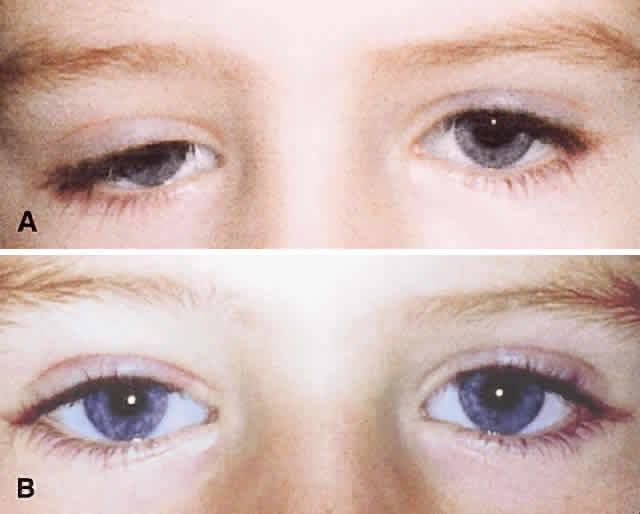
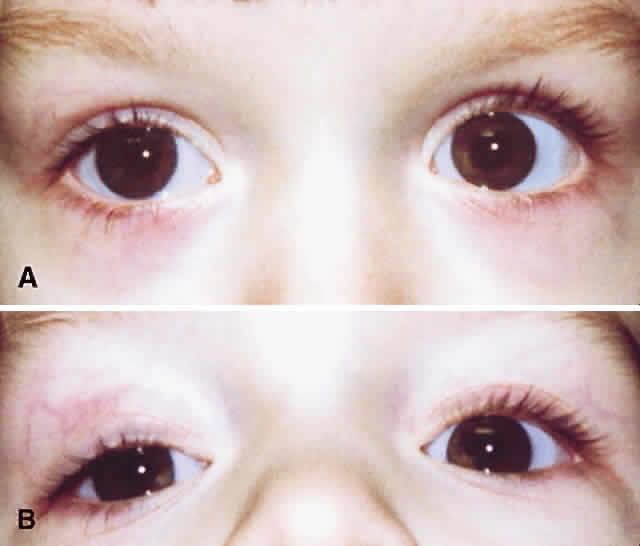
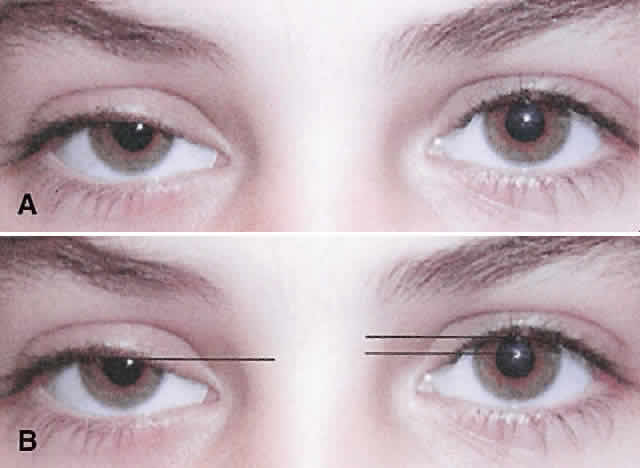
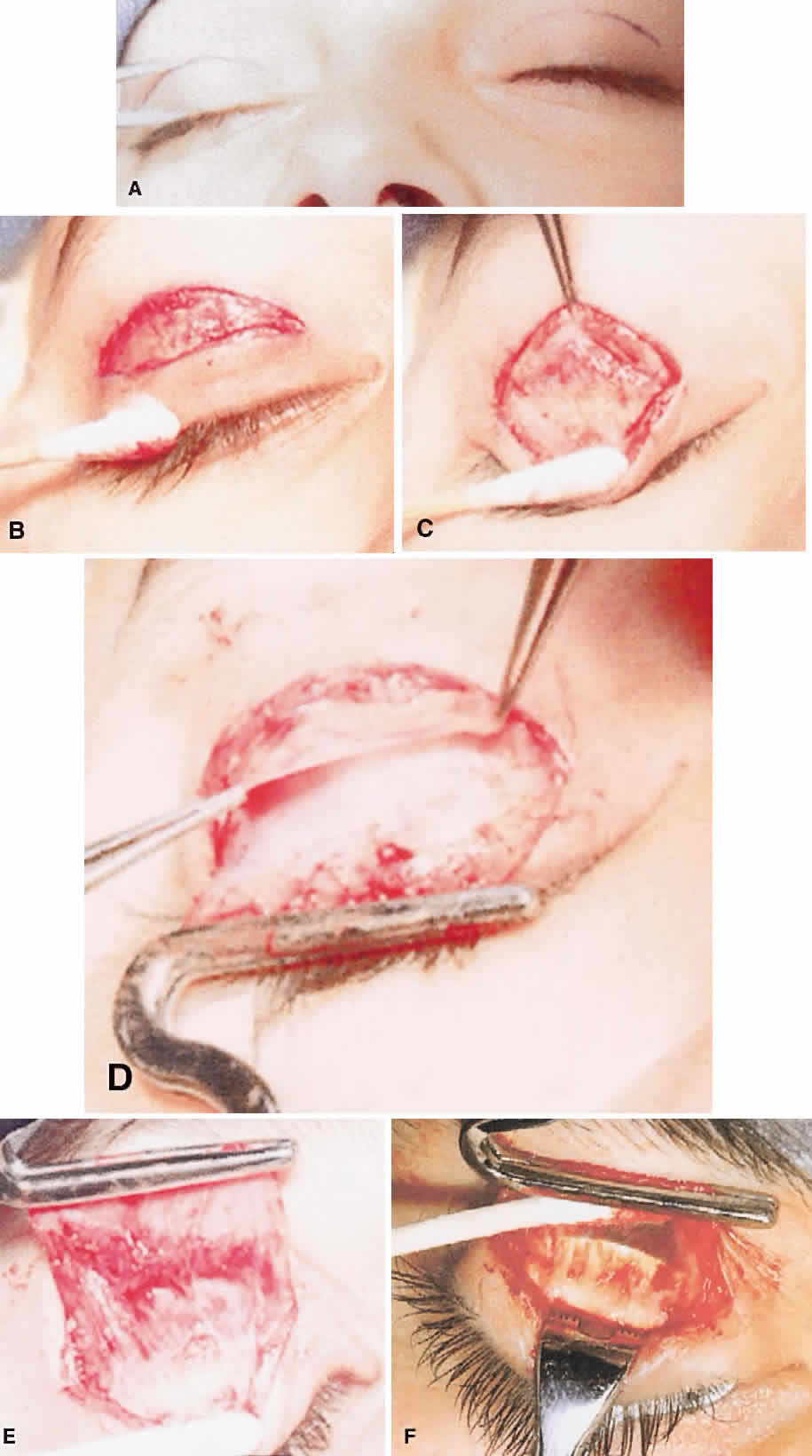
1. Edmunds B, Manners RM, Weller RO et al: Levator palpebrae superioris fibre size in normals and patients with congenital
ptosis. Eye 12:47–50, 1998 2. Merriam WW, Ellis FD, Helveston EM: Congenital blepharoptosis, anisometropia, and amblyopia. Am J Ophthalmol 89:401–497, 1980 3. Hornblass A, Kass LG, Ziffer AJ: Amblyopia in congenital ptosis. Ophthalmic Surg 26:334–337, 1995 4. Fiergang DL, Wright KW, Foster JA: Unilateral or asymmetric congenital ptosis, head posturing, and amblyopia. J Pediatr Ophthalmol Strabismus 36:74–77, 1999 5. Kao SC, Tsai CC, Lee SM et al: Astigmatic change following congenital ptosis surgery. Chung Hua I Hsueh Tsa Chih (Taipei) 61:689–693, 1998 6. Holck DE, Dutton JJ, Wehrly SR: Changes in astigmatism after ptosis surgery measured by corneal topography. Ophthalmol Plast Reconstr Surg 14:151–158, 1998 7. Langford JD, Linberg JV, Blaylock WK et al: Axial myopia in congenital ptosis: An animal model. Ophthalmol Plast Reconstr Surg 14:261–265, 1998 8. Gair EJ, Adams GG: Normal visual development after unilateral complete ptosis at birth. J AAPOS 3:58–59, 1999 9. Beard C: Ptosis. 3rd ed. St Louis: CV Mosby, 1981 10. Marmor M: Malignant hyperthermia. Surv Ophthalmol 28: 117–127, 1983 11. Dodd MJ, Phattiyakul P, Silpasuvan S: Suspected malignant hyperthermia in a strabismus patient: A case report. Arch Ophthalmol 99:1247–1250, 1981 12. Sedwick LA, Romano PE: Malignant hyperthermia. Considerations for the ophthalmologist. Surv Ophthalmol 25:378–382, 1981 13. Negre F, Caujolle JP, Ghenassia C et al: Malignant hyperthermia in ophthalmologic surgery [original in French]. J Fr Ophthalmol 11:753–757, 1988 14. Sarver BL, Putterman AM: Margin limbal distance to determine amount of levator resection. Arch Ophthalmol 103: 354–356, 1985 15. Jeong S, Lemke BN, Dortzbach RK et al: The Asian upper eyelid: An anatomical study with comparison to the caucasian
eyelid. Arch Ophthalmol 117:907–912, 1999 16. Lam DS, Ng JS, Cheng GP et al: Autogenous palmaris longus tendon as frontalis suspension material for
ptosis correction in children. Am J Ophthalmol 126:109–115, 1998 17. Chen TH, Yang JY, Chen YR: Refined frontalis fascial sling with proper lid crease formation for blepharoptosis. Plast Reconstr Surg 99:34–40, 1997 18. Wheatcroft SM, Vardy SJ, Tyers AG: Complications of fascia lata harvesting for ptosis surgery. Br J Ophthalmol 81:581–583, 1997 19. Putterman AM, Fett DR: Muller's muscle in the treatment of upper eyelid ptosis: a ten-year
study. Ophthalmic Surg 17:354–360, 1986 20. Putterman AM: A clamp for strengthening Muller's muscle in the treatment of ptosis: Modification, theory, and clamp for the Fasanella-Servat ptosis operation. Arch Ophthalmol 87:665–667, 1972 21. Engle EC, Marondel I, Houtman WA et al: Congenital fibrosis of the extraocular
muscles (autosomal dominant congenital external ophthalmoplegia): Genetic
homogeneity, linkage refinement, and physical mapping on chromosome 12. Am
J Hum Genet 57:1086–1094, 1995 (published erratum
appears in Am J Hum Genet 58:252, 1996) 22. Reck AC, Manners R, Hatchwell E: Phenotypic heterogeneity may occur in congenital fibrosis of the extraocular
muscles. Br J Ophthalmol 82:676–679, 1998 23. Nowaczyk MJ: Blepharophimosis, minor facial anomalies, genital anomalies, and mental
retardation: Report of two sibs with a unique syndrome. Am J Med Genet 87:78–81, 1999 24. Maede Y, Fujiwaki R, Watanabe Y et al: Bilateral granulosa cell tumor in a patient with blepharophimosis syndrome. Gynecol Oncol 73:335–336, 1999 25. Day-Salvatore D, McLean D: Blepharophimosis, hypoplastic radius, hypoplastic left heart, telecanthus, hydronephrosis, fused
metacarpals, and “prehensile” halluces: a
new syndrome? Am J Med Genet 80:309–313, 1998 26. Toomes C, Dixon MJ: Refinement of a translocation breakpoint associated with blepharophimosis-ptosis-epicanthus
inversus syndrome to a 280-kb interval at chromosome 3q23. Genomics 53:308—314, 1998 27. Cunniff C, Curtis M, Hassed SJ et al: Blepharophimosis: A causally heterogeneous malformation frequently associated
with developmental disabilities. Am J Med Genet 75:52–54, 1998 28. Tsaloumas MD, Willshaw HE: Congenital oculomotor palsy: associated neurological and ophthalmological
findings. Eye 11:500–503, 1997 29. Holmes JM, Mutyala S, Maus TL et al: Pediatric third, fourth, and sixth nerve palsies: A population-based study. Am J Ophthalmol 127:388–392, 1999 30. Schumacher-Feero LA, Yoo KW, Solari FM et al: Third cranial nerve palsy in children. Am J Ophthalmol 128:216–221, 1999 31. Mudgil AV, Repka MX: Ophthalmologic outcome after third cranial nerve palsy or paresis in childhood. J AAPOS 3:2–8, 1999 32. Munoz M, Page LK: Acquired double elevator palsy in a child with a pineocytoma. Am J Ophthalmol 118:810–811, 1994 33. Verma MJ, Faridi MM: Ocular motility disturbances (Duane retraction syndrome and double elevator
palsy) with congenital heart disease, a rare association with Goldenhar
syndrome: a case report. Indian J Ophthalmol 40:61–62, 1992 34. Ziffer AJ, Rosenbaum AL, Demer JL et al: Congenital double elevator palsy: Vertical saccadic velocity utilizing
the scleral search coil technique. J Pediatr Ophthalmol Strabismus 29:142–149, 1992 35. Cadera W, Bloom JN, Karlik S et al: A magnetic resonance imaging study of double elevator palsy. Can J Ophthalmol 32:250–253, 1997 36. Metz HS: Double elevator palsy. J Pediatr Ophthalmol Strabismus 18:31–35, 1981 37. Steel DH, Harrad RA: Unilateral congenital ptosis with ipsilateral superior rectus muscle overaction. Am J Ophthalmol 122:550–556, 1996 38. Callahan MA: Surgically mismanaged ptosis associated with double elevator palsy. Arch Ophthalmol 99:108–112, 1981 39. Woodruff G, Buncic JR, Morin JD: Horner's syndrome in children. J Pediatr Ophthalmol Strabismus 25:40–44, 1988 40. Segura P, Speeg-Schatz C, Wagner JM et al: Claude Bernard-Horner syndrome and its opposite, Pourfour du Petit syndrome, in
anesthesia and intensive care [in French]. Ann Fr Anesth Reanim 17:709–724, 1998 41. Brodsky MC: Hereditary external ophthalmoplegia synergistic divergence, jaw winking, and
oculocutaneous hypopigmentation: A congenital fibrosis syndrome
caused by deficient innervation to extraocular muscles. Ophthalmology 105:717–725, 1998 42. Bartkowski SB, Zapala J, Wyszynska-Pawelec G: Marcus Gunn jaw-winking phenomenon: Management and results of treatment
in 19 patients. J Craniomaxillofac Surg 27:25–29, 1999 43. Khwarg SI, Tarbet KJ, Dortzbach RK et al: Management of moderate-to-severe Marcus-Gunn jaw-winking ptosis. Ophthalmology 106:1191–1196, 1999 44. Li FY: Mapping of autosomal dominant progressive external ophthalmoplegia to a 7-cM
critical region on 10q24. Neurology 53:1265–1271, 1999 45. Tabaku M: A novel 7.4 kb mitochondrial deletion in a patient with congenital progressive
external ophthalmoplegia, muscle weakness and mental retardation. Genet Couns 10:285–293, 1999 46. De Coo IF, Gussinklo T, Arts PJ et al: A PCR test for progressive external ophthalmoplegia and Kearns-Sayre syndrome
on DNA from blood samples. J Neurol Sci 149:37–40, 1997 47. Burnstine MA: Upper blepharoplasty: A novel approach to improving progressive myopathic
blepharoptosis. Ophthalmology 106:2098–2100, 1999 48. Wallace DK, Sprunger DT, Helveston EM et al: Surgical management of strabismus associated with chronic progressive external
ophthalmoplegia. Ophthalmology 104:695–700, 1997 49. Maytal J, Spiro AJ, Sinnar S et al: The coexistence of myasthenia gravis and myotonic dystrophy in one family. Neuropediatrics 18:8–10, 1987 50. Aberfeld DC: Hereditary ptosis. Birth Defects Orig Artic Ser 7:63–65, 1971 51. Visentin DE, Yang WH, Karsh J: C1-esterase inhibitor transfusions in patients with hereditary angioedema. Ann Allergy Asthma Immunol 80:457–461, 1998 52. Daroff RB: Ocular myasthenia: Diagnosis and therapy. In Glaser J (ed): Neuro-ophthalmology, pp 62–71. St Louis: Mosby, 1980 53. Ertas M, Arac N, Kumral K et al: Ice test as a simple diagnostic aid for myasthenia gravis. Acta Neurol Scand 89:227–229, 1994 54. Evoli A, Tonali P, Bartoccioni E et al: Ocular myasthenia: Diagnostic and therapeutic problems. Acta Neurol Scand 77:31–35, 1988 55. Golnik KC, Pena R, Lee AG et al: An ice test for the diagnosis of myasthenia gravis. Ophthalmology 106:1282–1286, 1999 56. Pena RA, Golnik K, Lee A et al: An ice test for the diagnosis of myasthenia
gravis: A prospective study. Annual Meeting of the American Academy
of Ophthalmology, New Orleans, 1998 57. Saavedra J, Femminini R, Cochen S et al: Cold test for myasthenia gravis. Neurology 29:1075, 1979 58. Sethi K, Rivner M, Swift T: Ice pack test for myasthenia gravis. Neurology 37:1383–1385, 1987 59. Waller RR. Management of myogenic (myopathic) ptosis. Trans Am Acad Ophthalmol
Otolaryngol 79:697–702, 1975 |