Nystagmus (derived from the Greek word, νμσταγμοσ, meaning drowsiness, is derived from νμσταζειν, meaning “to nod in one's sleep”), the rhythmic to-and-fro oscillation of the eyes, has been regarded as enigmatic. In fact, the distinguished neuro-ophthalmologist Wilbrand once advised “never write on nystagmus, it will lead you nowhere.”1
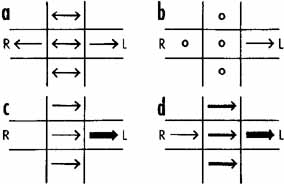
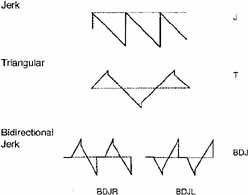
Although technologic advances have permitted quantitative insights into nystagmus analysis, the clinician should not be daunted. Many useful, often diagnostic, observations can be made by physical examination alone. Figures 1 and 2 are examples of one convenient method of diagramming nystagmus. Also, nystagmus can be further described when the globes are inspected under slit-lamp magnification or when the fundus is viewed. However, due to the complexity of nystagmus waveforms and the possibility of combinations of different types of nystagmus, only ocular motility recordings can guarantee diagnosis that is both accurate and repeatable.
|
This chapter is a coalescence of the traditional neuro-ophthalmologic approach to nystagmus diagnosis and the impact of the newer capabilities of electronic eye movement recording and mathematical “biomodeling,” specifically, top-down, behavioral models capable of simulating human ocular motor responses to known target inputs in the presence of nystagmus and saccadic disorders.
Eye movement recordings have allowed definition of 49 types of nystagmus (Table 1) and new insights into their pathophysiology. For precise analysis, special recording techniques are necessary, such as infrared, magnetic search-coil, or high-speed digital video recording systems, which can faithfully reproduce the eye-movement trajectories and provide accurate information on eye position without drift or noise. For quantitative purposes, all systems should record by way of direct current, with a bandwidth of 100 Hz. The eyes should be recorded separately in horizontal, vertical, and (if possible) torsional directions, with the tracing analogs written on rectilinear graph paper. Recording should be performed during fixation of visible targets and sometimes in the dark with eyes open (see Chapter 9). For detailed quantitative analysis, the data should be digitized at 200 Hz or higher.
TABLE 1. Forty-nine Types of Nystagmus*
| Acquired | Gaze-evoked | Pursuit-defect1 |
| “fixation” | Deviational | Pursuit-system |
| Anticipatory | Gaze-paretic | Infantile |
| Induced | “Neurasthenic” | Pseudospontaneous |
| Arthrokinetic | “Seducible” | Induced |
| Induced | “Setting-in” | Rebound |
| Somatosensory | Horizontal | Reflex |
| Associated | Induced | Baer's |
| Induced | Provoked | See-saw |
| Stransky's | Infantile | Somatosensory |
| Audiokinetic | Congenital | Induced |
| Induced | “Fixation” | Spontaneous |
| Bartels' | Hereditary | Stepping around |
| Induced | Pursuit-system | Apparent/real |
| Bruns' | Intermittent vertical | Induced |
| Centripetal | Jerk | Somatosensory |
| Cervical | Lateral medullary | Torsional |
| Neck torsion | Lid | Rotary |
| Vertebral-basilar artery insufficiency | Miner's1 | Uniocular |
| Circular/Elliptic/Oblique | Occupational | Upbeat |
| Alternating windmill | Muscle-paretic | Vertical |
| Circumduction | Myasthenic | Vestibular |
| Diagonal | Nucleus of the optic tract | A(po)geotropic/geotropic |
| Elliptic | Optokinetic | Alternating current |
| Gyratory | Induced | Bechterew's |
| Oblique | “Kinetic” | Caloric/caloric-after |
| Radiary | “Optic” | Compensatory |
| Convergence | Optomotor | Electrical/faradic/galvanic |
| Convergence-evoked | Panoramic | Head-shaking |
| Dissociated | “Railway” | Induced |
| Disjunctive | Sigma | L- |
| Downbeat | “Train” | Labyrinthine |
| Drug-induced | Optokinetic after- | Perverted |
| Barbiturate | Induced | Pneumatic/compression |
| Bow tie | Post-optokinetic | Positional/alcohol |
| Induced | Reverse post-optokinetic | Positioning |
| Epileptic | Pendular | Pseudocaloric |
| Ictal | Talantropia | Rotational/perrotary |
| Fusion maldevelopment | Periodic/Aperiodic alternating | Secondary phase |
| Latent/manifest latent | Alternans | |
| Monocular “fixation” | Physiologic | |
| Unimacular | End-point | |
| Flash-induced | Fatigue | |
| Flicker-induced | Pursuit after- | |
| Induced | Induced |
*Synonyms and other terms are indented under either the preferred or the more inclusive designation; some nystagmus types may be acquired or congenital; quoted terms are erroneous or nonspecific.
1May not exist.
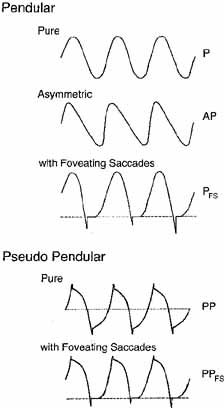
Nystagmus has traditionally been divided into two types on the basis of the clinical impression of the waveform. Thus, if the eyes appeared to oscillate with equal speed in either direction, it was called “pendular” nystagmus; if movement in one direction was faster than in the other, it was called “jerk” nystagmus. True pendular nystagmus is sinusoidal, whereas jerk nystagmus has a slow phase away from the object of regard, followed by a fast (saccadic) phase toward the target. The direction of the fast component, by convention, defines the nystagmus direction. Only accurate recordings can often assess these criteria. Nystagmus should be described not only by its waveform and direction but also by its amplitude and frequency, the product of which is intensity. The examiner should also note the positions of gaze in which the nystagmus occurs and whether the intensity changes with gaze direction. Jerk nystagmus is usually accentuated in amplitude on gaze in the direction of the fast component, a characteristic referred to as Alexander's law.2
The field of gaze in which nystagmus intensity is minimal is termed the null zone. The neutral zone is that eye position in which a reversal of direction of jerk nystagmus occurs and in which no nystagmus, any of several bidirectional waveforms, or pendular nystagmus is present. The null and neutral zones usually, but not always, overlap. Gaze-angle nulls usually result in head postures that allow use of the null to fixate targets that are directly in front of the patient.3
Based on quantitative eye-movement recordings, we identified three underlying mechanistic defects in the slow eye movement (SEM) subsystem (see Chapter 9) that produce nystagmus.
- High gain instability. In some persons, because of abnormally high gain in the SEM subsystem, a
runaway (increasing velocity) movement or a pendular oscillation
is evoked. In this chapter, the term high gain can also imply excessive delay for the gain present (i.e., the control
loop may have a normal gain, but an increased delay). Control
theory suggests how particular changes in gain can result in either
a pendular or a jerk nystagmus. Pendular nystagmus can be “congenital” (see
section on Infantile Nystagmus Syndrome) or
acquired, whereas horizontal jerk nystagmus with slow phases of increasing
velocity usually is associated with congenital nystagmus; however, the
latter may result from an Arnold-Chiari malformation.4 Vertical nystagmus with an exponential slow phase of increasing velocity
may be secondary to acquired cerebellar disease.5
- Visual-vestibular tone imbalance. The nystagmus of tone imbalance of the visual-vestibular subsystem
results from the imposition of asymmetric input on an inherently normal
horizontal gaze generator. This asymmetric input occurs if one vestibular
apparatus (labyrinths, nerve, and brain stem nuclei) functions
abnormally, if both sides are asymmetrically defective, or
if there is a central imbalance of the optokinetic subsystem. The nystagmus
recording always shows a linear (straight line) slow
phase, reflecting a persistent tone to drive the eyes toward the side
of the relatively damaged vestibular apparatus. The slow-phase
amplitude is reduced by fixation and enhanced by darkness, Frenzel (high-plus) lenses, or closing the eyes. Fixation inhibition
may be related to an opposing smooth-pursuit force and requires
the integrity of the cerebellar flocculus.
- Integrator leak. Nystagmus caused by a “leaky integrator”
occurs only in an eccentric gaze position; thus, it is gaze evoked.
The eyes are unable to maintain the eccentric position and drift back
to the primary position with a decreasing velocity, reflecting a passive
movement resisted by the viscous forces of orbital soft tissues. The
defect may reside in the brain stem “neural integrator”
or its connections, (such as in the cerebellum), which mediate eye deviation.
This form of gaze-evoked nystagmus is called “gaze-paretic”
nystagmus (see Chapter 9,
Fig. 8 for an illustration of the gaze-paretic waveform).
One means of classification of nystagmus is based on whether it is a gaze-evoked or gaze-modulated type; the former category requires that there be no primary-position nystagmus. The nystagmus exhibited in two benign syndromes (infantile nystagmus and binocular maldevelopment nystagmus), physiologic types (vestibular), and symptomatic types (vestibular) fall in the gaze-modulated category. Some physiologic types (end-point) and symptomatic types (gaze-paretic) are gaze evoked. Although these concepts of a control mechanism represent useful approaches toward a more meaningful classification of nystagmus, they are far from inclusive. For practical reasons, an empirical nystagmus classification is presented that will aid the clinician in bedside and office evaluation, without the use of sophisticated recording instrumentation. This classification continues to change as our understanding of nystagmus advances. It must be emphasized, however, that such clinical diagnoses should be considered speculative and that definitive diagnosis is often only possible by means of accurate ocular motor recordings.
The localizing significance of nystagmus is often a mere indication of dysfunction somewhere in the posterior fossa (i.e., vestibular end-organ, brain stem, or cerebellum). However, certain nystagmus patterns are quite specific and permit reasonably accurate neuroanatomic diagnosis. When possible, the specific and nonspecific forms are separated on the basis of clinical appearance and associated signs and symptoms.
NYSTAGMUS IN INFANCYThere are several types of benign nystagmus usually seen in infancy. The characteristic types of nystagmus in the Infantile Nystagmus Syndrome (INS, fka “congenital” nystagmus—CN) are the most common. Others are the nystagmus of the Fusion Maldevelopment Nystagmus Syndrome (FMNS, fka latent/manifest latent nystagmus—LMLN) and the pendular nystagmus of the Spasmus Nutans Syndrome (SNS).6 We have adopted the nomenclature recommended by the Classification of Eye Movement Abnormalities and Strabismus (CEMAS) Working Group in an attempt to eliminate the confusing and misleading terminology of some of the classical names found in the literature. The new terminology differentiates between a syndrome that includes nystagmus (often several different types of nystagmus) and a specific type of nystagmus. For example, the nystagmus seen in the INS may be any combination of two or three mechanistically different types of nystagmus (see previous) resulting in 12 to 14 specific waveforms. Using this terminology facilitates more accurate descriptions of each type of nystagmus when required (e.g., pendular, pursuit-system nystagmus) while still allowing for the inclusion of several types found in each syndrome by simply appending the word nystagmus to the syndrome (e.g., INS nystagmus). Similarly, if the shorthand, “IN” is used, it too must be understood to be a general description encompassing all of the specific types of nystagmus possible in the INS; the same applies to “FMN.”
Infantile Nystagmus SyndromeThe nystagmus of the INS is usually present at birth or noted in early infancy at the time of development of visual fixation, and it persists throughout life. The syndrome consists of one or more types of nystagmus with characteristic waveforms, head turns, tilts, or oscillations. Rarely, the nystagmus becomes manifest later in life,7 so the term congenital should be thought of as a congenital predisposition for this particular type of ocular motor instability, rather than taken literally. This syndrome may accompany primary visual defects, which led to the assumption that the nystagmus is secondary to poor vision and that both “sensory defect” and “motor defect” types existed. In fact, eye-movement recordings demonstrated that the specific types of nystagmus found in the INS had the same waveforms and underlying mechanism, regardless of the coincidental, perhaps facilitating, existence of a sensory deficit. The nystagmus itself is the direct result of an ocular motor control instability that may develop with or without an accompanying sensory deficit. Thus, for those cases in which a sensory deficit exists, it can only be a subordinate factor in the development of the nystagmus, perhaps interfering with the normal calibration of one or more of the ocular motor subsystems, thereby precipitating instability. The common association of “pendular” nystagmus with a sensory defect and “jerk” nystagmus with a primary motor abnormality was both simplistic and erroneous. Ocular motor studies of infants with INS showed no difference in waveforms associated with the presence or absence of sensory deficits; the infants exhibited the same waveforms that have been recorded in children and adults.8–10 Specifically, the development of foveation periods in INS waveforms begins early in infancy as acuity and fixation develop. This is clearly seen in infrared recordings of infants when they are attending to a visual task. INS may appear spontaneously or be familial. Hereditary INS may be sex linked, recessive, or dominant; the dominant form has been linked with chromosome 6p12.11
The relationship of the visual defect to the nystagmus possibly represents simple genetic association. Although the visual problem is not causal, it may contribute to the intensity of the nystagmus. At least one form of INS nystagmus represents a high-gain instability in a SEM subsystem,12 and fixation attempt (the effort to see) is its main driving force. Poor vision will increase fixation effort and increase the intensity of the nystagmus. Moreover, a subclinical motor instability may become manifest by this exaggerated visual effort. Although the exact anatomical location of the source of the instability present in INS nystagmus is unknown, we hypothesize that the various pendular waveforms (and some jerk waveforms) are due to a gain/delay problem in an internal (brain stem) feedback loop in the pursuit subsystem.12 That is, the pendular nystagmus waveforms of INS stem from “pursuit-system” nystagmus, modified by the saccadic system's attempts to foveate the target and the fixation subsystem's attempt to extend foveation; this hypothesis is embodied in an ocular motor system model (see Chapter 9).13–16 The much greater frequency of horizontal–torsional nystagmus, compared with vertical or diagonal nystagmus, probably reflects inherent differences in the stability of the respective pursuit subsystems (i.e., the horizontal is more unstable than the vertical). Although there is no torsional smooth pursuit system per se, the torsional component of the nystagmus reflects instability in torsional control.17 Another factor in support of the hypothesis of pursuit-system nystagmus is that no oscillopsia is perceived from oscillations in pursuit velocity, not in normals and not in those with INS. Thus, no additional adaptation mechanism need be proposed to account for the absence of oscillopsia in INS; it is suppressed by the same mechanism by which normals suppress it during pursuit. Initially, we proposed that excessive positive feedback around the common neural integrator might be responsible for the accelerating slow phases of INS nystagmus.18 We subsequently demonstrated that the common neural integrator is not the site of the INS instability.19 However, several models have been proposed that attempt to explain the genesis of some INS waveforms, based on that premise.20–22 Although each can generate limited, specific INS waveforms, the models exhibit behaviors inconsistent with data from individuals with INS and do not simulate the known broad range of human ocular motor responses (both normal and during nystagmus) to common stimuli (i.e., they are demonstrations of putative mechanisms to generate waveforms rather than models capable of simulating ocular motor system behavior). Because IN appears to be activated and intensified by fixation attempt, the deficit may also be linked to the fixation subsystem (see Chapter 9). The coexistence of a high-frequency pendular oscillation with a low-frequency jerk nystagmus (resulting in a dual-jerk waveform) in some INS subjects and also in FMNS, suggests that the high-frequency pendular oscillation is due to an instability at a different site. Recent evidence points to the nucleus of the optic tract (NOT) as the site of this oscillation shared by patients with either the INS or FMNS.23 Goldstein suggested that IN is caused by oscillations at two frequencies whose interactions may approximate some of the known INS waveforms.24 However, such interactions do not produce the absolutely motionless (i.e., “flat”) periods of extended foveation (300–400 msec) recorded in many patients.
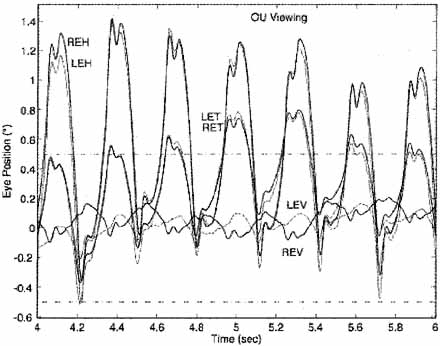
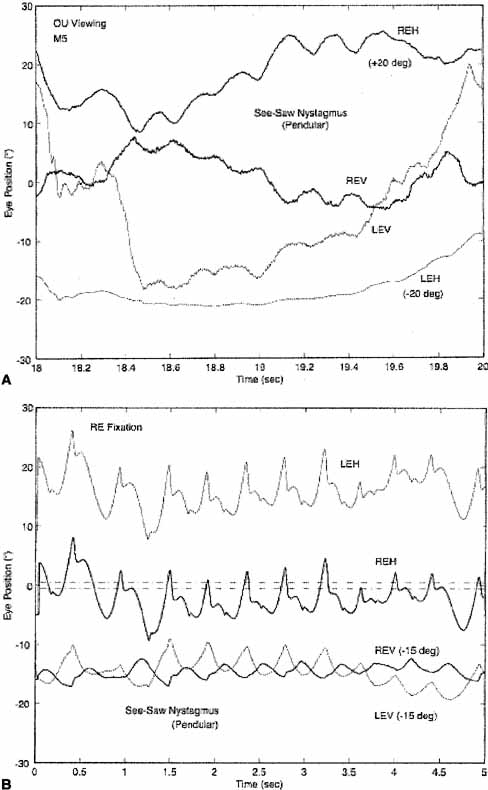
Distinguishing the lower frequency pendular nystagmus from jerk nystagmus may be difficult clinically, particularly in the INS. Certain forms of jerk nystagmus are invariably mislabeled as pendular, or the direction is misidentified. Even with oculographic recordings, the direction of the fast phase may be misinterpreted unless velocity tracings are obtained.25 In the absence of oculography, clinicians should describe the nystagmus carefully or use diagrammatic methods (see Figs. 1, 2, and 3). Monocular visual deprivation induced, in some monkeys, a diagonal nystagmus whose horizontal component initially looked like FMNS slow phases (see later discussion) and then developed to resemble INS slow phases. This deprivation took place from birth to 25 days and was followed by monocular deprivation of the other eye.26 The role of the NOT in FMN in monkeys has been more clearly defined recently.27,28
The INS nystagmus usually damps significantly with convergence. Although the exact mechanism responsible for this damping in unknown, there was speculation that it might result from co-contraction of antagonist muscles of each eye during convergence. However, recent work by Miller found no co-contraction.29 We hypothesized that damping during convergence might result from an effective increase in the stiffness of the ocular motor plant brought about by the increased innervation to the two medial recti. (i.e., co-contraction of antagonist muscles of the two eyes, rather than of each eye). The Orbit 1.8 simulation (J. M. Miller, personal communication), predicted that the 8 g primary-position tension in the medial rectus increased to 13 g at 20° adduction (40° of convergence) and to 18 g at 30° adduction (60° of convergence), 75% and 125% increases, respectively. Because convergence results in a change in the muscle pulley system,30,31 the latter may be the mechanism by which the stiffness is increased. The observations of convergence-induced damping of other types of nystagmus support this peripheral mechanism in preference to one relying on an inherent property of the nystagmus. As previously mentioned, the intensity of INS nystagmus is related to the fixation attempt, which probably explains why it sometimes persists with eyes open in darkness (when the subject will probably attempt to “see”) and damps behind closed lids (when the subject will, unless instructed to the contrary, reduce any attempt to “see”).25 The defining criterion is fixation attempt, not retinal illumination or lid position. Therefore, reports of the presence or absence of nystagmus with lid closure or darkness that lack a description of the instructions to the subject, provide little useful information.
The recognition of INS is of extreme importance, particularly in the adult patient, and may obviate unnecessary neurodiagnostic procedures; its characteristics are listed in Table 2. INS is almost always binocular and never shows more than minor amplitude dissociation between the two eyes. Clinically, the nystagmus usually appears uniplanar. Like vestibular end-organ nystagmus, horizontal nystagmus remains horizontal when the eyes are deviated vertically and does not convert to vertical nystagmus. Using new, sensitive techniques for recording torsional eye movements, we found small but significant torsional components in the nystagmus of subjects previously thought to have purely horizontal INS.17 Because the prominent horizontal movement masks the usually smaller torsional component, the latter appears to be a common characteristic of “horizontal” INS. In most patients, rightward movements were accompanied by clockwise torsion and leftward movements by counterclockwise torsion.32 We discuss later the superimposition of a latent component on an ongoing IN.
TABLE 2. Characteristics of Infantile Nystagmus
| Binocular with similar amplitude in both eyes |
| Usually horizontal and torsional (vertical rare) |
| Pendular or increasing velocity slow phases |
| Distinctive waveforms with foveation periods and braking saccades |
| Asymmetric aperiodic alternation possible (Baclofen ineffective) |
| Provoked or increased by fixation attempt |
| Abolished in sleep or inattention to visual tasks |
| Gaze-modulated, not gaze-evoked |
| Diminished (damped) by gaze-angle or convergence nulls |
| Superimposition of latent component possible |
| “Inversion” of the optokinetic reflex (actually, reversal of the IN) |
| Associated head oscillation (not compensatory) or turn |
| No oscillopsia except under rare conditions |
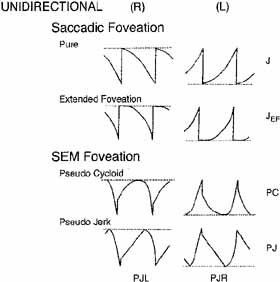
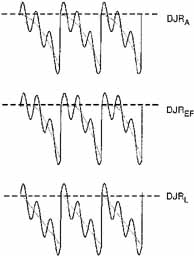
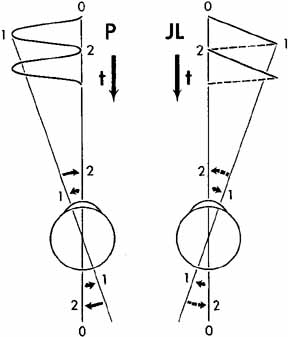
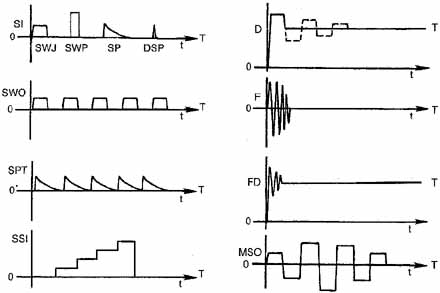
Eye-movement recordings of INS occasionally show a pure pendular waveform (sinusoidal) or a saw-toothed waveform (equiamplitude linear slow phase with foveating saccade) (see Fig. 8) typically seen in vestibular nystagmus. These pure forms are neither frequent nor pathognomonic for INS. More often, INS manifests distinctive waveforms that are not present in acquired nystagmus. These waveforms are an expression of the attempts by the ocular motor control system to increase foveation time, imposed on inherently unstable slow control. The INS waveforms shown in Figs. 4, 5, 6 and 7 (other than pure pendular or jerk) have never been recorded in acquired horizontal nystagmus.8,33 The target position is indicated by a dashed line; target position is problematic for pure and asymmetric pendular waveforms. We have regrouped the original waveforms based on hypothesized mechanisms to emphasize the different types of nystagmus that may be recorded in an individual with INS. This resulted in differentiating linear slow phases from the more common accelerating slow phases of jerk nystagmus, based on presumed mechanisms (visual-vestibular and pursuit-system, respectively). The presence of dynamic overshoots in the fast phases (saccades) of IN waveforms is both idiosyncratic and variable; for simplicity, we have not included them in Figs. 4, 5, 6 and 7. Because dynamic overshoots are not an abnormality, their presence does not represent a “different” waveform. Using our computer model,14,16 we demonstrated how the pendular waveforms in Figure 4 could be generated from a pursuit-system instability, and we are investigating two possible sources for the unidirectional jerk waveforms in Figure 5. We hypothesize that the linear-slow-phase jerk waveforms in Figure 6 arise from a tonic imbalance in the visual-vestibular subsystem. When the low-amplitude, high-frequency pendular oscillations, hypothesized to arise in the NOT, are present, they are superimposed on the basic IN waveforms as shown in Figure 7. Of the 15 waveforms shown in Figs. 4, 5, 6 and 7, we hypothesize that nine are due to pursuit-system oscillation (Figs. 4, 5), three to visual-vestibular imbalance (Fig. 6), and the three in Figure 7 to NOT nystagmus added to either type. We have also recorded the high-frequency NOT nystagmus superimposed on a low-frequency pendular IN. Twelve of these waveforms are pathognomonic of IN; P, AP, and JL are not. One important feature of IN waveforms is the presence of “braking saccades” that act to stop runaway slow phases.34 They are triggered by extraretinal eye-velocity information and, when properly measured, have the same velocity and duration characteristics as other types of saccades.35,36 For pendular waveforms, the target is foveated at the peaks that are more flattened, indicating extended foveation. Extended foveation in an adult with lifelong nystagmus secondary to a congenital brain stem hamartoma and in an adult given gabapentin for treatment of nystagmus secondary to an arteriovenous malformation37 supports the hypothesis that extended foveation periods in INS waveforms represent the action of a normal fixation system on the underlying INS oscillation. Figures 8 and 9 demonstrate how these waveforms serve to increase the time of foveal imaging.
|
|
|
The pure pendular (P) and jerk (J) waveforms in Figure 8 are not conducive to good acuity because of the extremely short foveation time (instants 0 and 2 on the time axis). Although these are common acquired waveforms, when afflicted with INS, the developing nervous system modifies pendular and jerk waveforms; therefore, foveation time (and thus acuity) is increased. Examples of some resultant waveforms are shown in Figure 9. In the pendular nystagmus with foveating saccades waveform (PFS), there is usually a substantial period of time when the target is imaged on the fovea and the eye is motionless (instant 3 on the time axis). In jerk-right nystagmus with extended foveation (JREF), the position from time 0 to 1 is when foveation takes place, and in the bidirectional jerk-left (BDJL) waveform, the position from instants 4 to 5 is conducive to good acuity. Waveform, gaze-angle nulls, and convergence nulls are affected by heredity.38 Members of the same family show more specific combinations of waveforms or of either waveform, having only a convergence null or no convergence null (i.e., having only a gaze-angle null), than do members of the general INS population. We found greater damping of INS nystagmus with convergence than with gaze angle, in patients who exhibited both types of null, and this translated into acuity increases.39 Comparison of the results of the Anderson-Kestenbaum and artificial divergence procedures also favored the latter.40
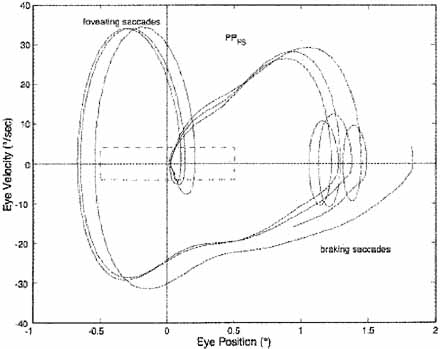
Increased foveation time is the most effective determinant of increased acuity.41–44 In most INS subjects, the best waveform (i.e., most foveation time per cycle) is in the null region associated with a particular gaze or convergence angle, but in other subjects it is not; these latter subjects prefer the gaze or convergence angle that yields the best waveform, even if it is not the waveform with the least amplitude. Decreased amplitude is, however, the major determining factor in cosmetic improvement and is appreciated by a young patient's parents. Despite a nulling of the nystagmus, an individual with INS may not show an increase in acuity with convergence if the resulting waveform has little foveation time per cycle or if acuity is primarily limited by a visual deficit. The fixation system of someone with INS is able to repeatedly foveate a target within minutes of arc, almost as accurately as a normal person.39,42,45 The use of phase-plane analysis allows definition of a “foveation window” (±0.5° by ±4.0°/second) for the study of fixation, smooth pursuit and the vestibulo-ocular reflex (VOR).39,46,47 These studies demonstrate the extremely accurate fixation, pursuit, and VOR possible in individuals with INS. In Figure 10, the tight overlap of foveation periods within the foveation window in the phase plane demonstrates how accurate the cycle-to-cycle target foveation in IN can be. The foveation window defines the time when the eye is within ±0.5° of the target and moving with less than ±4°/sec. Thus, despite an ocular oscillation whose amplitude is well outside the foveal extent and whose velocity exceeds ±30°/sec, the ocular motor system can foveate the target with extreme accuracy and allow normal visual acuity.
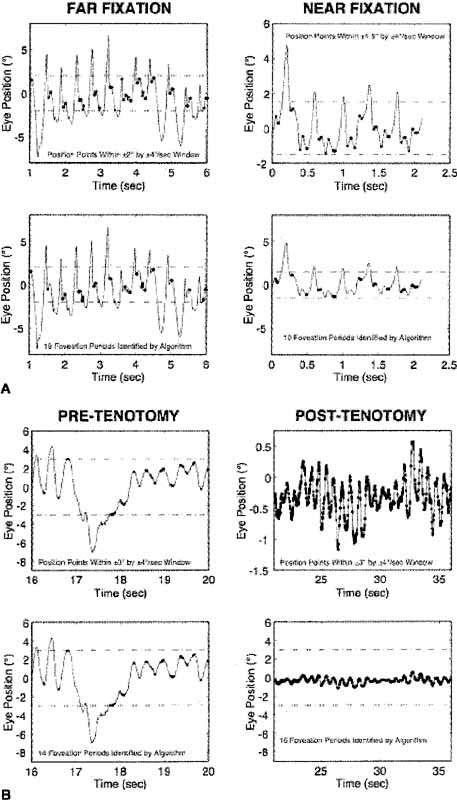
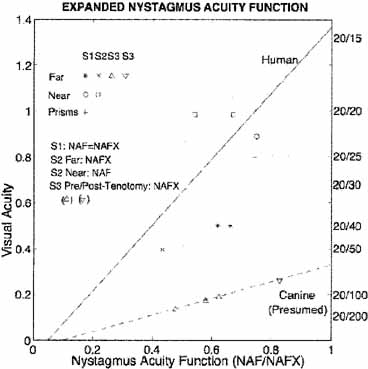
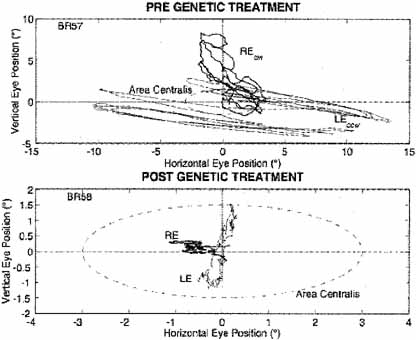
The nystagmus acuity function (NAF) provides an objective determination of potential visual acuity from measurements of the key characteristics of the INS waveform: foveation time and the standard deviations of foveation position and velocity means (for NAF) or position mean alone (NAFP).48 For those subjects whose foveation ability is not well developed (i.e., the target image always falls within the default foveation window), the window used for its calculation can be enlarged and the expanded NAF (NAFX) plotted versus gaze or convergence angle. Software is now available that calculates the NAFX from eye-movement data and provides a quantitative method for evaluating different therapies for their affect on potential visual acuity.49 Plots of the NAF or NAFX versus visual acuity reveal the linear relationship that allows intersubject prediction of potential visual acuity. The NAFX can also be used to compare potential acuity across subjects with different types of nystagmus (INS or FMNS) or to predict the acuity increase possible after therapeutic intervention in a given subject. The latter is accomplished by plotting the NAFX versus gaze or convergence angle. Figure 11A shows the NAFX outputs during near and far fixation from a subject with achiasma, recorded in Laboratory of H. Collewijn in Rotterdam (see Albinism and Achiasma section following).50 As the NAFX clearly shows, conditions for highest visual acuity occurred during near fixation where (as the bottom panels show) a smaller foveation window could be used to calculate the NAFX. Figure 11B shows the pre- and post-tenotomy NAFX outputs from an achiasmatic Belgian sheepdog, demonstrating the effectiveness of the treatment. As the bottom panels show, the post-tenotomy nystagmus is subclinical and well within the boundary of the area centralis; the nystagmus during the whole interval qualified as a foveation period. Figure 11C shows the NAFX versus potential (solid and dashed lines) and measured (symbols for the human data) visual acuity for two humans (S1 and S2) and a canine (S3). The NAFX (and, potential visual acuity) is higher at near than at far (S1 and S2) or while using base-out prisms at far (S1). For the canine, the post-tenotomy NAFX is higher than preoperatively, and the dashed line is inferred from veterinary data. Because of its direct relationship to potential visual acuity, the NAFX is the best available outcome measure of therapies designed to affect the IN waveform. Although amplitude is related to cosmetic appearance, neither it nor frequency is closely correlated with acuity.
|
|
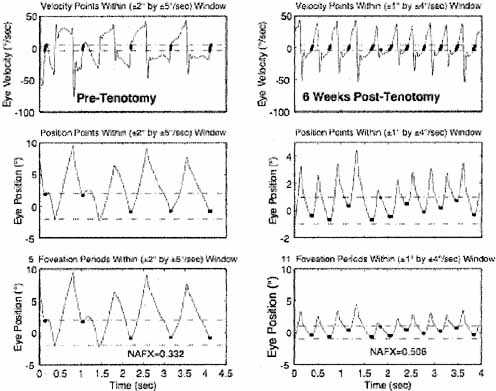
We hypothesized a new type of surgery to damp the IN of subjects that do not have gaze-angle, convergence or static nulls (i.e., they have asymmetric (a)periodic alternating nystagmus—APAN) or do have a primary-position null.51 The surgery consists of a simple tenotomy, dissection, and suture of the involved extraocular muscles in place, with neither recession nor resection. Results of a masked clinical trial of this surgery were positive in a phase 1 study of adults.52,53 The putative mechanism responsible for the damping effects of tenotomy is alteration of a proprioceptive tension-control loop.51 This is supported by the discoveries of the neural substrate for such a loop in both the musculotendon54–57 and enthesial end of the tendon (where the surgery takes place).58 Before any surgery is contemplated, IN should be definitively diagnosed using ocular motility recordings.59 Figure 12 shows the improvement in NAFX output of a patient after four-muscle tenotomy. As the increased NAFX values indicate, tenotomy damped the IN and improved the waveform. Both the foveation time per cycle and the mean foveation position improved and the post-tenotomy foveation window was smaller in both position and velocity. The various therapies available for INS, based on the presence or absence of gaze and convergence nulls, is summarized in Table 3. Note that for patients with both convergence and gaze-angle nulls, exploitation of the former (surgically or with vergence prisms) usually damps the nystagmus and increases acuity most; it is necessary to add -1.00 S (OU) to vergence prisms for prepresbyopic patients. Preliminary studies (at all gaze angles) of the broad damping effects of convergence on IN (see Fig. 14 and discussion of convergence following) suggest that composite prisms (unequal base-out) are not necessary in these cases; vergence prisms (equal base-out) will achieve the same damping.60 As indicated in Table 3, regardless of the presence of nulls, afferent stimulation can be used in all patients who exhibit nystagmus damping with active stimulation (see later discussion).
TABLE 3. Therapies for Infantile Nystagmus
| If the IN nulls ONLY with lateral gaze: |
| Resection and recession (four-muscle) |
| Version prisms |
| Afferent stimulation (passive or active) |
| If the IN nulls ONLY with convergence: |
| Bimedial recession1 (artificial divergence) plus bilateral tenotomy |
| 7D BO vergence prisms with (1.00 S1 (OU) |
| Afferent stimulation (passive or active) |
| If the IN nulls with BOTH lateral gaze and convergence: |
| Bimedial recession1 plus bilateral tenotomy or combined with resection and recession |
| 7D BO vergence prisms with (1.00 S1 (OU) |
| Afferent stimulation (passive or active) |
| If the IN nulls with NEITHER lateral gaze nor convergence or is asymmetric aperiodic alternating IN: |
| Four-muscle tenotomy, dissection and suture |
| Maximal recession (Four-muscle)* |
| Afferent stimulation (passive or active) |
1Damps IN only for nonstrabismic, binocular patients.
*This surgery should be replaced by the four-muscle tenotomy procedure that does not carry the risk of inducing diplopia.
The so-called inversion of the optokinetic reflex seems to occur only with INS.61 When optokinetic stimuli are presented to a patient with INS, a peculiar phenomenon may occur: the resulting nystagmus may be opposite in direction from what would be anticipated if the evoked optokinetic nystagmus (OKN) simply summated with the ongoing nystagmus. For example, in the presence of left-beating INS nystagmus, the response to right-going optokinetic targets (a leftward fast phase) should add to the left-beating INS nystagmus to produce enhancement of the nystagmus intensity. In “inversion,” the nystagmus may either damp or be converted to right-beating nystagmus. If right-going targets are presented at a gaze angle at which the nystagmus is either absent or pendular, a right-beating nystagmus may result. Inversion of the optokinetic reflex is present in 67% of INS patients. The observation of optokinetic inversion establishes the nystagmus as IN. The phenomenon is, in reality, merely a reversal of the INS nystagmus direction due to a null shift; it is not a true inversion of the optokinetic response (see discussion of reversed pursuit later). The basic function of the optokinetic system is to stabilize slowly moving retinal images, but the rapidly moving retina of an INS patient may interfere with this function. The optokinetic response appears suppressed in some patients; however, the perceived circularvection is in the proper direction, and OKN dynamics appear to be normal in individuals with INS.
The head oscillations that often accompany INS increase with visual intent and have traditionally been regarded as compensatory. For compensation to be achieved, head movements would have to be equal in amplitude and opposite in direction to the eye movements. For such a mechanism to work, the VOR would have to be totally inhibited (gain reduced to 0). Accurate objective observations of the head movements in patients with INS do not support that hypothesis.62 Rather, the head oscillation is merely an extension of the motor instability, and the VOR functions normally to cancel the effects of head oscillation during the periods of target foveation normally present in the INS waveform.47 The head tremor in INS can be distinguished from that in acquired disease; it is easily suppressed voluntarily in the former but not in the latter.
Point out the head tremor to the patient. If it stops, the nystagmus is IN; if it persists, both are acquired.
Individuals with INS usually do not experience an illusory oscillatory movement of their environment (oscillopsia).63,64 This lack of oscillopsia in INS, and also in FMNS, suggests that both oscillations occur within an efference copy feedback loop that serves to nullify the effects of retinal-image oscillation induced by either of these instabilities.65 Like most ocular oscillations (myoclonus being the exception), INS nystagmus disappears in sleep. In two patients with INS plus an acquired nystagmus, their acquired oscillopsia seemed to be related to an inability to maintain repeatable periods of good foveation in a particular plane.66,67 However, that inability was an epiphenomenon caused by the addition of a transitory acquired nystagmus to the ever-present INS nystagmus.65 Oscillopsia suppression in INS and other types of nystagmus appears to be accomplished by efference copy of the nystagmus signal.65,68–72 Oscillopsia may occur in some patients with very poor foveation stability73 or may occur in later life secondary to afferent deficits.74 The thresholds for motion detection in INS differ from normal and may also play a role in oscillopsia suppression.75,76 We reported oscillopsia of a migraine aura in an individual with INS,77 as well as vertical oscillopsia secondary to a decompensated phoria; the latter event led to the discovery of subclinical see-saw nystagmus in the horizontal–torsional waveforms of IN.78 As Figure 13 shows, the subclinical see-saw nystagmus is phase locked with the horizontal and torsional components allowing foveation periods to occur in all planes simultaneously. Torsional motion does not seem to impair orientation perception thresholds, perhaps due to extraretinal information.79 We hypothesize that subclinical see-saw nystagmus results from slight mismatches between the forces exerted by the vertical recti and oblique muscles and does not represent a true vertical-system instability; this contrasts with the see-saw nystagmus accompanying achiasma (discussed later).
|
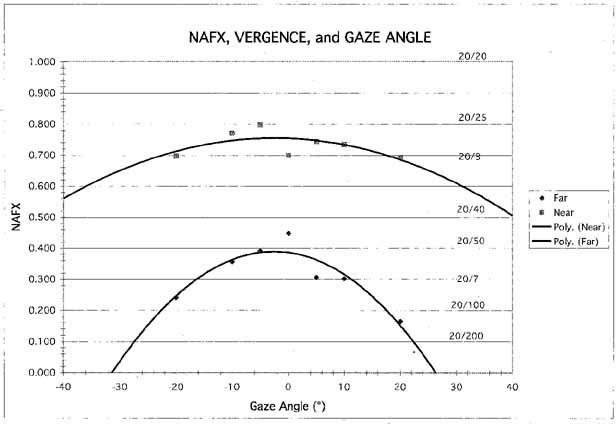
During fixation of stationary targets, many individuals with INS have a permanent null region representing the gaze angle at which the intensity of the nystagmus is the lowest and the waveform most conducive to highest NAFX and acuity. They often turn their heads to permit straight-ahead viewing with the eyes in the null region. Such patients benefit from appropriate version prism spectacles that alleviate the necessity for the head turn and the resulting increased fixation attempt.25,41 However, as Figure 14 shows, if the IN damps with convergence, the higher NAFX values will persist over a broader range of gaze angles than during fixation on a far target. This allows higher acuity over most useful gaze angles and demonstrates the advantage of either base-out prisms or the bimedial rectus recession procedure over therapies aimed at moving a gaze-angle null to primary position.
Some INS patients may exhibit a superimposed latent component that induces null shifts toward an eye that is covered (Fig. 15).80 Demonstration of such a shift and maintenance of any of the INS waveforms establish the nystagmus as belonging to INS rather than the FMNS (see later discussion). Rarely, a null shift is toward the viewing eye.25
Some studies of INS and smooth pursuit have led to confusion between the reversal of INS direction that may occur during pursuit and reversed pursuit. This confusion is similar to that discussed earlier for the optokinetic response. Accurate eye-movement recordings show that neither the optokinetic nor the pursuit subsystem responds in a reversed manner, as should be obvious both by the absence of any symptoms of such a grave deficit and the normal abilities of individuals with INS in sports. Also, their perceptions of both the direction and magnitude of movements in the periphery and on the fovea are normal. Just as the INS waveform is distorted by SEM (creating periods of extended foveation) during fixation of a stationary target, the pursuit system is able to generate pursuit movements with a direction and velocity that match those of a moving target during these same periods of the INS waveform.46,68,81 This ensures extended foveation of the moving target and results in accurate smooth pursuit during the periods when the target image is on the fovea. Pursuit during foveation is all that is necessary for good acuity; the same conditions are met during smooth pursuit as are met during fixation of a stationary target. During smooth pursuit (or during optokinetic or VOR stimuli) the gaze angle at which the INS null region occurs shifts in the direction opposite to the pursuit (optokinetic grating or VOR-induced eye motion).46,47 The amount of null shift is a function of the pursuit or VOR velocity. This measurable shift in the INS null angle causes the INS nystagmus reversal that has been mistakenly equated with “reversed” responses of both the optokinetic and pursuit subsystems.
In many individuals with INS, afferent stimulation of the ophthalmic division of the trigeminal nerve or of the neck may damp the nystagmus and improve the waveform, allowing increased visual acuity.48,82 Neck or forehead vibration prolonged foveation periods, yielding higher values of the NAF and improved visual acuity in 9 of 13 patients with INS.48 This noninvasive and benign therapy (active afferent stimulation) may prove useful in both INS and acquired nystagmus. The use of soft contact lenses to improve the acuity of individuals with INS takes advantage of the damping effect on nystagmus of (passive) afferent stimulation.83–86
Soft contact lenses are not contraindicated in INS and can provide better acuity than spectacles in patients whose nystagmus damps with afferent stimulation. Plano soft contact lenses can be used if no refractive correction is required.
Both surgical and nonsurgical treatments for INS have been reviewed elsewhere.3,87 Relatives of individuals with INS may have saccadic instabilities,88 and carriers of blue-cone monochromatism may have vertical (upbeat and downbeat) nystagmus and FMNS.89
Fusion Maldevelopment Nystagmus SyndromeThe FMNS consists of: a jerk nystagmus with either a linear or decreasing-velocity exponential slow phase identical to that of gaze-paretic nystagmus; strabismus; alternating hyperphoria/dissociated vertical deviation; and pendular torsional nystagmus in primary position. Occasionally, when both eyes are closed, a jerk nystagmus with a linear slow phase is present. Classically, the nystagmus evoked by the “pure” or “true” latent condition (LN) occurs only with uniocular viewing. There is no nystagmus with both eyes viewing, but when one eye is occluded, nystagmus develops in both eyes, with the fast phase toward the uncovered eye (Fig. 16). The fast phases of FMN may contain dynamic overshoots,90 whose characteristics in both FMN and IN are normal.91 Dynamic overshoots are integral parts of the saccadic fast phases and not the beginnings of the slow phases. The occurrence of square-wave jerks (SWJ, see following) is also normal, and their presence during binocular viewing in FMN is unpredictable and variable. Thus, the presence of either does not represent a different “type” of FMN;92 it is merely the same FMN including these common, normal saccadic dynamics or intrusions. This also applies to the occurrence of other types of nystagmus in addition to FMN (e.g., an undefined torsional nystagmus or the high-frequency pendular nystagmus thought to arise from the NOT, resulting in a dual-jerk waveform). In an individual patient, both FMN and IN may coexist with another type of nystagmus or saccadic intrusion/oscillation. The clearest way to delineate a group of patients with two mechanistically independent conditions is to describe them as having a syndrome consisting of two conditions (e.g., FMN with dynamic overshoots, FMN plus SWJ, or FMN plus torsional nystagmus). For simplicity, the basic FMN waveforms shown in Figure 16 do not contain dynamic overshoots in their fast phases nor the confounding addition of other types of nystagmus. When the additional pendular oscillation of NOT nystagmus is present, the resulting waveform would be the same as shown in Figure 7 for linear slow phases or consist of a pendular oscillation superimposed on decelerating slow phases.38 FMN is always congenital. However, several cases have been recorded of the manifest form of FMN (MLN occurring with both eyes open), associated with retrolental fibroplasia.90 FMN is common in Down syndrome, where it may coexist with IN.93
Early theories postulated that a unilateral retinal stimulus was the necessary condition for FMN, but this concept was discounted by observations of FMN in monocular fixation with a blind eye or with an acoustic stimulus in complete darkness. Similarly, the hypothesis that the FMNS is caused by nasal-temporal asymmetries in the optokinetic reflex is not supported by evidence that subjects with FMNS are able to use retinal slip information to adapt motion-detection sensitivities94 and are able to pursue symmetrically.95 Also, because nasal-temporal asymmetries exist in individuals with strabismus but not FMN,94 this cannot be the primary causal factor in the genesis of the nystagmus. Asymmetries in the monocular optokinetic response of monkeys deprived of binocular input early in life may result from, rather than cause, their nystagmus. In normal monkeys, each nucleus of the optic tract (NOT) is driven binocularly; in these monkeys, they are driven by the contralateral eye.96 Although the resulting imbalance may provide the tonic signal that produces the FMNS slow phases (inactivation of the NOT with muscimol abolishes the nystagmus), the cause of the imbalance appears to lie in higher centers. The spontaneous reversal of FMN in the dark has led to the speculation that eye dominance is predetermined.97,98 Shallo-Hoffmann et al. identified an alternating vertical component to FMN,99 and Brodsky linked the genesis of FMN and dissociated vertical divergence to the dorsal light reflex present in many animals.100
We relate the FMNS to the cortical switching that must occur in the calculation of egocentric direction when going from binocular to monocular viewing.90 Under binocular conditions, the gaze angle of each eye is summed with the other and divided by two to obtain the egocentric direction, referenced to the “cyclopean eye.” However, with monocular viewing, egocentric direction depends only on the viewing eye, and the cortical operation of summing and dividing by two must be altered to process unchanged information from the viewing eye. The shift in egocentric direction toward the nonviewing eye causes the slow drift of the eyes in that direction. Both eyes are then corrected by a saccade in the direction of the viewing eye, which brings the eyes to the target (or, in darkness, to the intended gaze angle). This contention is supported by unilateral strabismus surgery causing central effects on egocentric localization.101 Thus, FMNS nystagmus may be generated by this inability to properly alter the cortical mathematical operation normally used to define egocentric direction (i.e., this deficit in higher centers may result in a tonic imbalance in the visual-vestibular subsystem, producing the linear slow phases of FMN).
The shift to monocular egocentric localization can also produce a mode whereby the saccadic system generates defoveating saccades that momentarily carry the fixating eye past the target in a temporal direction, followed by a decelerating-velocity nasal drift back toward the target.102 This results from generating a pulse, but not a step, of innervation to drive the fast phases of the FMNS nystagmus. Presumably, the common neural integrator is kept from integrating these defoveating pulses by the signal representing the correct eye position vis-à-vis the target. These hypotheses have been combined in an ocular motor system model (see Chapter 9) capable of simulating responses of an individual with FMNS.15,103,104
FMNS nystagmus occurs in patients with strabismus who, although viewing with both eyes open, are fixing monocularly. The slow phases are either linear or decelerating, and the fast phases are always in the direction of the viewing eye.90 The nystagmus of patients with strabismus, alternating fixation, and FMNS nystagmus with both eyes open has fast phases always in the direction of the fixating eye. Such patients may be easily misdiagnosed as having INS, because the nystagmus is present with both eyes open. Recordings are required to document the decelerating or linear slow-phase waveforms characteristic of FMNS from the accelerating slow phases predominant in the INS.
Strabismus is a necessary (but not sufficient) condition for FMN.105 That is, all individuals with FMN have strabismus, consisting of a phoria under cover and a tropia with both eyes open, if nystagmus is present under these respective conditions. Conversely, FMNS is not significantly associated with early-onset strabismus.106 Rarely, on occlusion of a preferred eye, during which fixation with an amblyopic eye is forced, both eyes drift in the direction of the covered eye without corrections by fast phases; this is called latent deviation. Early surgical correction of infantile strabismus may convert the nystagmus of FMNS present with both eyes open (the manifest condition) to nystagmus present only upon occlusion of one eye (the latent condition),107 thereby supporting a previous hypothesis.105
Because the good acuity of INS patients is related to the long, postsaccadic foveation periods of many waveforms, it was difficult to explain the equally good acuity of FMNS patients, given the absence of such periods. However, accurate studies of FMNS foveation revealed a dual strategy.102 During the low-amplitude, linear-slow-phase FMNS waveform, the saccadic fast phases foveate the target, and the low-velocity slow phases take the eye away from the target with little effect on acuity. During the higher amplitude, decelerating slow-phase FMNS waveform, the saccadic fast phases defoveate the target, allowing foveation during the low-velocity, tail ends of the slow phases (see Fig. 16); this ensures the best acuity possible.
Although most patients have nystagmus from either the INS or the FMNS, some have both; three unambiguous patient groups have been identified: INS, FMNS, and INS + FMNS.38,108,109 The three groups exhibit different clinical signs and relations to strabismus; most INS patients do not have strabismus, but all FMNS patients do. Thus, INS and FMNS are specific, easily differentiated syndromes and do not, as has been suggested,110 represent a unitary disorder with a broad spectrum of expression. Because no acquired, time-independent, primary-position jerk nystagmus reverses direction with alternate eye cover, a simple reverse-cover test can be a powerful clinical tool.
Nystagmus Blockage SyndromeTo distinguish between benign, infantile, primary-position, jerk nystagmus, and that which is acquired and symptomatic, first verify that there is no periodic alternation in direction and then perform a reverse-cover test. If the cover test causes a reversal in the nystagmus direction consistent with FMNS, the nystagmus is benign (FMNS or INS with a latent component). If not, attempt to rule out INS (by history, clinical signs [see Table 2], and waveforms).
The nystagmus blockage syndrome (NBS) is both a poorly understood and an overdiagnosed phenomenon related to INS. As the name suggests, the nystagmus of these patients diminishes or disappears with the act of willed esotropia while fixating a distant target. This should not be confused with the damping of INS nystagmus during convergence on a near target. There are two mechanisms by which blockage of the ongoing nystagmus can be accomplished.111 During the willed esotropia, some INS nystagmus merely damps or stops, in much the same way as with true convergence. In the second type of NBS, the INS waveform converts to a FMNS waveform with the onset of the strabismus. Normally, the substitution of the FMNS slow phases for the INS waveforms that allow for better foveation would not be advantageous. However, in these few patients, the small FMNS amplitude results in better acuity than the larger INS amplitude. NBS is often misdiagnosed in FMNS patients with a strong Alexander's law variation of their nystagmus, which causes them to fixate with their adducting eye.111
Spasmus Nutans SyndromeSpasmus nutans syndrome (SNS) is a rare constellation of ocular oscillation, head nodding, and torticollis that begins in infancy (usually between 4 and 18 months of age) and disappears clinically in childhood (usually before 3 years of age). The nystagmus is generally bilateral (but can differ in each eye and may even be strictly monocular), and it oscillates in horizontal, torsional, or vertical directions. Hoyt reported an instance of SNS presenting with monocular nystagmus in monozygous twins.112
The nystagmus tends to be asymmetric in the two eyes, to vary in different directions of gaze, and to be rapid and of small amplitude. The head nodding is inconstant and irregular and can be horizontal, vertical, or both. The average duration of SNS is 12 to 24 months; rarely, it lasts a number of years. Studies of quantitative head- and eye-movement recordings indicate that the head movement may, using the normal VOR, actually serve to abolish the eye movements.113 In some patients, it may be only compensatory with suppression of the VOR. Compare this to INS, where the head oscillation is an extension of the nystagmus and the VOR is normal (see preceding discussion).
The pendular oscillation of SNS is characterized by a variable phase difference between the oscillations of each eye.114 These phase differences can appear from minute to minute and during the child's development. The dissociated nystagmus is usually of a higher frequency than INS nystagmus, and the result can be disjugate, conjugate, or uniocular. We hypothesize that SNS reflects a yoking abnormality, perhaps due to delayed development. Recordings show that SNS nystagmus may not disappear completely but may recede to a subclinical level; neither INS nor FMNS nystagmus disappears with age.
Acquired
SECONDARY TO VISUAL LOSS.
Nystagmus occurring in early childhood consequent to progressive bilateral visual loss should not be classified as INS unless INS waveforms are documented. The conceptual problems in the classification were discussed earlier. Usually, nystagmus secondary to visual loss cannot be distinguished from INS in a patient with coexisting primary visual abnormalities.
The nystagmus associated with rod monochromacy (complete congenital achromatopsia) is said to be distinguishable from other forms of nystagmus of infancy on the basis of slow buildup of the slow component velocity of OKN. This occurs during monocular stimulation with directional asymmetry of OKN when the temporal-to-nasal direction is compared with the nasal-to-temporal direction.115 Patients with blindness from birth and nystagmus may have an impaired VOR and an inability to initiate saccades voluntarily, despite the presence of quick phases of nystagmus.116 Adults with “eye movements of the blind” may exhibit features similar to those of patients with cerebellar disease116 or may exhibit jerk nystagmus, see-saw nystagmus, or a combination of oscillations. Cats reared from birth in stroboscopic illumination develop low-amplitude nystagmus; this is believed to be an animal model for nystagmus secondary to visual loss.117
Monocular visual loss may produce monocular nystagmus, usually vertical, at any age from birth through adult life. That the nystagmus is monocular and usually vertical makes it distinguishable from INS, but it may mimic the nystagmus of spasmus nutans, particularly if there is associated head nodding.
SPASMUS NUTANS.
The nystagmus of the benign spasmus nutans syndrome may sometimes be mimicked by tumors of the optic nerve, chiasm, or third ventricle,118 although both the true SNS nystagmus and that secondary to tumor have been called spasmus nutans, we prefer to limit that term to the benign condition. Any child with nystagmus resembling SNS nystagmus should have brain imaging if tumor cannot be ruled out by other clinical signs. Retinal disease may mimic the clinical signs of the SNS,119 as may opsoclonus-myoclonus.120
ACQUIRED PENDULAR NYSTAGMUS (ADULTS)
Acquired pendular nystagmus may reflect brain stem or cerebellar dysfunction, or both. It occurs in patients with vascular or demyelinating disease. In the latter, it has been regarded as a sign of cerebellar nuclear lesions. The nystagmus is multivectorial (i.e., horizontal, vertical, diagonal, elliptic, or circular) and usually is associated with a head tremor. Marked dissociation between the two eyes often exists and may not correlate with differences in visual acuity from coexisting optic neuropathy.121 Despite the dissociation, the oscillations of the two eyes in a patient with MS are phase-locked, even though they may differ in their frequencies.122 Das et al. postulated a neural-net model for acquired pendular nystagmus in MS; the model duplicated the resetting effect of saccades on the oscillation.123 Acquired pendular nystagmus also occurs in an autosomal peroxisomal disorder.124 Gabapentin is effective in treating some forms of acquired pendular nystagmus.124–126 Averbuch-Heller et al. published a comprehensive review of the pathogenesis of acquired pendular nystagmus in 1995.127
Rarely, acquired pendular nystagmus in the adult becomes manifest with acquired amblyopia, as mentioned earlier. Scopolamine may be an effective treatment,128 but botulinum toxin is of limited efficacy in treating acquired pendular nystagmus.129 A review of current therapeutic approaches to various types of nystagmus and saccadic oscillations, based on known physiology and pharmacology, points out the need for more precise, double-blind studies.130
Miner's nystagmus is a rarity limited presumably to mine workers in the United Kingdom. This historical anachronism was described as a small-amplitude, horizontal, and vertical nystagmus that is often more pronounced in upward gaze. The pathogenesis of this putative dysfunction is uncertain, but functional contamination with voluntary “nystagmus” is suspected; a secondary gain setting is usually present.
Except for the possible dissociation between the two eyes, acquired pendular nystagmus may be similar to a pendular INS waveform; both can have associated head tremor and characteristically damp with eyelid closure. Studies into the pathogenesis of acquired pendular nystagmus have ruled out delayed visual feedback and increased gain in the visually enhanced VOR as causal factors.127
ACQUIRED HORIZONTAL JERK NYSTAGMUS
Vestibular
We generally delimit vestibular nystagmus as being consequent to dysfunction of the vestibular end-organ, nerve, or nuclear complex within the brain stem. It is a horizontal-torsional or purely horizontal, primary-position jerk nystagmus with a linear slow phase. The nystagmus intensity increases with gaze toward the fast phase (obeying Alexander's law); it decreases and, with central lesions, may reverse directions on gaze toward the direction of the slow phase. The symptom of vertigo usually coexists. As might be expected, acute lesions of the cerebellar flocculus (the vestibulocerebellum) can produce a similar nystagmus (see Chapter 10). For practical clinical purposes, the responsible lesion in vestibular nystagmus is located in either the end-organ, nerve, or brain stem. Such localization requires an appreciation of the manifestations of end-organ dysfunction. In normal subjects, some degree of nystagmus and vertigo develops when the labyrinth (end-organ) is stimulated with warm or cold water applied to the tympanic membrane. The direction of the resulting nystagmus, in terms of the fast (jerk) phase, can be remembered by the mnemonic “COWS” (Cold, Opposite; Warm, Same). Cold water in the left ear (or warm water in the right) induces a right-beating nystagmus; cold water in the right ear (or warm water in the left) induces a left-beating nystagmus. In addition, the subject experiences vertigo and, with eye closure, past-points with an outstretched arm and falls in a consistent direction on Romberg testing. The apparent direction of the vertiginous movement, whether of the environment or self, is always in the direction of the fast phase of the nystagmus. The past-pointing and Romberg fall are always in the direction of the slow phase. For example, with cold water placed in the external canal of the left ear, the subject develops a right-beating jerk nystagmus and experiences environmental or bodily movement to the right (paradoxically appearing to move continuously in one direction).131 With the eyes closed, the patient's attempts at pointing an outstretched finger at a target in front of him result in past-pointing to the left; on standing there is a tendency to fall to the left (in the direction of the slow phase of the nystagmus). This Romberg fall can be directionally altered by head turning: with the head turned to the left, the slow phase is directed toward the rear and the fall is backward; with the head turned to the right, the fall is forward.132
These manifestations of cold-water irrigation mimic the effects of a destructive lesion of the vestibular end-organ; warm-water irrigation mimics an irritative lesion. Clinically, most diseases of the end-organ create destructive effects. Irritative phenomena occur but are transient, often subclinical, and usually of interest only to the electronystagmographer. During an attack of Ménière's disease, there may be ipsilateral (jerk toward the affected side) nystagmus. Perhaps the most common cause of ipsilateral nystagmus secondary to end-organ disease is recovery nystagmus.133 Here, spontaneous nystagmus that occurs after a unilateral labyrinthine lesion may transiently reverse direction as some function is restored in the damaged end-organ. This probably reflects the compensatory “central rebalancing” of the vestibular nuclei. This compensation can also change a primary-position vestibular nystagmus (of peripheral or central etiology) to a paroxysmal positional nystagmus.134
A patient with unidirectional jerk nystagmus, vertigo in the direction of the fast-phase component, and past-pointing and Romberg fall in the direction of the slow component is suffering acute dysfunction of the vestibular end-organ on the side of the nystagmus slow phase. When the pattern of direction for the nystagmus, vertigo, past-pointing, and Romberg fall is not as just described but varies in some aspect, the symptom complex represents a central vestibular abnormality. Thus, in central vestibular disease, the vertigo may be in the direction of the slow phase of the nystagmus, and the past-pointing or Romberg fall may be toward the fast phase.
Other factors distinguish peripheral from central vestibular nystagmus. Pure vertical or pure torsional nystagmus is never peripheral and always represents central dysfunction. Similarly, pure horizontal nystagmus without a torsional component is suggestive of central disease.131 Nystagmus that is reduced in intensity by visual fixation is peripheral, whereas nystagmus due to central lesions is usually not reduced, and may even be enhanced, by fixation. Peripheral vestibular nystagmus is best visualized clinically behind Frenzel lenses (+20 diopters), which eliminate the inhibiting effects of visual fixation and magnify the eyes.135 A marked bidirectionality to the nystagmus (left-beating on left gaze and a similarly severe right-beating nystagmus on right gaze) is almost always central. Nystagmus may accompany episodic attacks of ataxia.136 Evidence has been presented supporting a specific chromosomal abnormality in some cases137 and brain stem lesions in others.138 Table 4 presents the differential features of peripheral and central vestibular nystagmus.
| Symptom or Sign | Peripheral (End-Organ) | Central (Nuclear) |
| Direction of nystagmus | Unidirectional, fast phase opposite lesion | Bidirectional or unidirectional |
| Purely horizontal nystagmus without torsional component | Uncommon | Common |
| Vertical or purely torsional nystagmus | Never present | May be present |
| Visual fixation | Inhibits nystagmus and vertigo | No inhibition |
| Severity of vertigo | Marked | Mild |
| Direction of spin | Toward fast phase | Variable |
| Direction of pastpointing | Toward slow phase | Variable |
| Direction of Romberg fall | Toward slow phase | Variable |
| Effect of head turning | Changes Romberg fall | No effect |
| Duration of symptoms | Finite (minutes, days, weeks) but recurrent | May be chronic |
| Tinnitus and/or deafness | Often present | Usually absent |
| Common causes | Infection (labyrinthitis), Ménière's disease, neuronitis, vascular, trauma, toxicity | Vascular, demyelinating, and neoplastic disorders |
Gaze-Evoked (Gaze-Paretic) Nystagmus
Gaze-evoked nystagmus is elicited by the attempt to maintain an eccentric eye position, and it is the most common form of nystagmus encountered in clinical practice. Patients recovering from a central gaze palsy show a phase in which lateral gaze movement is possible but cannot be maintained in the deviated position; that is, the eyes drift back slowly toward primary position (see Chapter 10). A corrective saccade repositions the eyes eccentrically, and repetition of this pattern produces nystagmus, aptly designated “gaze-paretic.” This was a clinical description that was not based on oculographic findings. Information about the role of the brain stem neural integrators (see Chapter 9) led to the presumption that a defective integrator would result in the inability of the eyes to maintain an eccentric position, causing them to drift toward the center with a decreasing-velocity exponential waveform. This is indeed the waveform that defines the “gaze-paretic” subtype of gaze-evoked nystagmus, which is particularly prevalent in patients with cerebellar disease that especially involves the flocculus (see Chapter 10). There appears to be an inherent “leakiness” of the brain stem neural integrators, namely, a tendency to drift from a given firing level. The cerebellar flocculus normally corrects for this drift. With a floccular lesion, the leakiness and drift are unchecked, and gaze-paretic nystagmus develops. If the integrator leak is small and the time-constant long, a gaze-paretic nystagmus could have a slow phase shape that is linear rather than a decreasing-velocity exponential. Such nystagmus cannot be designated as gaze-paretic with any degree of certainty and may only be described as gaze-evoked.
In summary, the term gaze-paretic nystagmus is restricted to a subgroup of gaze-evoked nystagmus with a decreasing-velocity exponential slow phase. It is integrator nystagmus with a defect in the step function of neural firing frequency constituting the pathophysiologic mechanism. The same integrators are probably responsible for smooth-pursuit eye movements, which seem to be invariably abnormal in patients with gaze-paretic nystagmus.
The most common cause of pathologic, bidirectional, gaze-evoked nystagmus is sedative or anticonvulsant medication. The nystagmus fast phase is always in the direction of gaze (toward the right on right gaze, left-beating on left gaze, and upbeating on upward gaze; down gaze is usually without nystagmus). In the absence of drugs, horizontal gaze-evoked nystagmus with linear slow phases can be localized only enough to indicate brain stem or, if unilateral, labyrinthine dysfunction. Analysis of the associated neurologic signs and symptoms would be required for more precise localization.
Gaze-evoked vertical nystagmus almost always coexists with the horizontal variety. Primary-position vertical jerk nystagmus (upbeat and downbeat) is discussed later.
SPECIAL NYSTAGMUS TYPES
Physiological (End-Point)
There are three basic types of nystagmus that are regarded as normal (physiological) phenomena.139
Fatigue nystagmus begins during extended maintenance of an extreme gaze position and occurs in up to 60% of normals when horizontal gaze is maximally deviated for a time exceeding 30 seconds. It may become increasingly torsional with prolonged deviation effort and may be greater in the adducting eye. Fatigue nystagmus is not a clinically important phenomenon, because routine examinations do not include the maintenance of far eccentric gaze.
Unsustained end-point nystagmus is certainly the most frequently encountered physiologic nystagmus. Its characteristics have never been studied quantitatively. All experienced clinicians recognize that a few beats of nystagmus are within perfectly normal limits at gaze deviations of 30° or more.
Sustained end-point nystagmus begins immediately upon, or within several seconds of, reaching an eccentric lateral-gaze position. It occurs in more than 60% of normal subjects with horizontal-gaze maintenance greater than 40°. Quantitative oculography reveals that physiologic nystagmus can begin with only a 20° deviation139 and is almost universal at deviations of 40° or more.140 The slow phase is linear, except with an extreme 40° to 50° deviation, in which a decreasing-velocity exponential may develop. The nystagmus may be different in the two eyes, but it is symmetric in the two lateral directions. The amplitude of physiologic nystagmus does not exceed 3°.139 Thus, small-amplitude gaze-evoked nystagmus may be a normal phenomenon, provided the slow phase, with gaze angles up to 40°, is linear. The onset of end-point nystagmus is related to slow drift velocity,141 and the reduction of drift velocity during fixation (probably by the fixation subsystem) inhibits the nystagmus. Gaze-evoked nystagmus is by necessity “pathologic” if any of three features are present: (1) asymmetry in the two directions, (2) amplitude of 4° or more, or (3) exponential slow phase within a gaze angle of less than 40°.
DissociatedNystagmus in which the two eyes show a significant asymmetry in either amplitude or direction is designated “dissociated.” The most common type of dissociation is in internuclear ophthalmoplegias where it is most marked in the abducting eye (see Chapter 10). Abduction nystagmus, which is sometimes designated by the confusing term ataxic nystagmus, is not really a nystagmus. This saccadic oscillation is secondary to lesions of the medial longitudinal fasciculus and is discussed under Saccadic Pulses/Pulse Trains later in this chapter and in Chapter 10.
The pendular nystagmus in patients with multiple sclerosis is usually dissociated. There are a variety of nystagmus dissociations with lesions of the posterior fossa (e.g., asymmetric vertical nystagmus greater in one eye on looking up and in the other eye on looking down).
TorsionalTorsional nystagmus describes a torsional movement of the globe about its anteroposterior axis; the term rotary nystagmus is used interchangeably. Most nystagmus consequent to vestibular end-organ dysfunction has a torsional component admixed with a major horizontal or vertical nystagmus. A purely torsional nystagmus never occurs with vestibular end-organ disease. When of small amplitude, torsional nystagmus may reflect a medullary lesion. Larger-amplitude torsional nystagmus may be congenital, but when it is acquired it often indicates diencephalic (thalamic) involvement, in which case it is the underlying pattern in see-saw nystagmus. Torsional nystagmus can be classified into two groups.142 In one the nystagmus is in primary position, and in the other it is gaze evoked. The etiologies of both are demyelinating, vascular, or neoplastic.
For consistency, nystagmus in the torsional plane should be defined with respect to the subject, not the observer. When a subject's nystagmus beats to the right, it is called jerk-right nystagmus; if it beats toward the forehead, it is upbeat (even if the subject is in the head-hanging position). Therefore, when the nystagmus fast phases bring the top of the eye toward the subject's right shoulder, it is clockwise nystagmus. Clinical descriptions of torsional nystagmus, made from the observer's point of view, deviate from the accepted convention that applies to both horizontal and vertical movements. Maintaining the subject-based directions reduces confusion, eases understanding of anatomic substrates, and anchors the subject's perception of oscillopsia to the direction of his nystagmus. Just as leftward horizontal slow phases cause rightward, perceived world motion, clockwise torsional slow phases cause counterclockwise, perceived world motion (see elliptic nystagmus, discussed later).
See-SawSee-saw nystagmus is characterized by a conjugate, pendular, torsional oscillation with a superimposed disjunctive vertical vector. The intorting eye rises, and the opposite, extorting eye falls. Repetition of this sequence in the alternate direction provides the see-saw effect. The torsional movements predominate in all fields of gaze, but the see-saw feature may be restricted to the primary position or, more commonly, to downward or lateral gaze. See-saw nystagmus can be of the jerk type (with one phase being slow and the other fast) with unilateral meso-diencephalic lesions.143 Most patients with acquired pendular see-saw nystagmus have bitemporal hemianopias consequent to large parasellar tumors expanding within the third ventricle. It is occasionally evoked transiently after blinks or saccades.144 Upper brain stem vascular disease and severe head trauma are the next most common etiologies. Posttraumatic see-saw may be temporarily abolished by ingestion of alcohol.145 Rarely, the nystagmus is associated with septo-optic dysplasia, an Arnold-Chiari malformation,146 multiple sclerosis,147 or loss of vision alone.148 See-saw nystagmus probably reflects diencephalic (thalamic) dysfunction possibly of a pathway or pathways from the zona incerta to the interstitial nucleus of Cajal. A study of visual–vestibular interaction concluded that see-saw nystagmus resulted from a loss of retinal error signals secondary to disruption of chiasmal crossing fibers.149
Congenital see-saw nystagmus manifests either in constant vertical disjugacies without a significant torsional component or in conjugate torsional nystagmus with a vertical component said to be opposite that of the acquired variety; that is, the intorting eye falls while the extorting eye rises. However, all cases of congenital see-saw nystagmus that have been recorded follow the rule that the rising eye intorts while the falling eye extorts. Congenital see-saw nystagmus is also seen in canine and human achiasma (see Albinism and Achiasma section later in the chapter). An unusual instance of congenital see-saw nystagmus in two mentally retarded adult siblings, associated with retinitis pigmentosa, also had the typical vertical/torsional relationship associated with acquired see-saw.150
The ocular tilt reaction, described in Chapter 10, is actually one-half of a see-saw cycle.
Convergence/Convergence-EvokedThe act of convergence usually damps nystagmus, particularly the congenital type.151 Convergence can also damp151 or evoke152 lid nystagmus and may damp or enhance downbeat nystagmus.153 Upbeat nystagmus may change to downbeat with convergence.154
Repetitive divergence is a term describing a slow divergence movement followed by a rapid convergence to the primary position. The movements occur at irregular intervals, distinguishing this from nystagmus.155 In the single reported instance of this phenomenon in a patient with hepatic encephalopathy, an entire cycle lasted from 4 to 10 seconds, and the interval between cycles was 1 to 15 seconds.
Conjugate nystagmus evoked by convergence (convergence-evoked nystagmus) must be distinguished from convergence nystagmus. Convergence-retraction nystagmus as a manifestation of the dorsal midbrain syndrome is discussed in Chapter 10; here, the initiating convergence movements are saccadic156 and, thus not a true nystagmus. Fast divergent movements, followed by a slow convergence associated with epileptic electroencephalographic activity, occurred in a neonate with an intraventricular hemorrhage.157
With the exception of pure convergence nystagmus in infants with spasmus nutans, true pendular convergence nystagmus is uncommon. We observed it in a patient with presumed progressive supranuclear palsy who had paralysis of all volitional eye movements. In retrospect, however, the patient probably had central nervous system Whipple's disease, perhaps the most common cause of pendular convergence nystagmus (see Chapter 10). We studied three patients with convergence nystagmus with phase shifts of about 180° in both the horizontal and torsional planes with conjugate nystagmus in the vertical plane.158 Convergence increased the nystagmus in two of the patients. The waveforms were either sinusiodal or complex sums of sinusoids, and in one patient they were cycloidal. Unlike the pseudocycloid waveform of INS, there were no initiating saccades to these cycloidal movements. We hypothesized that the low-frequency convergence nystagmus resulted from a visually mediated vergence instability, whereas high-frequency forms might have arisen from instability of brain stem pathways associated with vergence.
Nystagmus evoked by convergence is unusual and may be either conjugate or disjugate, congenital or acquired.159 In two cases reported, no definite clinical correlation could be made with a specific lesion. The neuropathologic examination revealed no morphologic explanation for nystagmus in the patient with congenital convergence-evoked nystagmus; the patient with the acquired form had demyelinating disease with a spastic paraparesis and no cranial nerve abnormality other than the ocular motor findings.159 Horizontal pendular nystagmus rarely is evoked by accommodative vergence.160 This is to be distinguished from psychogenic flutter (the so-called voluntary nystagmus discussed later), which is often best induced when the eyes are slightly converged.
Periodic AlternatingPeriodic alternating nystagmus (PAN) is an extraordinary ocular motor phenomenon in which a persisting horizontal jerk nystagmus periodically changes directions. PAN may be congenital or acquired. The congenital variety (i.e., INS nystagmus), which may be associated with albinism,161,162 has slow-phase waveforms of both linear and increasing velocity, usually lacks the well-defined stereotyped periodicity seen in acquired PAN (i.e., it is asymmetric (a)periodic alternating nystagmus—APAN), and can persist for many minutes in either direction or spontaneously change direction after a few seconds.163,164 The addition of variable pendular nystagmus to these APAN jerk waveforms can produce complex waveforms that may mimic those of FMNS nystagmus.165–167 The periodicity of INS APAN is markedly influenced by changes in gaze position, supporting the hypothesis that the direction reversals are a result of a temporal shift in the null zone (Fig. 17).80
Patients with INS and a varying head turn should be examined for APAN.
In contrast, the usual, fixed sequence in acquired PAN consists of about 90 seconds of nystagmus beating in one direction, 10 seconds of a neutral phase in which the eyes stop or beat downward irregularly, and 90 seconds of beating in the opposite direction. This periodicity is continuous during waking hours and may prevail during sleep. Some patients demonstrate asymmetries in the timing of the two major phases, but the basic pattern for each patient is usually invariable.
In a detailed clinical and control-system study, Leigh and colleagues168 proposed that PAN arises from: (1) a defect in the brain stem neural networks that generates slow phases of vestibular and optokinetic nystagmus, (2) the action of an adaptive network that normally acts to null prolonged, inappropriate nystagmus, and (3) an inability to use retinal-error velocity information. They proposed a control system model that denied access of visual signals to the OKN-vestibular system. This model is particularly appealing because of the occasional relationship between impaired vision and PAN. Support for their hypothesis of impairment in the velocity storage element was presented by Furman et al.,169 who studied four PAN patients. PAN has occurred after bilateral vitreous hemorrhages (associated with a massive subarachnoid hemorrhage) and after cataracts and disappeared after bilateral vitrectomy and cataract surgery, respectively. Ablation of the nodulus and ventral uvula of the cerebellum in monkeys produces PAN.170
Although numerous conditions have been associated with PAN, including INS, head trauma, vascular insufficiency, encephalitis, syphilis, multiple sclerosis, spinocerebellar degenerations, and posterior fossa tumors, particular attention should be directed to an abnormality of the craniocervical junction, such as a Chiari malformation. PAN may coexist with downbeat nystagmus, which also suggests an abnormality in the same location.
Campbell described PAN secondary to phenytoin intoxication in a patient with alcoholic cerebellar degeneration.171 The antispasticity drug baclofen abolishes acquired PAN but has no effect on the INS variety.172 The drug abolished experimentally created PAN in the monkey,170 as well as a single case of aperiodic alternating nystagmus in a patient with vertebrobasilar insufficiency.173
PAN may be associated with a periodic alternating skew deviation.174 Periodic alternating gaze deviations, with and without associated alternating nystagmus or alternating head turning, are rare, related phenomena.175
DownbeatDownbeat nystagmus is defined as nystagmus in primary gaze position, with the fast phase beating in a downward direction. Patients with brain stem disease or drug intoxications usually lack gaze-evoked downward nystagmus despite nystagmus in all other fields of gaze. Thus, nystagmus beating downward in the primary position is a striking phenomenon and is highly suggestive of a craniocervical junction lesion, such as a Chiari malformation.176,177 Downbeat nystagmus is usually of sufficient amplitude in primary position to cause oscillopsia, and, contrary to Alexander's law,2 it is not maximum at the extreme of downward gaze. Rather, it is usually of maximal intensity when the eyes are deviated laterally and slightly below the horizontal;177 the nystagmus may be intermittent.
The other major cause of downbeat nystagmus is spinocerebellar degeneration. A variety of miscellaneous conditions may also be associated with downbeat nystagmus,153,177 including anticonvulsants, alcohol and lithium intoxication, magnesium deficiency, B12 deficiency, brain stem encephalitis, alcoholic cerebellar degeneration, and brain stem or cerebellar infarcts.178 Rarely, downbeat nystagmus is secondary to upper brain stem or supratentorial lesions, or as a form of INS nystagmus. Schmidt's179 article is an excellent clinical review of downbeat nystagmus.
Downbeat nystagmus was regarded as the prototype of nystagmus secondary to a “pursuit defect,” but studies have cast serious doubt as to whether a unidirectional pursuit defect is a cause of any form of nystagmus.176 The slow-phase waveform of downbeat nystagmus can vary from a linear, to an increasing-velocity, to a decreasing-velocity exponential in the same patient, presumably reflecting short-term gain changes by cerebellar compensation for leaky brain stem neural integrators.5 One patient with a pseudocycloid waveform damped with convergence, prompting treatment with base-out prism spectacles.153 This has subsequently proved superior to retinal image stabilization as a means of reducing oscillopsia.180 Downbeat is usually increased in intensity with head-hanging176 as well as linear acceleration, and occasionally convergence. The mechanism of downbeat is postulated to be disruption of central vestibular pathways,176 possibly involving specific connections from the otoliths or an asymmetry of the vertical semicircular canal gains.181
UpbeatPrimary-position nystagmus with the fast phase beating upward may reflect drug or other intoxications182 but usually indicates structural disease, usually of the brain stem.183–185 The location of the lesions in patients with upbeat nystagmus after meningitis or Wernicke's encephalopathy is uncertain. Büttner et al. discussed the localizing value of upbeat and other types of nystagmus.186 With convergence, upbeat may enhance184 or convert to downbeat.154 The slow-phase waveform is usually linear but may be an increasing-velocity exponential.
Upbeat may be enhanced184 or suppressed187 by head tilt. Therefore, it is conceptualized hypothetically as secondary to a disruption of central otolithic pathways; the effects of convergence on the nystagmus are presumed to reflect vergence effects on these pathways. Tobacco smoking causes upbeat nystagmus in normal human subjects in darkness, but the nystagmus is suppressed by visual fixation.188 The mechanism is presumed to be the excitatory effects of nicotine on central vestibular pathways.
ReboundRebound nystagmus is either the diminution and direction change of gaze-evoked horizontal nystagmus during sustained ocular deviation or a horizontal gaze-evoked nystagmus that, on refixation to primary position, transiently beats in the opposite direction. The sign is often present in patients with cerebellar disease.189 Rebound nystagmus is conceptualized as a smooth eye movement bias generated to oppose gaze-evoked centripetal drift of the eyes. Normal subjects may demonstrate rebound nystagmus after prolonged far lateral gaze if the lights are shut off the moment the eyes are returned to primary position and may even occur in normals during fixation in a fully lit room.140 Rebound and centripetal nystagmus may occur in Creutzfeldt-Jakob disease.190
Circular, Elliptic, and ObliqueCircular nystagmus, sometimes confusingly mislabeled as “rotary,” is a form of pendular nystagmus in which the globe oscillates continuously in a fine, rapid, circular path. Unlike torsional nystagmus, in which the 12 o'clock meridian of the limbus torts laterally, this point maintains its position in circular nystagmus. The nystagmus represents the summation of simultaneous, equal-amplitude, horizontal, and vertical pendular oscillations that are 90° out of phase. Elliptic nystagmus is produced when the horizontal and vertical oscillations are 90° out of phase but are of unequal amplitude. Analysis of the vertical and horizontal pendular nystagmus movements, which summate to form circular and elliptic nystagmus, indicates that a true circular pattern is rarely sustained. More often, the nystagmus varies between elliptic and circular. Unlike all other types of nystagmus, the oscillopsia induced by elliptical nystagmus is in the same direction as the slow phases.191 Thus, if the subject's eye moves in a clockwise, circular-elliptic manner (from the subject's point of view), the direction of perceived world motion will also be clockwise. Acquired circular-elliptic nystagmus occurs in multiple sclerosis, is often dissociated in the two eyes, and almost always coexists with truncal or extremity ataxia. Drug treatment with isoniazid may be effective.191 This type of nystagmus is often part of the INS and is identical in the two eyes in patients otherwise free of neurologic signs.
Oblique or diagonal nystagmus results when simultaneous pendular, horizontal, and vertical vectors are either in phase or 180° out of phase. The angle of the diagonal vector depends on the relative amplitudes of the horizontal and vertical components. This type is more often acquired than infantile and has the same significance as acquired circular-elliptic nystagmus.
CervicalThe literature on cervical (cord) nystagmus contains numerous examples of spontaneous or positional nystagmus allegedly secondary to lesions of the cervical spinal cord or roots.192–194 Although this form of positional nystagmus occurs only rarely, it is claimed to support the “legitimacy” of dizziness in patients who have sustained “whiplash” injuries and are involved in litigation. For that reason, the entire concept of cervical nystagmus has become highly suspect.193,194 We have never seen a patient with convincing cervical nystagmus.
Muscle-Paretic (Myasthenic)A paretic eye muscle, from whatever cause, can fatigue quickly during contraction, and muscle-paretic nystagmus can be observed. This is often evident as gaze-evoked nystagmus in myasthenia gravis, in which there is usually asymmetry between the two eyes.195 Another form of oscillation in myasthenia is nystagmus of the abducting eye (may be saccadic pulse trains, discussed below), coexisting with paresis of adduction; this mimics an internuclear ophthalmoplegia. Here the oscillation is not due to lateral rectus paresis, but rather to excessive innervation by increased central gain, the result of paresis of the contralateral yoke medial rectus. Cessation of both muscle-paretic and contralateral yoke nystagmus in myasthenia usually follows administration of anticholinesterase medication.
LidLid nystagmus is a rhythmic, upward jerking of the upper eyelids that usually consists of the normally coordinated movements of the lids and eyes during vertical ocular nystagmus. Several types of pathologic lid nystagmus are recognized. Type I, with no specific localizing value coexists synchronously with vertical ocular nystagmus, but the amplitude of the lid movements exceeds significantly that of the eyes. Type II is evoked by lateral gaze and is characterized by rapid phasic twitches of the lids that are synchronous with the fast phases of horizontal ocular nystagmus. This second type may be a sign of the lateral medullary syndrome and can be inhibited by near-effort.151 Type III lid nystagmus is provoked by ocular convergence.152,196 In a patient with this type of nystagmus studied pathologically, there was a large area of subacute demyelinization in the rostral medulla. Lid nystagmus, differing from Types I–III, may occur with midbrain lesions.197
EpilepticNystagmus associated with epileptic activity includes retraction, pendular, torsional, and divergent-convergent forms.157 At times, the pupils may constrict and dilate synchronously with the nystagmus.198 The usual form of epileptic nystagmus is horizontal jerk with the fast phase contralateral to a posterior parietal focus, but a vertical nystagmus may also be present.199 The eyes may tonically deviate toward or away from the side of the epileptic focus.200 The decreasing-velocity slow phases suggest that a gaze-holding failure201 causes the slow phases, and the relationship of the ictal activity suggests activation of the saccadic system (see Chapter 10).199
INDUCED NYSTAGMUS
Caloric
We previously described the characteristics of caloric-induced vestibular nystagmus. With unilateral irrigation, the nystagmus is horizontal, torsional, or oblique, depending on the position of the head. Bilateral simultaneous caloric stimulation produces vertical nystagmus; the direction of the fast (jerk) phase can be remembered by the mnemonic CUWD (pronounced “cud” and designating Cold, Up; Warm, Down).
Traditionally, caloric nystagmus was regarded as being evoked entirely by thermal convection of the endolymph. However, the fact that such nystagmus exists in the zero-gravity conditions of outer space indicates the necessity of an alternative mechanism,202 which,203 in addition to the convection mechanism, is a direct temperature effect on the canal's sensory apparatus that is independent of head orientation. A review of the technique of quantitative bithermal caloric testing for evaluating vestibular function is provided elsewhere. Caloric and head rotation (doll's) are useful tests in the evaluation of comatose patients.204
RotationalRotating or accelerating head movements induce motion of endolymph in the semicircular canals, with a resultant jerk nystagmus. If the axis of rotation passes through the upright head, as happens with the Bárány chair, the nystagmus fast phase is in the same direction as head (or chair) rotation. After cessation of the rotation, the postrotary nystagmus is in the opposite direction. Rotational nystagmus is used in evaluating the ocular motor system in infants (see Chapter 3, Fig. 2), and to evaluate vestibulo-ocular gain and other aspects (visual suppression of the VOR) of the vestibular system.
PositionalIn patients complaining of vertigo related to shifts in position of the head or body, positional nystagmus should be considered. The test is performed by observing for nystagmus produced when the patient rapidly reclines from a sitting to a supine position, with the head either turned alternately to one side and then the other, or hyperextended (hanging). Two types of nystagmus, peripheral and central, can be differentiated (Table 5).
| Features | Peripheral | Central |
| Latency | 3–40 sec | None; nystagmus begins immediately |
| Fatigability | Yes | No |
| Rebound | Yes | No |
| Habituation | Yes | No |
| Intensity of vertigo | Severe | Mild |
| Reproducibility | Poor | Good |
| Directionality and Waveforms | Stereotyped | Variable |
Peripheral positional nystagmus is associated with marked vertigo, which begins after a delay of 2 to 20 seconds. The nystagmus and vertigo eventually fatigue, usually within 1 minute. A rapid return to the sitting position causes another brief burst of nystagmus and vertigo (“rebound”). Repositioning in the provocative supine posture again induces nystagmus and vertigo, but to a lesser extent. Repetition of the shifts of posture ultimately results in diminution and disappearance of the nystagmus and vertigo. Characteristic of the peripheral variety is variable reproducibility: the nystagmus and vertigo may not be present every time the offending position is attained. Whereas latency, fatigability, and habituation have traditionally been regarded as the major features distinguishing peripheral from central positional nystagmus, Baloh et al.205 emphasize that the nystagmus vector is the critical determinant. Positional nystagmus with a torsional-vertical vector, with the torsional component greater in the undermost eye and the vertical component (upbeating) greater in the uppermost eye, is always peripheral, irrespective of the nature of latency, fatigability, and habituation. The torsional component increases when gaze is directed toward the down eye, and the upbeating component increases with gaze directed toward the upper eye.205 With head hanging (extended), a downbeating nystagmus (fast phase beating toward the chin) probably has the same significance as downbeating nystagmus with the patient upright.
Central positional nystagmus, which is invariably reproducible, begins immediately on movement to the provoking position; the nystagmus neither fatigues nor habituates, and the vertigo is usually mild. Positional nystagmus that changes direction while the head position remains fixed has long been regarded as a central sign, also with peripheral disease and even in normal subjects.206
The peripheral variety, designated “benign paroxysmal positional vertigo” (BPPV), is indicative of labyrinthine disease, and neuroradiologic procedures are rarely indicated. The most common specific etiology is head trauma. However, no cause is found in most cases. The malady increases in prevalence with age. Nystagmus, identical to the benign peripheral variety, may result from the slow drift produced by central vestibular compensation consequent to either peripheral or central vestibular dysfunction.134
The most reasonable etiology of BPPV is “canalolithiasis.” The hypothesis is that otoconia detach from a utricle, congeal to form a “plug,” and float freely in the endolymph of a posterior semicircular canal. Because the plug is of greater specific gravity than the endolymph, the canal becomes a gravity receptor, and symptoms and signs are produced when the head is in a particular position. This concept has led to a form of “physical therapy” in which repetitive movement, or a single provocative movement, dislodges the displaced otoconia.207 Most patients with the disorder recover spontaneously after several weeks. However, if a person has persistent symptoms and the attacks are consistently evoked with the head in the offending position, a liberatory maneuver is performed. With BPPV, in addition to positional vertigo, patients develop symptoms on arising, bending over, leaning forward, and with head movements while upright.
The central type of positional nystagmus is often associated with neoplastic, vascular, demyelinating, or degenerative disorders involving the brain stem or cerebellum.208 Intermediate forms of positional nystagmus, such as those that fatigue and habituate but have no noticeable latency, should raise suspicions of central disease. The difference between central paroxysmal positioning nystagmus and that of BPPV is in the direction; the latter beats in the direction aligned with the affected canal plane and any other direction indicates a central origin.209
Testing for positional nystagmus is fairly standardized and does not require electronystagmography. We prefer self-illuminated Frenzel glasses (highly convex lenses, +20 D, which blur vision), but testing can also be performed in a completely darkened room, with the examiner periodically observing the eyes with a dim light. Routine testing for positional nystagmus in patients without the specific symptom of positional vertigo does not generally yield useful results.
The BPPV just mentioned is due to dysfunction of a posterior semicircular canal. Some patients have horizontal-canal BPPV210 or anterior-canal BPPV,211 which have different liberatory maneuvers.212 The term positional nystagmus is often used interchangeably with positional vertigo in designating a syndrome, and positional and positioning are used interchangeably to describe nystagmus.
OptokineticThe localizing value of OKN with cerebral hemispheric lesions is discussed in Chapter 10. OKN is used for several other important functions in clinical neuro-ophthalmology. It provides evidence of at least gross levels of visual function in infants or patients with functional visual loss. As mentioned in Chapter 10, OKN in the downward direction is used to induce convergence-retraction “nystagmus,” and horizontal OKN is used to demonstrate the adduction insufficiency in internuclear ophthalmoparesis. “Inversion” of OKN is diagnostic of INS. OKN can also be used to diagnose oculomotor nerve misdirection.
Drug/Toxin-InducedDrug-induced nystagmus is a common sequela of barbiturate, tranquilizer, phenothiazine, and anticonvulsant therapy.213 The nystagmus is generally gaze evoked and is usually horizontal or horizontal-torsional in direction. Vertical nystagmus is often present on upward gaze and only rarely on downward gaze. At times the nystagmus may be dissociated in the two eyes despite the lack of structural disease to account for the asymmetry. Although primary-position nystagmus is usually indicative of severe drug intoxication, it may persist for 10 hours after the oral ingestion of only 100 mg of secobarbital.134 In addition, this amount of secobarbital can produce positional nystagmus. As mentioned, lithium can produce downbeat nystagmus,214,215 tobacco can induce upbeat nystagmus,188 and severe alcohol intoxication can produce downbeat nystagmus.216 Toluene (glue-sniffing) can induce pendular nystagmus with both horizontal and vertical components.217
Unfortunately, that alcohol can produce horizontal gaze-evoked nystagmus has led to a “roadside sobriety” test conducted by law-enforcement officers.218 Nystagmus as an indicator of alcohol intoxication is fraught with extraordinary pitfalls: many normal individuals have physiologic end-point nystagmus; small doses of tranquilizers that wouldn't interfere with driving ability can also produce nystagmus; nystagmus may be congenital or consequent to structural neurologic disease; and often a neuro-ophthalmologist or sophisticated oculographer is required to determine whether nystagmus is pathologic. Such judgments are difficult for experts to make under the best conditions and impossible to make accurately under roadside conditions. It is unreasonable to have cursorily trained law officers using the test, no matter how intelligent, perceptive, and well meaning they might be. As noted, meticulous history taking and drug-screening blood studies are often essential in evaluating patients with nystagmus.
SPECIAL ANATOMIC CATEGORIES
Acoustic Neuroma
Schwannomas of the eighth nerve grow so slowly that adaptive mechanisms often obscure clinical vestibular manifestations. Vestibular nystagmus beating contralateral to the lesion may be present, particularly if fixation is eliminated. As the tumor expands to compress the brain stem, a slow, gaze-evoked ipsilateral nystagmus is often added. The combination of a small-amplitude, rapid primary-position jerk nystagmus beating contralateral to the lesion and a slower, larger-amplitude, gaze-evoked (Bruns') nystagmus ipsilateral to the lesion also occurs with other extra-axial masses, including cerebellar tumors, compressing the brain stem. Rarely, Bruns' nystagmus is inverted.219
Lateral Medullary SyndromeThe lateral medullary syndrome (Wallenberg) is a distinctive constellation of signs. The nystagmus in this syndrome tends to be stereotyped. With the eyes open there is horizontal-torsional jerk nystagmus beating contralateral to the lesion; when recorded with the eyes closed, the nystagmus beats ipsilateral to the lesion. Other rare manifestations, confined to single cases, are gaze-evoked eyelid and ocular nystagmus inhibited by the near reflex151 and horizontal gaze-evoked monocular downbeat nystagmus.
An extraordinarily dramatic eye-movement abnormality, “saccadic lateropulsion,” may occur with lateral medullary infarction. Eye movements as well as body and limb movements are biased toward the side of the lesion (ipsipulsion). The ocular motor abnormality is most striking during shifts of fixation; all ipsilateral saccades are too large (hypermetric), whereas those to the opposite side are too small (hypometric). Spontaneous drifts to the side of the lesion are gaze dependent and may reflect disruptions of the gaze-holding mechanism.220 There is also reduced capability to adjust saccadic gain in response to the dysmetria,221 significant tilt in the subjective vertical, with ipsilateral excyclotropia,222 and torsional nystagmus and torsipulsion.223,224 Upward or downward refixations veer ipsilaterally along an oblique rather than a vertical line. Lateropulsion away from the side of the lesion (contrapulsion) has occurred with a unilateral disorder of the rostral cerebellum.225 Both types of lateropulsion are regarded as saccadic instabilities (Table 6). The gaze deviation may reflect increased inhibition of the ipsilateral vestibular nucleus and ipsipulsion may reflect decreased excitation of contralateral premotor areas in the pontine paramedian reticular formation.226
TABLE 6. Saccadic Intrusions and Oscillations*
| Bobbing/dipping | Saccadic lateropulsion |
| inverse bobbing | ipsipulsion |
| reverse bobbing | contrapulsion |
| Convergence-retraction “nystagmus” | Saccadic pulses/pulse trains |
| “nystagmus” retractoris | abduction “nystagmus” |
| Double saccadic pulses (single/multiple) | ataxic “nystagmus” |
| saccadic intrusions/oscillations | saccadic intrusions/oscillations |
| Dynamic overshoot | stepless saccades |
| “quiver” | Square-wave jerks/oscillations |
| Dysmetria | Gegenrucke |
| Flutter | hopping “nystagmus” |
| Flutter dysmetria | “lightening eye movements” |
| Macro saccadic oscillations | myoclonus |
| Myoclonus | saccadic intrusions/oscillations |
| laryngeal “nystagmus” | Zickzakbewegungen |
| “lightning eye movements” | Square-wave pulses (bursts/single) |
| pharyngeal “nystagmus” | “macro square-wave jerks” |
| Opsoclonus | Kippdeviationen/“Kippnystagmus” |
| “dancing eyes” | “pendular macro-oscillations” |
| “lightning eye movements” | saccadic “nystagmus” |
| saccadomania | saccadic oscillations/intrusions |
| Psychogenic flutter | Staircase saccadic intrusions |
| hysterical flutter | Superior oblique myokymia |
| hysterical “nystagmus” | |
| “ocular fibrillation” | |
| “ocular shuddering” | |
| psychological “nystagmus” | |
| voluntary flutter | |
| voluntary “nystagmus” |
*Synonyms and other terms are indented under either the preferred or the more inclusive designation; quoted terms are erroneous or misleading.
ALBINISM AND A(HEMI)(HYPO)CHIASMA
Ocular albinism is associated with anomalous visual projections that result in a variety of eye movement disturbances, with considerable intersubject variability. These individuals may have pendular or jerk nystagmus, absent OKN, “inverted” pursuit, or “defective” pursuit (see section on INS) when targets are projected onto the temporal half-retina.227 Periodic alternating nystagmus may also occur.161,162 Albinism may also exist in the absence of any nystagmus.228,229
Achiasma is a very rare condition, first recognized in dogs (ca. 1974) and then in humans (ca. 1992). It is associated with the combination of INS and see-saw nystagmus; we first diagnosed the see-saw nystagmus from videotape (in canines in 1991 and in a human in 1993) and subsequently studied it using eye-movement recordings.50,230–235 In achiasma, all retinal fibers remain ipsilateral, passing to the ipsilateral lateral geniculates and visual cortexes. Thus, each visual cortex has a representation of the entire visual field, but stereopsis is impossible; there is no bitemporal hemianopia. Figure 18 shows both the horizontal INS nystagmus and the vertical component of the see-saw nystagmus in an achiasmatic canine (Fig. 18A) and an achiasmatic human (Fig. 18B). The horizontal and vertical waveforms of the canines' nystagmus and the human's vertical nystagmus were pendular, whereas the human's horizontal waveforms were pendular with foveating saccades (PFS) or pseudopendular with foveating saccades (PPFS). Because the achiasmatic human subject was not always able to achieve well-developed foveation, as shown by her inability to repeatedly foveate the target with her fixating right eye, a larger than normal foveation window for the NAFX would be required to assess the best potential visual acuity for this segment. Unlike INS, which may or may not be associated with a particular sensory deficit, the see-saw nystagmus appears to be directly associated with achiasma in both species. In achiasma, there is no lesion, indeed no chiasm, and disruption of thalamic inputs to the nucleus of Cajal, postulated as the cause of acquired see-saw nystagmus, cannot be causal. In hemichiasma, abnormalities at the chiasm prevent decussation of the retinal fibers from one eye, the other decussating normally.236 Whereas achiasma appears to be sufficient for see-saw nystagmus, hemichiasma is not. Ocular motility and retinal studies of humans with achiasma and hypochiasma (a maldeveloped chiasm) revealed INS, see-saw nystagmus, strabismus, reduced vision, optic nerve dysplasia.237 Because of the rarity of congenital see-saw nystagmus and its identification in both canines and humans with achiasma and in one of two canines with hemichiasma, we regard its presence in infants as a diagnostic sign of possible achiasma or hemichiasma.238 The presence of see-saw nystagmus in an infant is a strong indication for imaging of the optic chiasm to rule out achiasma or structural abnormalities (hypochiasma) conducive to hemichiasma.
Leber's congenital amaurosis may be caused by several abnormalities, among which is a defect in retinal pigment epithelium due to absence of the RPE65 gene. Recent studies of a new genetic treatment for this blinding disease conducted on members of a colony of RPE65-deficient canines have shown initial success.239 Within several weeks of a subretinal injection of healthy RPE65 genetic material in an adenovirus solution, the previously absent ERG and pupillary responses were markedly improved, as was the visually guided behavior of the injected dogs. Comparisons of the pre- and postinjection ocular motility recordings revealed that the INS nystagmus damped to such an extent that it was clinically absent.240 The nystagmus scanpaths in Figure 19 show the dramatic difference between the INS nystagmus in two litter mates, one untreated and the other genetically treated. Note that the elliptical trajectories of the untreated nystagmus were in opposite directions; prior to treatment, the clinical appearance of the treated dog's nystagmus was judged to be equal to that of the untreated litter mate. Further studies are being conducted to assess both the effectiveness and safety of this promising genetic treatment to prevent blindness in affected children.
Some of the various eye signs seen with cerebellar system disease are discussed in previous sections and in Chapter 10. Descriptions of others will follow.