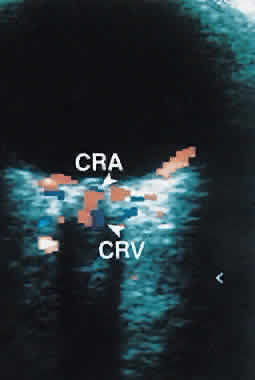
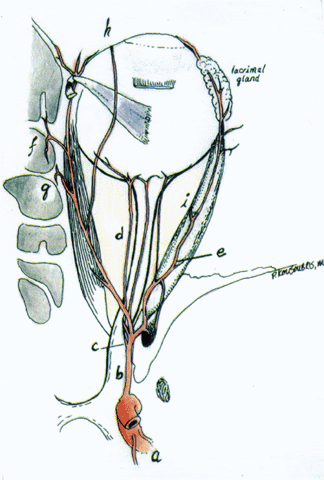
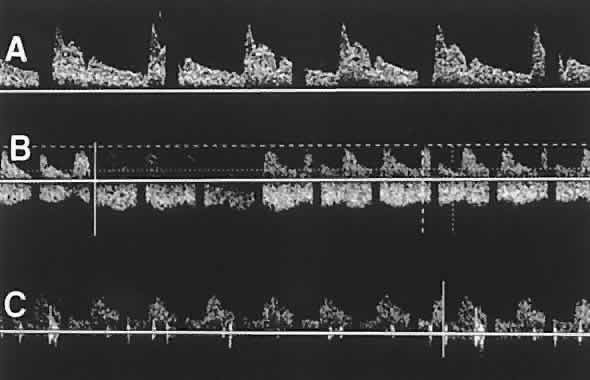
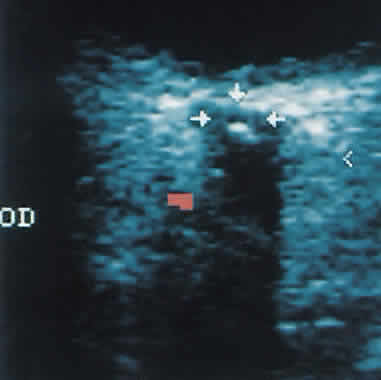
1. Eyer MK, Brandestini MA, Phillips DJ et al: Color digital echo/Doppler image presentation. Ultrasound Med Biol 7:21–31, 1981 2. Erickson SJ, Hendrix LE, Massaro BM et al: Color Doppler flow imaging of the normal and abnormal orbit. Radiology 173:511–516, 1989 3. White DN: Johann Christian Doppler and his effect: a brief history. Ultrasound Med Biol 8:583–591, 1982 4. Yoshii I, Ikeda A: A new look at the blood supply of the retro-ocular space. Anat Rec 233:321–328, 1992 5. Hayreh SS, Dass R: The ophthalmic artery. II. Intraorbital course. Br J Ophthalmol 46:165–185, 1962 6. Goebel W, Lieb WE, Ho A et al: Color Doppler imaging: A new technique to assess orbital blood flow in
patients with diabetic retinopathy. Invest Ophthalmol Vis Sci 36:864–870, 1995 7. Galassi F, Nuzzaci G, Sodi A et al: Color Doppler imaging in evaluation of optic nerve blood supply in normal
and glaucomatous subjects. Int Ophthalmol 16:273–276, 1992 8. Lieb WE, Cohen SM, Merton DA et al: Color Doppler imaging of the eye and orbit. Arch Ophthalmol 109:527–531, 1991 9. Greenfield DS, Heggerick PA, Hedges TR: Color Doppler imaging of normal orbital vasculature. Ophthalmology 102:1598–1605, 1995 10. Aburn NS, Sergott RC: Orbital colour Doppler imaging. Eye 7:639–647, 1993 11. Guthoff RF, Berger RW, Winkler P et al: Doppler ultrasonography of the ophthalmic and central retinal vessels. Arch Ophthalmol 109:532–536, 1991 12. Williamson TH, Lowe GDO, Baxter GM: Influence of age, systemic blood pressure, smoking and blood viscosity
on orbital blood velocities. Br J Ophthalmol 79:17–22, 1995 13. Berges O: Colour Doppler flow imaging of the orbital veins. Acta Ophthalmol 204:55–58, 1992 14. Lizzi FL, Coleman DJ, Driller J et al: Effects of pulsed ultrasound on ocular tissue. Ultrasound Med Biol 7:245–252, 1981 15. Lizzi FL, Packer AJ, Coleman DJ: Experimental cataract production by high frequency ultrasound. Ann Ophthalmol 10:934–942, 1978 16. Mitchell DG: Colour Doppler imaging principles, limitations, and artifacts. Radiology 177:1–10, 1990 17. Sergott RC, Flaharty PM, Lieb WE et al: Color Doppler imaging identifies four syndromes of the retrobulbar circulation
in patients with amaurosis fugax and central retinal artery occlusions. Trans Am Ophthalmol 90:383–398, 1992 18. Knapp MEP, Flaharty PM, Sergott RC et al: Gaze-induced amaurosis from central retinal artery compression. Ophthalmology 99:238–240, 1992 19. Mawn LA, Hedges TR, Rand W et al: Orbital color Doppler imaging in carotid occlusive disease. Arch Ophthalmol 115: 492–496, 1997 20. Ho AC, Lieb WE, Flaharty PM et al: Color Doppler imaging of the ocular ischemic syndrome. Ophthalmology 99:1453–1462, 1992 21. Hu H, Sheng W, Yen M et al: Color Doppler imaging of orbital arteries for detection of carotid occlusive
disease. Stroke 24:1196–1203, 1993 22. Ho AC, Sergott RC, Regillo CD et al: Color Doppler hemodynamics of giant cell arteritis. Arch Ophthalmol 112:938–945, 1994 23. Williamson TH, Baxter G, Paul R et al: Colour Doppler ultrasound in the management of a case of cranial arteritis. Br J Ophthalmol 76:690–691, 1992 24. Flaharty PM, Lieb WE, Sergott RC et al: Color Doppler imaging. A new noninvasive technique to diagnose and monitor
carotid cavernous sinus fistulas. Arch Ophthalmol 109: 522–526, 1991 25. Kotval PS, Weitzner I, Tenner MS: Diagnosis of carotid-cavernous fistula by periorbital color Doppler imaging
and pulsed Doppler volume flow analysis. J Ultrasound Med 9:101–106, 1990 26. Trible JR, Sergott RC, Spaeth GL et al: Trabeculectomy is associated with retrobulbar hemodynamic changes. Ophthalmology 101:340–351, 1994 27. Baxter GM, Williamson TH, McKillop G et al: Color Doppler ultrasound of orbital and optic nerve blood flow effects
of posture and Timolol 0.5%. Invest Ophthalmol Vis Sci 33:604–610, 1992 28. Mansberger S, Harris A, Caldemeyer K et al: Acute effect of topical apraclonidine on perimacular and orbital hemodynamics (abstract). Invest Ophthalmol Vis Sci 35(Suppl): 2176, 1994 29. Harris A, Shoemaker JA, Sergott RC et al: Vasospasm in normal tension glaucoma color Doppler imaging assessment of
carbon dioxide reactivity in orbital vessels (abstract). Invest Ophthalmol Vis Sci 35:1254, 1994 30. Flaharty PM, Phillips W, Sergott RC et al: Color Doppler imaging of superior ophthalmic vein thrombosis. Arch Ophthalmol 109:582–583, 1991 31. Lieb WE, Merton DA, Shields JA et al: Colour Doppler imaging in the demonstration of an orbital varix. Br J Ophthalmol 74:305–308, 1990 32. Jain R, Sawhney S, Berry M: Real-time sonography of orbital tumours colour Doppler characterization
initial experience. Acta Ophthalmol 204(Suppl):46–49, 1992 33. Wolff-Korman PG, Kormann BA, Hasenfratz GC et al: Duplex and color Doppler ultrasound in the differential diagnosis of choroidal
tumors. Acta Ophthalmol 204(Suppl):66–70, 1992 34. Guthoff RF, Berger RW, Winkler P et al: Doppler ultrasonography of malignant melanomas of the uvea. Arch Ophthalmol 109:537–541, 1991 35. Lieb WE, Shields JA, Cohen SM et al: Color Doppler imaging in the management of intraocular tumors. Ophthalmology 97:1660–1664, 1990 36. Mittra RA, Sergott RC, Flaharty PM et al: Optic nerve decompression improves hemodynamic parameters in papilledema. Ophthalmology 100:987–997, 1993 37. Williamson TH, Baxter GM: Central retinal vein occlusion, an investigation by color Doppler imaging. Ophthalmology 101:1362–1372, 1994 38. Regillo CD, Sergott RC, Ho AC et al: Hemodynamic alterations in the acute retinal necrosis syndrome. Ophthalmology 100:1171–1176, 1993 39. Regillo CD, Sergott RC, Brown GC: Successful scleral buckling procedures decrease central retinal artery
blood flow velocity. Ophthalmology 100:1044–1049, 1993 40. Wong D, Restori M: Ultrasonic Doppler studies of the vitreous. Eye 2:87–91, 1988 41. Wells RG, Miro P, Brummond R: Color-flow Doppler sonography of persistent hyperplastic primary vitreous. J Ultrasound Med 10:405–407, 1991 42. Canning CR, Restori M: Doppler ultrasound velocity mapping of extra-ocular muscles: A preliminary
report. Eye 3:409–414, 1989 |