effect of indomethacin in preventing surgically induced miosis. Br J Ophthalmol 67:94, 1983 155. Gimbel H, Van Westenbrugge J, Chetham JK, et al: Intraocular availability and pupillary effect of flurbiprofen and indomethacin
during cataract surgery. J Cataract Refract Surg 22:474, 1996 156. Gimbel HV: The effect of treatment with topical nonsteroidal anti-inflammatory
drugs with and without intraoperative epinephrine on the maintenance
of pupillary mydriasis during cataract surgery. Ophthalmology 96:585, 1989 157. Psilas K, Kalogeropoulos C, Loucatzicos E, et al: The effect of indomethacin, diclofenac, and flurbiprofen on the maintenance
of mydriasis during extracapsular cataract extraction. Doc Ophthalmol 81:293, 1992 158. Sachdev MS, Mehta MR, Dada VK, et al: Pupillary dilatation during cataract surgery-relative efficacy of
indomethacin and flurbiprofen. Ophthalmic Surg 21:557, 1990 159. Bito LZ: Surgical miosis: Have we been misled by a bunch of rabbits? Ophthalmology 97:1, 1990 160. Katz IM: Indomethacin. Ophthalmology 88:455, 1987 161. Sawa M, Masuda K: Topical indomethacin in soft cataract aspiration. Jpn J Ophthalmol 20:514, 1976 162. Roberts CW: Pretreatment with topical diclofenac sodium to decrease postoperative inflammation. Ophthalmology 103:636, 1996 163. Eguchi K, Ohara K, Tobari I, et al: Effects of topical indomethacin on suppression of inflammation after ocular
surgeries—Double masked study of anti-inflammatory effects
on the intracapsular lens extraction. Acta Soc Ophthalmol Jpn 86:2198, 1982 164. Araie M, Sawa M, Takase M: Effect of topical indomethacin on the blood aqueous barrier after intracapsular
extraction of senile cataract—A fluorophotometric study. Jpn J Ophthalmol 25:237, 1981 165. Sanders DR, Kraff MC, Lieberman HL, et al: Breakdown and reestablishment of the blood aqueous barrier following implant
surgery. Arch Ophthalmol 100:588, 1982 166. Garner A, Klintworth GK, eds: Pathobiology of Ocular Disease. New York: Marcel Dekker, 1982:59–89 167. Swan KC, Hart WM: A comparative study of the effects of Mecholyl, Doryl, Eserine, pilocarpine, atropine, and
epinephrine on the blood-aqueous barriers. Am J Ophthalmol 23:1311, 1940 168. Van Alphen G, Macri FJ: Entrance of fluorescein into the aqueous humor of cat eye. Arch Ophthalmol 75:247, 1966 169. Adler FH: The action of atropine in ocular inflammations. Arch Ophthalmol 55:484, 1926 170. Novack GD, Leopold IH: The blood-aqueous and blood-brain barriers to permeability. Am J Ophthalmol 705:412, 1988 171. Miyake K, Kensaku K, Asakura M, et al: Consensual reactions of human blood-aqueous barrier to implant operations. Arch Ophthalmol 102:558, 1984 172. Albert DM, Diaz-Rohena R: A historical review of sympathetic ophthalmia and its epidemiology. Surv Ophthalmol 34:1, 1989 173. Kwito GM, Shin DH, Ahn RH, et al: Bilateral effects of long-term monocular timolol therapy. Am J Ophthalmol 104:591, 1987 174. Freddo TF, Barsotti M, Bartels SP, et al: Combined aqueous fluorophotometric and tracer localization studies of owl
monkeys with endotoxin induced uveitis. Invest Ophthalmol 30:375, 1989 175. Hara Y, Yuasa T: Disturbances of the blood-ocular barrier in uveitis. Invest Ophthalmol 30:450, 1989 176. Komuro Y, Matsumoto S, Shirato S, et al: A new apparatus for automatic counting of aqueous floaters. Acta Soc Ophthalmol Jpn 89:556, 1985 177. Sawa M, Tsurimka , Tsuru T, et al: New quantitative method to determine protein and cells in aqueous in vivo. Jpn J Ophthalmol 32:132, 1988 178. DeBeijer-Dominicus JA, Van Rij G, DeLavalette JCC: Indomethacin eye drops and the postoperative irritation after extracapsular
cataract extraction combined with intraocular lens implantation. Doc Ophthalmol 56:167, 1983 179. Kraff MC, Martin RG, Neumann AC, et al: Efficacy of diclofenac sodium ophthalmic solution versus placebo in reducing
inflammation following cataract extraction and posterior chamber
lens implantation. J Cataract Refract Surg 20:138, 1994 180. Goa KL, Chrisp P: Ocular diclofenac. Drugs Aging 2:473, 1992 181. Vickers FF, McGuigan LJB, Ford C, et al: The effect of diclofenac sodium ophthalmic on the treatment of postoperative
inflammation [Abstract]. Invest Ophthalmol Vis Sci 32(suppl):793, 1991 182. Ronen S, Rozenman Y, Zylbermann R, et al: Treatment of ocular inflammation with diclofenac: Double blind trial following
cataract surgery. Ann Ophthalmol 17:577, 1985 183. Noonan WD, Samples JR: New drugs in ophthalmology: Diclofenac sodium. J Toxicol Cut Ocular Toxicol 12:265, 1993 184. Flach AJ, Lavelle CJ, Olander KW, et al: The effect of ketorolac 0.5% solution in reducing postsurgical inflammation
following ECCE with IOL: Double-masked, parallel comparison
with vehicle. Ophthalmology 5:1279, 1988 185. Solomon KD, Cheetham JK, DeGryse R, et al: Topical ketorolac tromethamine 0.5% ophthalmic solution in ocular
inflammation after cataract surgery. Ophthalmology 108:331, 2001. 186. Heier J, Cheetham JK, DeGryse R, et al: Ketorolac tromethamine 0.5% ophthalmic solution in the treatment
of moderate to severe ocular inflammation after cataract surgery: A randomized, vehicle-controlled clinical trial. Am J Ophthalmol 127:253, 1999 187. Flach AJ, Dolan BJ, Donahue ME, et al: Comparative effects of ketorolac 0.5% or diclofenac 0.1% ophthalmic
solutions on inflammation after cataract surgery. Ophthalmology 105:1775, 1998 188. Sabiston MB, Tessler D, Sumersk H, et al: Reduction of inflammation following cataract surgery by flurbiprofen. Ophthalmic Surg 18:873, 1987 189. Diestelhorst M, Aspacher F, Konen W, et al: The effect of flurbiprofen 0.03% eye drops on the blood aqueous
barrier in extracapsular cataract extraction with IOL implantation. Int Ophthalmol 15:69, 1991 190. Blaydes JE, Kelley EP, Walt JG, et al: Flurbiprofen 0.03% for the control of inflammation following cataract
extraction by phacoemulsification. J Cataract Refract Surg 22:481, 1993 191. Rossetti L, Bujtar E, Castoldi D, et al: Effectiveness of diclofenac eyedrops in reducing inflammation and the incidence
of cystoid macular edema after cataract surgery. J Cataract Refract Surg 22:794, 1996 192. Italian Diclofenac Study Group. Efficacy of diclofenac eyedrops in preventing postoperative inflammation
and long-term cystoid macular edema. J Cataract Refract Surg 23:1183, 1997 193. Kraff MC, Sanders DR, McGuigan L, et al: Inhibition of blood-aqueous humor barrier breakdown with diclofenac: A
fluorophotometric study. Arch Ophthalmol 108:380, 1990 194. Alpar JJ, McCuigan L, Alpar AJ, et al: Effect of diclofenac sodium ophthalmic 0.1% on inflammation following
cataract extraction: A fluorophotometry study. Ophthalmology 96(suppl):109, 1989 195. Flach AJ, Jaffe NS, Akers WA, et al: The effect of ketorolac 0.5% solution in reducing postoperative
inflammation: Double mask parallel comparison with dexamethasone phosphate 0.1% solution. Ann Ophthalmol 21:407, 1989 196. Flach AJ, Kraff MC, Sanders DR, et al: The quantitative effect of 0.5% ketorolac tromethamine solution
and dexamethasone phosphate 0.1% solution on postsurgical blood
aqueous barrier. Arch Ophthalmol 106:480, 1988 197. Othenin-Girard P, Tritten J, Pittet N, et al: Dexamethasone versus diclofenac sodium eyedrops to treat inflammation after
cataract surgery. J Cataract Refract Surg 20:9, 1994 198. Roberts CW, Brennan K: A comparison of topical diclofenac with prednisolone for postoperative
inflammation after cataract surgery. Arch Ophthalmol 113:725, 1995 199. El-Harazi SM, Ruiz RS, Feldman RM, et al: A randomized double-masked trial comparing ketorolac tromethamine 0.5%, diclofenac
sodium 0.1%, and prednisolone acetate 1% in
reducing post-phacoemulsification flare and cells. Ophthalmic Surg Lasers 29:539, 1998 200. Simone JN, Pendelton RA, Jenkins JE: Comparison of the efficacy and safety of ketorolac tromethamine 0.5% and
prednisolone acetate 1% after cataract surgery. J Cataract Refract Surg 25:699, 1999 201. Ostrov CS, Sirkin SR, Deutsch WE, et al: Ketorolac, prednisolone and dexamethasone for postoperative inflammation. Clin Ther 19:259, 1997 202. Diestelhorst M, Aspacher F, Konen W, et al: Effect of dexamethasone 0.1% and prednisolone acetate 1% eye
drops on the blood-aqueous barrier after cataract surgery: A
controlled randomized fluorophotometric study. Graefe's Arch Clin Exp Ophthalmol 230:451, 1992 203. Diestelhorst M, Schmidl B, Konen W, et al: Efficacy and tolerance of diclofenac sodium 0.1%, flurbiprofen 0.03%, and
indomethacin 1.)% in controlling postoperative
inflammation. J Cataract Refract Surg 22:788, 1996 204. Samiy N and Foster CS: The role of nonsteroidal anti-inflammatory drugs in ocular inflammation. Int Ophthalmol Clin 36:195, 1996 205. Hotchkiss IL, Robin AL, Pollack IP, et al: Nonsteroidal anti-inflammatory agents after argon laser trabeculoplasty: Flurbiprofen
and indomethacin. Ophthalmology 91:969, 1984 206. Levine SB, Leopold IH: Advances in ocular corticosteroid therapy. Med Clin North Am 57:1167, 1973 207. Burde RM, Waltman SR: Topical corticosteroids after cataract surgery. Ann Ophthalmol 4:290, 1972 208. Corby JM: Corticosteroid therapy for the reduction of postoperative inflammation
after cataract extraction. Am J Ophthalmol 82:923, 1976 209. Diamond JG, Kaplan HJ: Lensectomy and vitrectomy for complicated cataract secondary to uveitis. Arch Ophthalmol 96:1798, 1978 210. Huang K: Indomethacin inhibition of prostaglandin mediated inflammation following
intraocular surgery. Invest Ophthalmol 16:760, 1977 211. Kaplan HJ, Diamond JG, Brown SA: Vitrectomy in experimental uveitis. Arch Ophthalmol 97:336, 1979 212. Knopf MM: A double-blind study of fluorometholone. Am J Ophthalmol 69:739, 1970 213. Mustaka A, Kaufman HE, Johnston G, et al: Corticosteroid efficacy in postoperative uveitis. Ann Ophthalmol 6:719, 1993 214. Assil KK, Massry G, Lehmann R, et al: Control of ocular inflammation after cataract extraction with rimexolone 1% ophthalmic
suspension. J Cataract Refract Surg 23:750, 1997 215. Hoskins HD, Hetherington J, Minckler DS, et al: Complications of laser trabeculoplasty. Ophthalmology 90:796, 1983 216. Hurvitz LM, Spaeth GL, Zakhour I, et al: Comparison of the effect of flurbiprofen, dexamethasone and placebo on
cyclocryotherapy-induced inflammation. Ophthalmic Surg 15:394, 1984 217. Mermond A, Pittet N, Herbort CP: Inflammation patterns after laser trabeculoplasty measured with the laser
flare meter. Arch Ophthalmol 110:368, 1992 218. Herbort CP, Mermond A, Schnyder C, et al: Anti-inflammatory effect of diclofenac drops after argon laser trabeculoplasty. Arch Ophthalmol 111:481, 1993 219. Goethals M, The Belgian Study Group on Glaucoma: Efficacy and safety of indomethacin 0.1% versus flurbiprofen 0.03% eyedrops
in inflammation after argon laser trabeculoplasty. Documenta Ophthalmologica 85:287, 1994 220. Diestelhorst M, Thull D and Krieglstein GK: The effect of argon laser trabeculoplasty on the blood-aqueous barrier
and intraocular pressure in human glaucomatous eyes treated with
diclofenac 0.1%. Graefe's Arch Clin Exp Ophthalmol 233:559, 1995 221. Cox SN, Hay E, Bird AC: Treatment of chronic macular edema with acetazolamide. Arch Ophthalmol 106:1190, 1988 222. Jampol LM, Sanders DR, Kraff MC: Prophylaxis and therapy of aphakic cystoid macular edema. Surv Ophthalmol 28(suppl):535, 1984 223. Jacobson DR, Dellaporta A: Natural history of cystoid macular edema after cataract extraction. Am J Ophthalmol 77:445, 1974 224. Drolsum L, Haaskjold E: Causes of decreased visual acuity after cataract extraction. J Cataract Refract Surg 21:59, 1995 225. Henkind P, ed: The First International Cystoid Macular Edema Symposium. Surv Ophthalmol 28(suppl):431, 1984 226. Jampol LM: Pharmacologic therapy of aphakic and pseudophakic cystoid macular edema: 1985 update. Ophthalmology 92:807, 1985 227. Jampol LM: Pharmacologic therapy of aphakic cystoid macular edema: A review. Ophthalmology 89:891, 1982 228. Miyake K: Indomethacin in the treatment of postoperative cystoid macular edema. Surv Ophthalmol 78(suppl):554, 1984 229. Rossetti L, Chaudhuri J, Dickersin K: Medical prophylaxis and treatment of cystoid macular edema after cataract
surgery. Ophthalmology 105:397, 1998 230. Koay P: The emerging roles of topical non-steroidal anti-inflammatory
agents in ophthalmology. Br J Ophthalmol 80:480, 1996 231. Rocha G, Deschenes J: Pathophysiology and treatment of cystoid macular edema. Can J Ophthalmol 31:282, 1996 232. Fechner PU: Die prophylaxe des zystoiden Makulaodems mit indomethacin–Augentropfen. Klin Monatsbl Augenheilkd 180:169, 1980 233. Hollwich F, Jocobi K, Kuchle HJ, et al: Zur prophylaxe des zystoiden makulaodems mit Indomethacin-Augentropfen. Klin Monatsbl Augenheilkd 183:477, 1983 234. Klein RM, Katzin HM, Yannuzzi LA: The effect of indomethacin pretreatment on aphakic cystoid macular edema. Am J Ophthalmol 87:487, 1979 235. Kraff MC, Sanders DR, Jampol LM, et al: Prophylaxis of pseudophakic cystoid macular edema with topical indomethacin. Ophthalmology 89:885, 1982 236. Miyake K: Prevention of cystoid macular edema after lens extraction by topical indomethacin. II. A
control study in bilateral extractions. Jpn J Ophthalmol 2:80, 1978 237. Miyake K: Prevention of cystoid macular edema after lens extraction by topical indomethacin. Albrecht von Graefes Arch Klin Ophthalmol 203:81, 1977 238. Miyake K, Miyake Y, Makubo K, et al: Incidence of cystoid macular edema after retinal detachment surgery and
the use of topical indomethacin. Am J Ophthalmol 95:451, 1983 239. Miyake K, Sakamura S, Miura H: Long-term follow-up study on the prevention of aphakic cystoid
macular edema by topical indomethacin. Br J Ophthalmol 64:324, 1980 240. Sholiton DB, Reinhart WJ, Frank KE: Indomethacin as a means of preventing cystoid macular edema following intracapsular
cataract surgery. Am Intraocular Implant Soc J 5:137, 1979 241. Tanake N, Saga Y, Hara K: Effect of topical indomethacin in treatment for cystoid macular edema after
lens extraction. Folia Ophthalmol Jpn 34:178, 1983 242. Tennant JL: Is cystoid macular edema reversible by oral indomethacin, yes or no? In Emery JM, Paton D, eds. Current Concepts in Cataract Surgery. Selected Proceedings of the Fourth
Biannual Cataract Surgical Congress. St. Louis: CV Mosby, 1976:310–312 243. Yannuzzi LA, Landau AN, Turtz AL: Incidence of aphakic cystoid macular edema with the use of topical indomethacin. Ophthalmology 88:947, 1981 244. Yannuzzi LA, Wallyn RH: Cystoid macular edema. A controlled treatment study utilizing indomethacin. In Emery JM, Paton D, eds. Current Concepts in Cataract Surgery. Selected Proceedings of the Fourth
Biannual Cataract Surgical Congress. St. Louis: CV Mosby, 1976:313–316 245. Mizuno K, Miyake K: Pre- and postoperative therapy in cataract extraction. Ophthalmic Surg 18:143, 1987 246. Solomon LD: Flurbiprofen-CME Study Group I. Efficacy of topical flurbiprofen
and indomethacin in preventing pseudophakic cystoid macular edema. J Cataract Refract Surg 21:73, 1995 247. Ibanez HE, Lesher MP, Singerman LJ, et al: Prospective evaluation of the effect of pseudophakic cystoid macula edema
on contrast sensitivity. Arch Ophthalmol 111:1635, 1993 248. Waitzman MB: Topical indomethacin in treatment and prevention of intraocular inflammation. Ann Ophthalmol 11:489, 1979 249. Heier JS, Topping TM, Baumann W, et al: Ketorolac versus prednisolone versus combination therapy in the treatment
of acute pseudophakic cystoid macular edema. Ophthalmology 107:2034, 2000 250. Yannuzzi LA, Klein RM, Wallyn RH: Ineffectiveness of indomethacin in the treatment of chronic cystoid macular
edema. Am J Ophthalmol 84:517, 1977 251. Burnett J, Tessler H, Isenberg S, et al: Double-masked trial of fenoprofen sodium: Treatment of chronic aphakic
cystoid macular edema. Ophthalmic Surg 14:150, 1983 252. Flach AJ, Dolan BJ, Irvine AR: Effectiveness of ketorolac 0.5% solution for chronic aphakic and
pseudophakic cystoid macular edema. Am J Ophthalmol 103:479, 1987 253. Flach AJ, Jampol LM, Yannuzzi LA: Improvement in visual acuity in chronic aphakic and pseudophakic cystoid
macular edema after treatment with topical 0.5% ketorolac tromethamine. Am J Ophthalmol 112:514, 1991 254. Epstein DL: Cystoid macular edema occurring 13 years after cataract extraction. Am J Ophthalmol 83:501, 1977 255. Weisz JM, Bressler NM, Bressler SB, et al: Ketorolac treatment of pseudophakic cystoid macular edema identified more
than 24 months after cataract extraction. Ophthalmology 106:1656, 1999 256. Severin SL: Late cystoid macular edema in pseudophakia. Am J Ophthalmol 90:223, 1980 257. Flach AJ: Discussion of Ketorolac versus prednisolone versus combination therapy
in the treatment of acute pseudophakic cystoid macular edema. Ophthalmology 107:2039, 2000 258. Smith RT, Campbell CJ, Koester CJ: The barrier function in extracapsular cataract surgery. Ophthalmology 97:90, 1990 259. Sorr EM, Everett WH, Hurite FC: Incidence of fluorescein angiographic subclinical macular edema following
phacoemulsification of senile cataracts. Ophthalmology 86:201, 1979 260. Wetzig PC, Thatcher DB, Christiansen JM: The intracapsular versus extracapsular cataract technique in relationship
to retinal problems. Trans Am Ophthalmol Soc 77:339, 1979 261. Gilbard SM, Peyman GA, Goldberg MF: Evaluation for cystoid maculopathy after pars plicata lensectomy-vitrectomy
for congenital cataracts. Ophthalmology 90:1201, 1983 262. Hoyt CS, Nickel B: Aphakic cystoid macular edema: Occurrence in infants and children after
transpupillary lensectomy and anterior vitrectomy. Arch Ophthalmol 100:746, 1982 263. Poer DV, Helveston EM, Ellis FD: Aphakic cystoid macular edema in children. Arch Ophthalmol 99:249, 1981 264. Stern AL, Taylor DM, Dalburg LA, et al: Pseudophakic cystoid maculopathy—Study of 50 cases. Ophthalmology 88:942, 1981 265. Federman JL, Annesley WH Jr, Sarin LK: Vitrectomy and cystoid macular edema. Ophthalmology 87:622, 1980 266. Fung WE: Surgical therapy for chronic aphakic cystoid macular edema. Ophthalmology 89:898, 1982 267. Fung WE: Anterior vitrectomy for chronic aphakic cystoid macular edema. Ophthalmology 87:189, 1980 268. Katzen LY, Fleischman J, Trokel S: YAG laser treatment of cystoid macular edema. Am J Ophthalmol 95:589, 1983 269. Pfoff DS, Thom SR: Cystoid macular edema treated with hyperbaric oxygen. Ophthalmology 97(suppl):120, 1990 270. Bito LZ, Nichols R, Siminoff S: Ocular effects of substance P and other biologically active peptides as
compared to prostaglandins E2, (PGE2). Proc Int Soc Eye Res 1:53, 1980 271. Pendergast SD, Marcherio RR, Williams GA, et al: Vitrectomy for chronic pseudophakic cystoid macular edema. Am J Ophthalmol 128:317, 1999 272. Wilkinson CP: Vitrectomy for chronic pseudophakic cystoid macular edema. Am J Ophthalmol 129:560, 2000. 273. Peyman GA, Canakis C, Livir-Rallatos C, et al: The effect of internal limiting membrane peeling on chronic recalcitrant
pseudophakic cystoid macular edema: A report of two cases. Am J Ophthalmol 133:571, 2002 274. Kobayashi H, Kobayashi K, Okinami S: Randomized clinical trial of topical betaxolol for persistent macular edema
after vitrectomy and epiretinal membrane removal. Am J Ophthalmol 136:244, 2003 275. Tach AB, Dugel PU, Flindall RJ, et al: A comparison of retrobulbar versus sub-tenon's corticosteroid
therapy for cystoid macular edema refractory to topical medications. Ophthalmology 104:2003, 1997 276. Abe T, Hayasaka S, Nagaki Y, et al: Pseudophakic cystoid macular edema treated with high-dose intravenous
methylprednisolone. J Cataract Refract Surg 25:1286, 1999 277. Benhamou N, Massin P, Haouchine B, et al: Intravitreal triamcinolone for refractory pseudophakic macular edema. Am J Ophthalmol 135:246, 2003 278. Jonas JB, Kreissig I, Degenring RF: Intravitreal triamcinolone acetonide for pseudophakic cystoid macular edema. Am J Ophthalmol 136:384, 2003 279. Jonas JB, Kreissig I, Degenring RF: Secondary chronic open-angle glaucoma after intravitreal triamcinolone
acetonide. Arch Ophthalmol 121:729, 2003 280. Friedlaender MH: Conjunctivitis of allergic origin: Clinical presentation and differential
diagnosis. Surv Ophthalmol 38(suppl):105, 1993 281. Jackson WB: Differentiating conjunctivitis of diverse origins. Surv Ophthalmol 38(suppl):91, 1993 282. Leopold IH: Advances in ocular therapy noncorticosteroid anti-inflammatory agents. Am J Ophthalmol 78:759, 1974 283. Greaves MW, McDonald W: Itch: Role of prostaglandins. Br Med J 22:608, 1973 284. Pakes GE, Brogden RN, Heel RD, et al: Flunisolide: A review. Drugs 19:397, 1980 285. Meyer E, Kraus E, Zonis S: Efficacy of antiprostaglandin therapy in vernal conjunctivitis. Br J Ophthalmol 71:497, 1987 286. Laibovitz RA, Koester J, Schaich L, et al: Safety and efficacy of diclofenac sodium 0.1% ophthtalmic solution
in acute seasonal allergic conjunctivitis. J Ocular Pharm Ther 11:361, 1995 287. Lewis RA, Roberts LJ, Holgate ST, et al: Immunologic generation of mast cell prostaglandins. J Allergy Clin Immunol 65:235, 1980 288. Gupta S, Khurang AK, Ahluwalia BK, et al: Topical indomethacin for vernal conjunctivitis. Acta Ophthalmol (Copenh) 69:95, 1991 289. Brooks CD, Nelson AL, Metzler C: Effect of flurbiprofen on induced allergic rhinitis. J Allergy Clin Immunol 73:584, 1984 290. Brooks CD, Karl KJ: Hay fever treatment in the combined antihistamine and cyclooxygenase-inhibiting
drugs. J Allergy Clin Immunol 81:1110, 1988 291. Brooks CD, Nelson AL, Metzler C: Treatment of ragweed hay fever with flurbiprofen. Ann Allergy 55:557, 1985 292. Abelson MB, Schaefer K: Conjunctivitis of allergic origin: Immunologic mechanisms and current approaches
to therapy. Surv Ophthalmol 38(suppl):115, 1993 293. Bishop K, Abelson MB, Cheetham J, et al: Evaluation of flurbiprofen in the treatment of antigen-induced allergic
conjunctivitis. Invest Ophthalmol Vis Sci 31:487, 1990 294. Allansmith MR, Ross RN: Ocular allergy and mast cell stabilizers. Surv Ophthalmol 30:229, 1986 295. Wood TS, Stewart RH, Bowman RW, et al: Suprofen treatment of GPC. Ophthalmology 95:822, 1988 296. Seitz B, Sorken K, LaBree LD, et al: Corneal sensitivity and burning sensation-Comparing topical ketorolac
and diclofenac. Arch Ophthalmol 114:921, 1996 297. Zaidman GW: Diclofenac and its effect on corneal sensation. Arch Ophthalmol 113:262, 1995. 298. Szerenyi K, Sorken K, Garbus JJ, et al: Decrease in normal human corneal sensitivity with topical diclofenac sodium. Am J Ophthalmol 118:312, 1994 299. Phillips AF, Szerenyi K, Campos M, et al: Arachidonic acid metabolites after excimer laser corneal surgery. Arch Ophthalmol 111:1273, 1993 300. Eiferman RA, Hoffman RS, Sher NA: Topical diclofenac reduces pain following photorefractive keratectomy. Arch Ophthalmol 111:1022, 1993 301. Gwon A, Vaughan ER, Cheetham JK, et al: Ocufen (flurbiprofen) in the treatment of ocular pain after radial
keratotomy. CLAO J 20:2, 1994 302. Sher NA, Frantz JM, Talley A, et al: Topical diclofenac in the treatment of ocular pain after excimer photorefractive
keratectomy. Refract Corneal Surg 9:425, 1993 303. Cherry PMH, Tutton MK, Adhikary H, et al: The treatment of pain following photorefractive keratectomy. J Cataract Refract Surg 10:S222, 1994 304. Yee RW, The Ketorolac Radial Keratotomy Study Group: Analgesic efficacy and safety of nonpreserved ketorolac tromethamine ophthalmic
solution following radial keratotomy. Am J Ophthalmol 125:472, 1998 305. Stein R, Stein H, Cheskes A, et al: Photorefractive keratectomy and postoperative pain. Am J Ophthalmol 117:403, 1994 306. Arshinoff S, D'Addario D, Sadler C, et al: Use of topical nonsteroidal anti-inflammatory drugs in excimer laser
photorefractive keratectomy. J Cataract Refract Surg 20:216, 1994 307. Maguen E, Salz JJ, Nesburn AB, et al: Results of excimer laser photorefractive keratectomy for the correction
of myopia. Ophthalmology 101:1548, 1994 308. Epstein RL, Laurence EP: Effect of topical diclofenac solution on discomfort after radial keratotomy. J Cataract Refract Surg 20:378, 1994 309. Sher NA, Frantz JM, Talley A, et al: Topical diclofenac in the treatment of ocular pain after excimer photorefractive
keratectomy. Refract Corneal Surg 9:425, 1993 310. Epstein RL, Laurence EP: Relative effectiveness of topical ketorolac and topical diclofenac on discomfort
after radial keratotomy. J Cataract Refract Surg 21:156, 1995 311. Kaiser PK, Pineda R, Corneal Abrasion Patching Study Group: A study of topical nonsteroidal anti-inflammatory drops and no pressure
patching in the treatment of corneal abrasions. Ophthalmology 104:1353, 1997 312. Donnenfeld ED, Selkin BA, Perry HD, et al: Controlled evaluation of a bandage contact lens and a topical nonsteroidal
anti-inflammatory drug in treating traumatic corneal abrasions. Ophthalmology 102:9790, 1995 313. Salz JJ, Reader AL, Schwartz LJ, et al: Treatment of corneal abrasions with soft contact lenses and topical diclofenac. J Refract Corneal Surg 10:640, 1994 314. Verma S, Corbett MC, Marshall J: A prospective, randomized double-masked trial to evaluate the role
of topical anesthetics in controlling pain after photorefractive keratectomy. Ophthalmology 102:1918, 1995 315. Rosenwasser GOD, Holland S, Pflugfelder SC, et al: Topical anesthetic abuse. Ophthalmology 97:967, 1990 316. Dornic DI, Thomas JM, Lass JH: Topical diclofenac sodium in the management of anesthetic abuse keratopathy. Am J Ophthalmol 125:719, 1998 317. Fry LL: Efficacy of diclofenac sodium solution in reducing discomfort after cataract
surgery. J Cataract Refract Surg 21:187, 1995 318. Apt L, Voo I, Isenberg SJ: A randomized clinical trial of the nonsteroidal eyedrop diclofenac after
strabismus surgery. Ophthalmology 105:1448, 1998 319. Morton WS, Benham SW, Lawson RA, et al: Diclofenac vs oxybuprocaine eye drops for analgesia in pediatric strabismus
surgery. Paediatr Anaesth 7:221, 1997 320. Apple DJ, Solomon KD, Tetz MR, et al: Posterior capsule opacification. Surv Ophthalmol 37:73, 1992 321. Nishi O, Nishi K, Fujiwara T, et al: Effects of diclofenac sodium and indomethacin on proliferation and collagen
synthesis of lens epithelial cells in vitro. J Cataract Refract Surg 21:461, 1995 322. Flach AJ: Incidence of postoperative posterior capsular opacification following treatment
with diclofenac 0.1% and ketorolac 0.5% ophthalmic
solutions: 3-year randomized, double-masked, prospective
clinical investigation. Trans Am Ophth Soc 98:101, 2000 323. Snir M, Axer-Siegel R, Friling R: Efficacy of diclofenac versus dexamethasone for treatment after strabismus
surgery. Ophthalmology 107:1884, 2000 324. Apt L, Voo I, Isenberg SJ: A randomized clinical trial of the nonsteroidal eyedrop diclofenac after
strabismus surgery. Ophthalmology 105:1448, 1998 325. Wright M, Butt Z, McIlwaine G, et al: Comparison of the efficacy of diclofenac and betamethasone following strabismus
surgery. Br J Ophthalmol 81:299, 1997 326. Morton NS, Benham SW, Lawson RA, et al: Diclofenac vs oxybuprocaine eye drops for analgesia in paediatric strabismus
surgery. Paediatr Anesth 7:221, 1997 327. Gordon YJ, Araullo-Cruz T, Romanowski EG: The effects of topical nonsteroidal anti-inflammatory drugs on adenoviral
replication. Arch Ophthalmol 116:900, 1998 328. Shiuey Y, Ambati BK, Adamis AP, et al: A randomized, double-masked trial of topical ketorolac versus artificial
tears for treatment of viral conjunctivitis. Ophthalmology 107:1512, 2000 329. Gartry DS, Kerr Muir MG, Marshall J: Excimer laser photorefractive keratectomy: 18-month follow-up. Ophthalmology 99:1209, 1992 330. Tuft Sj, Gartry DS, Rawe IM, et al: Photorefractive keratectomy: implications of corneal wound healing. Br J Ophthalmol 77:243, 1993 331. Gartry DS, Kerr Muir MG, Marshall J: The effect of topical corticosteroids on refraction and corneal haze following
excimer laser treatment of myopia: an update. Eye 7:584, 1993 332. Sher NA, Krueger RR, Teal P, et al: Role of topical steroids, corticosteroids and nonsteroidal anti-inflammatory
drugs in the etiology of stromal infiltrates after excimer
photorefractive keratectomy. J Refract Corneal Surg 10:587, 1994 333. Nassaralla BA, Szerenyi K, Wang XW, et al: Effect of diclofenac on corneal haze after photorefractive keratectomy
in rabbits. Ophthalmology 102:469, 1995 334. O'Brart DPS, Lohmann CP, Klonos G, et al: The effects of topical corticosteroids and plasmin inhibitors on refractive
outcome, haze, and visual performance after photorefractive keratectomy. Ophthalmology 101:1565, 1994 335. Talamo JH, Gollamudi S, Green WR, et al: Modulation of corneal wound healing after excimer laser keratomileusis
using topical mitomycin C and steroids. Arch Ophthalmol 109:1141, 1991 336. Morlet N, Gillies MC, Crouch R, et al: Effect of topical interferon alpha 2 beta on corneal haze after excimer
laser photorefractive keratectomy in rabbits. Refract Corneal Surg 9:443, 1993 337. Schaumberg DA, Sullivan DA, Dana MR: Epidemiology of dry eye syndrome. Adv Exp Med Biol 506:989, 2002 338. Avunduk AM, Avunduk MC, Varnell ED, et al: The comparison of efficacies of topical corticosteroids and nonsteroidal
anti-inflammatory drops on dry eye patients. Am J Ophthalmol 136:593, 2003 339. Chan CKM, Lam DSC: The comparison of efficacies of corticosteroids and nonsteroidal anti-inflammatory
drops on dry eye patients. Am J Ophthalmol 137:1157, 2004 340. Sall K, Stevenson OD, Mundorf , et al: Two multicenter, randomized studies of the efficacy and safety of cyclosporine
ophthalmic emulsion in moderate to severe dry eye disease. Ophthalmology 107:631, 2000 341. Jaros PA, DeLuise VP: Pingueculae and pterygia. Surv Ophthalmol 33:41, 1988 342. FruchtPery J, Siganos CS, Solomon A, et al: Topical indomethacin solution versus dexamethasone solution for treatment
of inflamed pterygium and pinquecula: A prospective randomized clinical
study. Am J Ophthalmol 127:148, 1999 343. Manthey KF: Nonsteroidal anti-inflammatory drugs in the treatment of intermediate
uveitis. Dev Ophthalmol 23:158, 1992 344. Maza MS, Jabbur NS, Foster CS: An analysis of therapeutic decision for scleritis. Ophthalmology 100:1372, 1993 345. Perianin A, Gougerot-Pocidalo ME, Giroud JP, et al: Diclofenac: A negative chemokinetic factor for neutrophil locomotion. Biochem Pharmacol 34:3433, 1985 346. Van Husen H: Lokale Behandlung mit Diclofenac Augentropfen bei Erkrankungen der vorderen
Augenabschmitte. Klin Monatsbl Augenheilkd 188:615, 1986 347. Harth M: Rare miscellaneous adverse drug reactions and interactions of nonsteroidal
anti-inflammatory drugs. In Borda T, Koff RS, eds. NSAIDs—A Profile of Adverse Effects. Philadelphia: Hanley and Belfur, 1992:219–231 348. Brooks PM: Side effects of non-steroidal anti-inflammatory drugs. Med J Aust 148:248, 1988 349. Calen A: In common clinical usage nonsteroidal anti-inflammatory drugs infrequently
produce adverse effects on the kidney. Am J Kidney Dis 11:485, 1983 350. Heinrich WI: Nephrotoxicity of nonsteroidal anti-inflammatory agents. Am J Kidney Dis 11:485, 1983 351. Koch-Weser J: Non-steroidal anti-inflammatory drugs. N Engl J Med 302:1179, 1980 352. Masuda K: Anti-inflammatory agents: Nonsteroidal anti-inflammatory
drugs. In Sears ML, ed. Pharmacology of the Eye. New York: Springer-Verlag, 1984:539–551 353. O'Brien WM: Pharmacology of nonsteroidal anti-inflammatory drugs. Am J Med 10:32, 1983 354. Stevenson DD: Diagnosis, prevention and treatment of adverse reactions to nonsteroidal
anti-inflammatory drugs. J Allergy Clin Immunol 74:617, 1984 355. Wolfe F: Gastro protective therapy and risk of gastrointestinal ulcers: Risk reduction
by COX-2. J. Rheumatol 29:3, 2002 356. Russell RI: Protective effects of prostaglandin on the gastric mucosa. Am J Med 81(Suppl 2A):2, 1986 357. Simmons TC, Weinstein WM, Shapiro N, et al: A therapeutic trial of 15-methyl prostaglandin E2 in rheumatoid arthritis patients with gastrointestinal lesions. Prostaglandins 21:165, 1981 358. Kivilaakso E, Silen W: Pathogenesis of experimental gastric-mucosal injury. N Engl J Med 301:364, 1979 359. Madhok R, MacKenzie JA, Lee FD: Small bowel ulceration in patients who received NSAIDs for rheumatoid arthritis. Q J Med 58:53, 1986 360. Somerville K, Faulkner G, Langman M: Non-steroidal anti-inflammatory drugs and bleeding ulcer. Lancet 1:462, 1986 361. Collier DSJ, Pain JA: NSAIDs and peptic ulcer perforation. Gut 26:359, 1985 362. Mitchell WS, Sturrock RD: Ulcers and anti-inflammatory agents. Br Med J 284:731, 1982 363. Carthbert MF: Adverse reactions to NSAIDs. Curr Med Res Opin 2:600, 1974 364. Rhymer AR, Gengos DC: Indomethacin. In Husklsson EC, ed. Antirheumatic Drugs. Eastbourne: Praeger, 1983:265–278 365. Collum LMT, Bowen DI: Ocular effects of ibuprofen. Br J Ophthalmol 55:472, 1971 366. Burns CA: Indomethacin reduced retinal sensitivity and corneal deposits. Am J Ophthalmol 66:825, 1968 367. O'Brien WM, Bigby GF: Rare adverse reactions to NSAIDs. J Rheumatol 12:13, 347, 562, 785, 1985 368. Ciabottoni G, Cinott GA, Prerucci A, et al: Effects of NSAIDs in patients with chronic glomerular disease. N Engl J Med 310:279, 1984 369. McGiff JC, Growshaw K, Itskovitz HD: Prostaglandins and renal function. Fed Proc 33:39, 1974 370. Clive DM, Stoff JS: Renal syndromes associated with NSAIDs. N Engl J Med 310:563, 1984 371. Koopmans PP, Thren TH, Thomas CMG, et al: The effects of NSAIDs on the anti-hypertensive and diuretic action
of thiazides in patients with hypertension. Br J Clin Pharmacol 21:417, 1986 372. Prescott LF: Overdose with NSAIDs. Soc Med Int Congress Symp Series 52:21, 1982 373. Warren GV, Korbet SM, Schwartz MM, et al: Minimal change glomerulopathy associated with nonsteroidal anti-inflammatory
drugs. Am J Kidney Dis 13:127, 1989 374. Porile JL, Bakris GL, Garella S: Acute interstitial nephritis with glomerulopathy due to nonsteroidal anti-inflammatory
agents. J Clin Pharmacol 30:468, 1990 375. Radford MG, Holley KE, Grande JP: Reversible membranous nephropathy associated with the use of nonsteroidal
anti-inflammatory drugs. JAMA 276:466, 1996 376. Mielke CH, Kaneshuo MM, Maher IA, et al: The standardized normal vey bleeding time and its prolongation by aspirin. Blood 34:204, 1969 377. Strom BL, Berlin JA, Kinman JL, et al: Parenteral ketorolac and risk of gastrointertinal and operative site bleeding: A
postmarketing surveillance study. JAMA 275:376, 1996 378. Gallager JE, Blauth J, Fornadley JA: Perioperative ketorolac tromethamine and postoperative hemorrhage in cases
of tonsillectomy and adenoidectomy. Laryngoscope 105:606, 1995 379. Strom BL, Berlin JA, Kinman JL: Risk of operative site bleeding with Parenteral ketorolac. JAMA 276:372, 1996 380. Lesko SM, Mitchell AA: An assessment of the safety of pediatric ibuprofen: a practitioner-based
randomized clinical trial. JAMA 273:929, 1995 381. Lesko SM, Mitchell AA: Pediatric ibuprofen and leucopenia. JAMA 275:986, 1996 382. Inman WHW: Study of fatal bone marrow depression. Br Med J 1:1500, 1977 383. Szczeklik A, Gryglewski RJ, Czerniawska-Mysik G: Clinical patterns of hypersensitivity to nonsteroidal anti-inflammatory
drugs and their pathogenesis. J Allergy Clin Immunol 60:276, 1977 384. Benson GD: Hepatotoxicity following therapeutic use of antipruritic analgesics. Am J Med 75:85, 1983 385. Juan J: Prostaglandins as modulators of pain. Gen Pharmacol 9:403, 1978 386. Bigby M, Stern R: Cutaneous reactions to NSAIDs. J Am Acad Dermatol 12:866, 1985 387. Sachs S. Phototoxicity to NSAIDs. N Engl J Med 309:187, 1983 388. Mahoney JM, Waterbury LD: RS 37619: A non-irritating ophthalmic anti-inflammatory agent. Invest Ophthalmol 24:151, 1983 389. Ocufen [product information package insert]. Alcon USA, Ft. Worth, Texas, 1990 390. Profenal [product information package insert]. Alcon USA, Ft. Worth, Texas, 1990 391. Araie M, Sawa M, Takase M: Topical flurbiprofen and diclofenac suppress blood-aqueous barrier
breakdown in cataract surgery: A fluorophotometric study. Jpn J Ophthalmol 27:535, 1983 392. Gannon R: Focus on ketorolac. Hospital Formulary 24:695, 1989 393. Waterbury LD, Flach AJ: Efficacy of low concentrations of ketorolac tromethamine in animal models
of ocular inflammation. J Ocular Pharm Ther 20:345, 2004 394. NSAID Adverse Reaction Report [press release). American Society
of Cataract and Refractive Surgery, Aug 3, 1999 395. NSAID Update [press release]. American Society of Cataract and
Refractive Surgery, American Society of Ophthalmic Administrators, Aug 11, 1999 396. Rosenthal KJ: ASCRS inquiry of NSAID problem leads to drug recall. Ophthalmol Times Nov 15. 1999 397. Gayton JL: Primary management problems symposium: Complications of NSAIDs. Presented
at the 104th Annual Meeting of the American Academy of Ophthalmology, Oct 24, 2000 398. Flach AJ: Topically applied nonsteroidal anti-inflammatory drugs and corneal
problems: An interim review and comment. Ophthalmology 107:1224, 2000 399. Congdon NG, Schein OD, von Kulajta P, et al: Corneal complications associated with topical ophthalmic use of nonsteroidal
anti-inflammatory drugs. J Cataract Refract Surg 27:622, 2001 400. Flach AJ: Corneal melts associated with topically applied nonsteroidal anti-inflammatory
drugs. (Discussion of comments) Tr Am Ophth Soc 99:210, 2001 401. Flach AJ: Corneal melts associated with topically applied nonsteroidal anti-inflammatory
drugs. Tr Am Ophth Soc 99:205, 2001 402. Thygeson P: Controversies in Ophthalmology. Philadelphia, Pa: WB Saunders 450, 1977 403. O'Brien TP, Li QJ, Sauerburger F, et al: The role of matrix metalloproteinases in ulcerative keratolysis associated
with perioperative diclofenac use. Ophthalmology 108:656, 2001 404. Lam S, Beck RW, Hall D, et al: Atonic pupil after cataract surgery. Ophthalmology 96:589, 1989 405. Percival SPB: Results after intracapsular extraction: The atonic pupil. Ophthalmic Surg 8:138, 1977 406. Sitenga GL, Ing EB, Van Dellen RG, et al: Asthma caused by topical application of ketorolac. Ophthalmology 103:890, 1996 407. Davies T, Lederer DA, Spencer AA, et al: The effect of flurbiprofen on platelet function and blood coagulation. Thromb Res 5:667, 1974 408. Brown SI, Weller CA, Vidrich AM: Effect of corticosteroids on corneal collagenase of rabbits. Am J Ophthalmol 70:744, 1970 409. Francois J: Cortisone et tension oculaire. Am Ocul 187:805, 1954 410. Nesburn AB: Immunological aspects of ocular herpes simplex disease. In Suran A, Gery I, Nussenblatt RB, eds. Immunology of the Eye: Workshop III. Washington, DC: Information Retrieval, 1979:21 411. Rooklin AR, Lampert SL, Jaeger EA, et al: Posterior subcapsular cataracts in steroid-requiring asthmatic children. J Allergy Clin Immunol 63:383, 1979 412. Schwartz B: Physiological effects of corticosteroids on the eye. In Schwartz B, ed. Corticosteroids and the Eye. Boston: Little, Brown & Co, 1980:753 413. Schwartz JT, Rueling FH, Feinleib M, et al: Twin study on ocular pressure following topically applied dexamethasone. Arch Ophthalmol 76:281, 1973 414. Skalka HW, Prichard JT: Effect of corticosteroids on cataract formation. Arch Ophthalmol 98:1773, 1980 415. Spaeth CL, Rodrigues MM, Weinreb S: Steroid induced glaucoma. Trans Am Ophthalmol Soc 75:353, 1977 416. Stewart RH, Kimbrough RL: Intraocular pressure response to topically administered fluorometholone. Arch Ophthalmol 97:2139, 1979 417. Urban RC, Cotlier E: Corticosteroid-induced cataracts. Surv Ophthalmol 31:102, 1986 418. Yablonski ME, Burde RM, Kolker AE, et al: Cataracts induced by topical dexamethasone in diabetics. Arch Ophthalmol 96:474, 1978 419. Raizman M: Corticosteroid therapy of eye disease-fifty years later. Arch Ophthalmol 114:1000, 1996. 420. Fan DSP, Ng JSK, Lam DSC: A prospective study on ocular hypertensive and anti-inflammatory
response to different dosages of fluorometholone in children. Ophthalmology 108:12973, 2001 421. Leibowitz HM, Bartlett JD, Rich R: Intraocular pressure-raising potential of 1.0a% rimexolone
in patients responding to corticosteroids. Arch Ophthalmol 114:933, 1996 422. Tan DTH, Chee SP, Lim L, et al: Randomized clinical trial of a new dexamethasone delivery system for treatment
of post-cataract surgery inflammation. Ophthalmology 106:223, 1999 423. Chang DF, Garcia IH, Hunkeler JD, et al: Phase II results of an intraocular steroid delivery system for cataract
surgery. Ophthalmology 106:1172, 1999 424. Gieser DK, Hoda PPE, Goldberg I, et al: Flurbiprofen and intraocular pressure. Ann Ophthalmol 13:831, 1981 425. Stjernschantz J, Sherk T, Sears M: Ocular responses to leukotriene C4 and D4 in the cat. Prostaglandins 27:5, 1984 426. Hersh PS, Rice BA, Baer JC, et al: Topical nonsteroidal agents and corneal wound healing. Arch Ophthalmol 108:577, 1990 427. Shimazaki j, Fujishima H, Yagi Y, et al: Effects of diclofenac eye drops on corneal epithelial structure and function
after small-incision cataract surgery. Ophthalmology 103:50, 1996 428. Fraser-Smith EB, Matthews TR: Effect of ketorolac on herpes simplex virus type one ocular infection in
rabbits. J Ocul Pharmacol 4:321, 1988 429. Fraser-Smith EB, Matthews TR: Effect of ketorolac on Candida albicans ocular infection in rabbits. Arch Ophthalmol 105:264, 1987 430. Hendricks RL, Barfknecht CF, Schoenwald RD, et al: The effect of flurbiprofen on herpes simplex virus type 1 stromal keratitis
in mice. Invest Ophthalmol 31:1503, 1990 431. Klein BE, Klein R, Lee KE, et al: Drug use and five-year incidence of age-related cataracts. Ophthalmology 108:1670, 2001 432. Age-Related Eye Disease Study Research Group: Risk factors associated with age-related nuclear and cortical cataract. Ophthalmology 108:1400, 2001 433. Napoli SA, Helm C, Insler MS, et al: External ocular inflammatory effects of lipoxygenase enzyme products. Ann Ophthalmol 22:30, 1990 434. Palmer RMJ, Stepney RJ, Higg GA, et al: Chemokinetic activity of arachidonic acid lipoxygenase products on leukocytes
of different species. Prostaglandins 20:411, 1980 435. Samuelsson B, Borgeat P, Hammarstrom SJ, et al: Leukotrienes: A new group of biologically active compounds. In Samuelsson B, Ramwell PW, Proletti R, eds. Leukotrienes and Other Lipoxygenase Products. New York: Raven Press, 1980:1–18 436. Rao NA, Patchett R, Fernandez MA, et al: Treatment of experimental granulomatous uveitis by lipoxygenase and cyclo-oxygenase
inhibitors. Arch Ophthalmol 105:413, 1987 437. Moreira H, McDonnell PJ, Fasano AP, et al: Treatment of experimental Pseudomonas keratitis with cyclo-oxygenase
and lipoxygenase inhibitors. Ophthalmology 98:1693, 1991 |