Chapter 1:
Anatomy & Embryology of the Eye
Author:
A thorough understanding of the anatomy of the eye, orbit, visual pathways, upper cranial nerves, and central pathways for the control of eye movements is a prerequisite for proper interpretation of diseases having ocular manifestations. Furthermore, such anatomic knowledge is essential to the proper planning and safe execution of ocular and orbital surgery. Whereas most knowledge of these matters is based on anatomic dissections, either postmortem or during surgery, noninvasive techniques-particularly magnetic resonance imaging (MRI) and ultrasonography-are increasingly providing additional information. Investigating the embryology of the eye is clearly a more difficult area because of the relative scarcity of suitable human material, and thus there is still great reliance on animal studies, with the inherent difficulties in inferring parallels in human development. Nevertheless, a great deal is known about the embryology of the human eye, and-together with the recent expansion in molecular genetics-this has led to a much deeper understanding of developmental anomalies of the eye.
I. NORMAL ANATOMY
THE ORBIT (![]() Figures 1-1 and
Figures 1-1 and ![]() 1-2)
1-2)
The orbital cavity is schematically represented as a pyramid of four walls that converge posteriorly. The medial walls of the right and left orbit are parallel and are separated by the nose. In each orbit, the lateral and medial walls form an angle of 45 degrees, which results in a right angle between the two lateral walls. The orbit is compared to the shape of a pear, with the optic nerve representing its stem. The anterior circumference is somewhat smaller in diameter than the region just within the rim, which makes a sturdy protective margin.
The volume of the adult orbit is approximately 30 mL, and the eyeball occupies only about one-fifth of the space. Fat and muscle account for the bulk of the remainder.
The anterior limit of the orbital cavity is the orbital septum, which acts as a barrier between the eyelids and orbit (see below).
The orbits are related to the frontal sinus above, the maxillary sinus below, and the ethmoid and sphenoid sinuses medially. The thin orbital floor is easily damaged by direct trauma to the globe, resulting in a "blowout" fracture with herniation of orbital contents into the maxillary antrum. Infection within the sphenoid and ethmoid sinuses can erode the paper-thin medial wall (lamina papyracea) and involve the contents of the orbit. Defects in the roof (eg, neurofibromatosis) may result in visible pulsations of the globe transmitted from the brain.
Orbital Walls
The roof of the orbit is composed principally of the orbital plate of the frontal bone. The lacrimal gland is located in the lacrimal fossa in the anterior lateral aspect of the roof. Posteriorly, the lesser wing of the sphenoid bone containing the optic canal completes the roof.
The lateral wall is separated from the roof by the superior orbital fissure, which divides the lesser from the greater wing of the sphenoid bone. The anterior portion of the lateral wall is formed by the orbital surface of the zygomatic (malar) bone. This is the strongest part of the bony orbit. Suspensory ligaments, the lateral palpebral tendon, and check ligaments have connective tissue attachments to the lateral orbital tubercle.
The orbital floor is separated from the lateral wall by the inferior orbital fissure. The orbital plate of the maxilla forms the large central area of the floor and is the region where blowout fractures most frequently occur. The frontal process of the maxilla medially and the zygomatic bone laterally complete the inferior orbital rim. The orbital process of the palatine bone forms a small triangular area in the posterior floor.
The boundaries of the medial wall are less distinct. The ethmoid bone is paper-thin but thickens anteriorly as it meets the lacrimal bone. The body of the sphenoid forms the most posterior aspect of the medial wall, and the angular process of the frontal bone forms the upper part of the posterior lacrimal crest. The lower portion of the posterior lacrimal crest is made up of the lacrimal bone. The anterior lacrimal crest is easily palpated through the lid and is composed of the frontal process of the maxilla. The lacrimal groove lies between the two crests and contains the lacrimal sac.
Orbital Apex (![]() Figure 1-3)
Figure 1-3)
The apex of the orbit is the entry portal for all nerves and vessels to the eye and the site of origin of all extraocular muscles except the inferior oblique. The superior orbital fissure lies between the body and the greater and lesser wings of the sphenoid bone. The superior ophthalmic vein and the lacrimal, frontal, and trochlear nerves pass through the lateral portion of the fissure that lies outside the annulus of Zinn. The superior and inferior divisions of the oculomotor nerve and the abducens and nasociliary nerves pass through the medial portion of the fissure within the annulus of Zinn. The optic nerve and ophthalmic artery pass through the optic canal, which also lies within the annulus of Zinn. The inferior ophthalmic vein may pass through any part of the superior orbital fissure, including the portion adjacent to the body of the sphenoid that lies inferomedial to the annulus of Zinn. The inferior ophthalmic vein frequently joins the superior ophthalmic vein before exiting the orbit.
Blood Supply (![]() Figures 1-4,
Figures 1-4, ![]() 1-5 and
1-5 and ![]() 1-6)
1-6)
The principal arterial supply of the orbit and its structures derives from the ophthalmic artery, the first major branch of the intracranial portion of the internal carotid artery. This branch passes beneath the optic nerve and accompanies it through the optic canal into the orbit. The first intraorbital branch is the central retinal artery, which enters the optic nerve about 8-15 mm behind the globe. Other branches of the ophthalmic artery include the lacrimal artery, supplying the lacrimal gland and upper eyelid; muscular branches to the various muscles of the orbit; long and short posterior ciliary arteries; medial palpebral arteries to both eyelids; and the supraorbital and supratrochlear arteries. The short posterior ciliary arteries supply the choroid and parts of the optic nerve. The two long posterior ciliary arteries supply the ciliary body and anastomose with each other and with the anterior ciliary arteries to form the major arterial circle of the iris. The anterior ciliary arteries are derived from the muscular branches to the rectus muscles. They supply the anterior sclera, episclera, limbus, and conjunctiva as well as contributing to the major arterial circle of the iris. The most anterior branches of the ophthalmic artery contribute to the formation of the arterial arcades of the eyelids, which make an anastomosis with the external carotid circulation via the facial artery.
The venous drainage of the orbit is primarily through the superior and inferior ophthalmic veins, into which drain the vortex veins, the anterior ciliary veins, and the central retinal vein. The ophthalmic veins communicate with the cavernous sinus via the superior orbital fissure and the pterygoid venous plexus via the inferior orbital fissure. The superior ophthalmic vein is initially formed from the supraorbital and supratrochlear veins and from a branch of the angular vein, all of which drain the skin of the periorbital region. This provides a direct communication between the skin of the face and the cavernous sinus, thus forming the basis of the potentially lethal cavernous sinus thrombosis secondary to superficial infection of the periorbital skin.
THE EYEBALL
The normal adult globe is approximately spherical, with an anteroposterior diameter averaging 24.2 mm.
THE CONJUNCTIVA
The conjunctiva is the thin, transparent mucous membrane that covers the posterior surface of the lids (the palpebral conjunctiva) and the anterior surface of the sclera (the bulbar conjunctiva). It is continuous with the skin at the lid margin (a mucocutaneous junction) and with the corneal epithelium at the limbus.
The palpebral conjunctiva lines the posterior surface of the lids and is firmly adherent to the tarsus. At the superior and inferior margins of the tarsus, the conjunctiva is reflected posteriorly (at the superior and inferior fornices) and covers the episcleral tissue to become the bulbar conjunctiva.
The bulbar conjunctiva is loosely attached to the orbital septum in the fornices and is folded many times. This allows the eye to move and enlarges the secretory conjunctival surface. (The ducts of the lacrimal gland open into the superior temporal fornix.) Except at the limbus (where Tenon's capsule and the conjunctiva are fused for about 3 mm), the bulbar conjunctiva is loosely attached to Tenon's capsule and the underlying sclera.
A soft, movable, thickened fold of bulbar conjunctiva (the semilunar fold) is located at the inner canthus and corresponds to the nictitating membrane of some lower animals. A small, fleshy, epidermoid structure (the caruncle) is attached superficially to the inner portion of the semilunar fold and is a transition zone containing both cutaneous and mucous membrane elements.
Histology
The conjunctival epithelium consists of two to five layers of stratified columnar epithelial cells, superficial and basal. Conjunctival epithelium near the limbus, over the caruncle, and near the mucocutaneous junctions at the lid margins consists of stratified squamous epithelial cells. The superficial epithelial cells contain round or oval mucus-secreting goblet cells. The mucus, as it forms, pushes aside the goblet cell nucleus and is necessary for proper dispersion of the precorneal tear film. The basal epithelial cells stain more deeply than the superficial cells and near the limbus may contain pigment.
The conjunctival stroma is divided into an adenoid (superficial) layer and a fibrous (deep) layer. The adenoid layer contains lymphoid tissue and in some areas may contain "follicle-like" structures without germinal centers. The adenoid layer does not develop until after the first 2 or 3 months of life. This explains why inclusion conjunctivitis of the newborn is papillary in nature rather than follicular and why it later becomes follicular. The fibrous layer is composed of connective tissue that attaches to the tarsal plate. This explains the appearance of the papillary reaction in inflammations of the conjunctiva. The fibrous layer is loosely arranged over the globe.
The accessory lacrimal glands (glands of Krause and Wolfring), which resemble the lacrimal gland in structure and function, are located in the stroma. Most of the glands of Krause are in the upper fornix, the remaining few in the lower fornix. The glands of Wolfring lie at the superior margin of the upper tarsus.
Blood Supply, Lymphatics, & Nerve Supply
The conjunctival arteries are derived from the anterior ciliary and palpebral arteries. The two arteries anastomose freely and-along with the numerous conjunctival veins that generally follow the arterial pattern-form a considerable conjunctival vascular network. The conjunctival lymphatics are arranged in superficial and deep layers and join with the lymphatics of the eyelids to form a rich lymphatic plexus. The conjunctiva receives its nerve supply from the first (ophthalmic) division of the fifth nerve. It possesses a relatively small number of pain fibers.
TENON'S CAPSULE (Fascia Bulbi)
Tenon's capsule is a fibrous membrane that envelops the globe from the limbus to the optic nerve. Adjacent to the limbus, the conjunctiva, Tenon's capsule, and the episclera are fused together. More posteriorly, the inner surface of Tenon's capsule lies against the sclera, and its outer aspect is in contact with orbital fat and other structures within the extraocular muscle cone. At the point where Tenon's capsule is pierced by tendons of the extraocular muscles in their passage to their attachments to the globe, it sends a tubular reflection around each of these muscles. These fascial reflections become continuous with the fascia of the muscles, the fused fasciae sending expansions to the surrounding structures and to the orbital bones. The fascial expansions are quite tough and limit the action of the extraocular muscles and are therefore known as check ligaments. The lower segment of Tenon's capsule is thick and fuses with the fascia of the inferior rectus and the inferior oblique muscles to form the suspensory ligament of the eyeball (Lockwood's ligament), upon which the globe rests.
THE SCLERA & EPISCLERA
The sclera is the fibrous outer protective coating of the eye consisting almost entirely of collagen (![]() Figure 1-7). It is dense and white and continuous with the cornea anteriorly and the dural sheath of the optic nerve posteriorly. Across the posterior scleral foramen are bands of collagen and elastic tissue, forming the lamina cribrosa, between which pass the axon bundles of the optic nerve. The outer surface of the anterior sclera is covered by a thin layer of fine elastic tissue, the episclera, which contains numerous blood vessels that nourish the sclera. The brown pigment layer on the inner surface of the sclera is the lamina fusca, which forms the outer layer of the suprachoroidal space.
Figure 1-7). It is dense and white and continuous with the cornea anteriorly and the dural sheath of the optic nerve posteriorly. Across the posterior scleral foramen are bands of collagen and elastic tissue, forming the lamina cribrosa, between which pass the axon bundles of the optic nerve. The outer surface of the anterior sclera is covered by a thin layer of fine elastic tissue, the episclera, which contains numerous blood vessels that nourish the sclera. The brown pigment layer on the inner surface of the sclera is the lamina fusca, which forms the outer layer of the suprachoroidal space.
At the insertion of the rectus muscles, the sclera is about 0.3 mm thick; elsewhere it is about 0.6 mm thick. Around the optic nerve, the sclera is penetrated by the long and short posterior ciliary arteries and the long and short ciliary nerves (![]() Figure 1-8). The long posterior ciliary arteries and long ciliary nerves pass from the optic nerve to the ciliary body in a shallow groove on the inner surface of the sclera at the 3 and 9 o'clock meridians. Slightly posterior to the equator, the four vortex veins draining the choroid exit through the sclera, usually one in each quadrant. About 4 mm posterior to the limbus, slightly anterior to the insertion of the respective rectus muscle, the four anterior ciliary arteries and veins penetrate the sclera. The nerve supply to the sclera is from the ciliary nerves.
Figure 1-8). The long posterior ciliary arteries and long ciliary nerves pass from the optic nerve to the ciliary body in a shallow groove on the inner surface of the sclera at the 3 and 9 o'clock meridians. Slightly posterior to the equator, the four vortex veins draining the choroid exit through the sclera, usually one in each quadrant. About 4 mm posterior to the limbus, slightly anterior to the insertion of the respective rectus muscle, the four anterior ciliary arteries and veins penetrate the sclera. The nerve supply to the sclera is from the ciliary nerves.
Histologically, the sclera consists of many dense bands of parallel and interlacing collagen bundles, each of which is 10-16 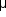 m thick and 100-140
m thick and 100-140 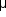 m wide. The histologic structure of the sclera is remarkably similar to that of the cornea. The reason for the transparency of the cornea and the opacity of the sclera is the relative deturgescence of the cornea.
m wide. The histologic structure of the sclera is remarkably similar to that of the cornea. The reason for the transparency of the cornea and the opacity of the sclera is the relative deturgescence of the cornea.
THE CORNEA
The cornea is a transparent tissue comparable in size and structure to the crystal of a small wristwatch (Figure 1-9). It is inserted into the sclera at the limbus, the circumferential depression at this junction being known as the scleral sulcus. The average adult cornea is 0.52 mm thick in the center, about 0.65 mm thick at the periphery, and about 11.75 mm in diameter horizontally and 10.6 mm vertically. From anterior to posterior, it has five distinct layers (Figure 1-10): the epithelium (which is continuous with the epithelium of the bulbar conjunctiva), Bowman's layer, the stroma, Descemet's membrane, and the endothelium. The epithelium has five or six layers of cells. Bowman's layer is a clear acellular layer, a modified portion of the stroma. The corneal stroma accounts for about 90% of the corneal thickness. It is composed of intertwining lamellae of collagen fibrils 10-250 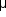 m in width and 1-2
m in width and 1-2 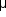 m in height that run almost the full diameter of the cornea. They run parallel to the surface of the cornea and by virtue of their size and proximity are optically clear. The lamellae lie within a ground substance of hydrated proteoglycans in association with the keratocytes that produce the collagen and ground substance. Descemet's membrane, constituting the basal lamina of the corneal endothelium, has a homogeneous appearance on light microscopy but a laminated appearance on electron microscopy due to structural differences between its pre- and postnatal portions. It is about 3
m in height that run almost the full diameter of the cornea. They run parallel to the surface of the cornea and by virtue of their size and proximity are optically clear. The lamellae lie within a ground substance of hydrated proteoglycans in association with the keratocytes that produce the collagen and ground substance. Descemet's membrane, constituting the basal lamina of the corneal endothelium, has a homogeneous appearance on light microscopy but a laminated appearance on electron microscopy due to structural differences between its pre- and postnatal portions. It is about 3 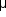 m thick at birth but increases in thickness throughout life, reaching 10-12
m thick at birth but increases in thickness throughout life, reaching 10-12 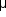 m in adulthood. The endothelium has only one layer of cells, but this is responsible for maintaining the essential deturgescence of the corneal stroma. The endothelium is quite susceptible to injury as well as undergoing loss of cells with age. Endothelial repair is limited to enlargement and sliding of existing cells, with little capacity for cell division. Failure of endothelial function leads to corneal edema.
m in adulthood. The endothelium has only one layer of cells, but this is responsible for maintaining the essential deturgescence of the corneal stroma. The endothelium is quite susceptible to injury as well as undergoing loss of cells with age. Endothelial repair is limited to enlargement and sliding of existing cells, with little capacity for cell division. Failure of endothelial function leads to corneal edema.
Sources of nutrition for the cornea are the vessels of the limbus, the aqueous, and the tears. The superficial cornea also gets most of its oxygen from the atmosphere. The sensory nerves of the cornea are supplied by the first (ophthalmic) division of the fifth (trigeminal) cranial nerve.
The transparency of the cornea is due to its uniform structure, avascularity, and deturgescence.
THE UVEAL TRACT
The uveal tract is composed of the iris, the ciliary body, and the choroid (![]() Figure 1-7). It is the middle vascular layer of the eye and is protected by the cornea and sclera. It contributes blood supply to the retina.
Figure 1-7). It is the middle vascular layer of the eye and is protected by the cornea and sclera. It contributes blood supply to the retina.
Iris
The iris is the anterior extension of the ciliary body. It presents as a flat surface with a centrally situated round aperture, the pupil. The iris lies in contiguity with the anterior surface of the lens, dividing the anterior chamber from the posterior chamber, each of which contains aqueous humor. Within the stroma of the iris are the sphincter and dilator muscles. The two heavily pigmented layers on the posterior surface of the iris represent anterior extensions of the neuroretina and retinal pigment epithelium.
The blood supply to the iris is from the major circle of the iris (![]() Figure 1-4). Iris capillaries have a nonfenestrated endothelium and hence do not normally leak intravenously injected fluorescein. Sensory nerve supply to the iris is via fibers in the ciliary nerves.
Figure 1-4). Iris capillaries have a nonfenestrated endothelium and hence do not normally leak intravenously injected fluorescein. Sensory nerve supply to the iris is via fibers in the ciliary nerves.
The iris controls the amount of light entering the eye. Pupillary size is principally determined by a balance between constriction due to parasympathetic activity transmitted via the third cranial nerve and dilation due to sympathetic activity. (See Chapter 14.)
The Ciliary Body
The ciliary body, roughly triangular in cross-section, extends forward from the anterior end of the choroid to the root of the iris (about 6 mm). It consists of a corrugated anterior zone, the pars plicata (2 mm), and a flattened posterior zone, the pars plana (4 mm). The ciliary processes arise from the pars plicata (Figure 1-11). They are composed mainly of capillaries and veins that drain through the vortex veins. The capillaries are large and fenestrated and hence leak intravenously injected fluorescein. There are two layers of ciliary epithelium: an internal nonpigmented layer, representing the anterior extension of the neuroretina; and an external pigmented layer, representing an extension of the retinal pigment epithelium. The ciliary processes and their covering ciliary epithelium are responsible for the formation of aqueous.
The ciliary muscle is composed of a combination of longitudinal, circular, and radial fibers. The function of the circular fibers is to contract and relax the zonular fibers, which originate in the valleys between the ciliary processes (![]() Figure 1-12). This alters the tension on the capsule of the lens, giving the lens a variable focus for both near and distant objects in the visual field. The longitudinal fibers of the ciliary muscle insert into the trabecular meshwork to influence its pore size.
Figure 1-12). This alters the tension on the capsule of the lens, giving the lens a variable focus for both near and distant objects in the visual field. The longitudinal fibers of the ciliary muscle insert into the trabecular meshwork to influence its pore size.
The blood vessels supplying the ciliary body are derived from the major circle of the iris. The sensory nerve supply of the iris is via the ciliary nerves.
The Choroid
The choroid is the posterior segment of the uveal tract, between the retina and the sclera. It is composed of three layers of choroidal blood vessels: large, medium, and small. The deeper the vessels are placed in the choroid, the wider their lumens (Figure 1-13). The internal portion of the choroid vessels is known as the choriocapillaris. Blood from the choroidal vessels drains via the four vortex veins, one in each of the four posterior quadrants. The choroid is bounded internally by Bruch's membrane and externally by the sclera. The suprachoroidal space lies between the choroid and the sclera. The choroid is firmly attached posteriorly to the margins of the optic nerve. Anteriorly, the choroid joins with the ciliary body.
The aggregate of choroidal blood vessels serves to nourish the outer portion of the underlying retina (![]() Figure 1-4).
Figure 1-4).
THE LENS
The lens is a biconvex, avascular, colorless and almost completely transparent structure, about 4 mm thick and 9 mm in diameter. It is suspended behind the iris by the zonule, which connects it with the ciliary body. Anterior to the lens is the aqueous; posterior to it, the vitreous. The lens capsule (see below) is a semipermeable membrane (slightly more permeable than a capillary wall) that will admit water and electrolytes.
A subcapsular epithelium is present anteriorly (Figure 1-14). The lens nucleus is harder than the cortex. With age, subepithelial lamellar fibers are continuously produced, so that the lens gradually becomes larger and less elastic throughout life. The nucleus and cortex are made up of long concentric lamellae. The suture lines formed by the end-to-end joining of these lamellar fibers are Y-shaped when viewed with the slitlamp (Figure 1-15). The Y is upright anteriorly and inverted posteriorly.
Each lamellar fiber contains a flattened nucleus. These nuclei are evident microscopically in the peripheral portion of the lens near the equator and are continuous with the subcapsular epithelium.
The lens is held in place by a suspensory ligament known as the zonule (zonule of Zinn), which is composed of numerous fibrils that arise from the surface of the ciliary body and insert into the lens equator.
The lens consists of about 65% water, about 35% protein (the highest protein content of any tissue of the body), and a trace of minerals common to other body tissues. Potassium is more concentrated in the lens than in most tissues. Ascorbic acid and glutathione are present in both oxidized and reduced forms.
There are no pain fibers, blood vessels, or nerves in the lens.
THE AQUEOUS
Aqueous humor is produced by the ciliary body. Entering the posterior chamber, it passes through the pupil into the anterior chamber (![]() Figure 1-7) and then peripherally toward the anterior chamber angle. The physiology of the aqueous is discussed in Chapter 11.
Figure 1-7) and then peripherally toward the anterior chamber angle. The physiology of the aqueous is discussed in Chapter 11.
THE ANTERIOR CHAMBER ANGLE
The anterior chamber angle lies at the junction of the peripheral cornea and the root of the iris (![]() Figures 1-12, 1-16, and 11-3). Its main anatomic features are Schwalbe's line, the trabecular meshwork (which overlies Schlemm's canal), and the scleral spur.
Figures 1-12, 1-16, and 11-3). Its main anatomic features are Schwalbe's line, the trabecular meshwork (which overlies Schlemm's canal), and the scleral spur.
Schwalbe's line marks the termination of the corneal endothelium. The trabecular meshwork is triangular in cross-section, with its base directed toward the ciliary body. It is composed of perforated sheets of collagen and elastic tissue, forming a filter with decreasing pore size as the canal of Schlemm is approached. The internal portion of the meshwork, facing the anterior chamber, is known as the uveal meshwork; the external portion, adjacent to the canal of Schlemm, is called the corneoscleral meshwork. The longitudinal fibers of the ciliary muscle insert into the trabecular meshwork. The scleral spur is an inward extension of the sclera between the ciliary body and Schlemm's canal, to which the iris and ciliary body are attached. Efferent channels from Schlemm's canal (about 30 collector channels and about 12 aqueous veins) communicate with the episcleral venous system.
THE RETINA
The retina is a thin, semitransparent, multilayered sheet of neural tissue that lines the inner aspect of the posterior two-thirds of the wall of the globe. It extends almost as far anteriorly as the ciliary body, ending at that point in a ragged edge, the ora serrata (![]() Figure 1-12). In adults the ora serrata is about 6.5 mm behind Schwalbe's line on the temporal side and 5.7 mm behind it nasally. The outer surface of the sensory retina is apposed to the retinal pigment epithelium and thus related to Bruch's membrane, the choroid, and the sclera. In most areas, the retina and retinal pigment epithelium are easily separated to form the subretinal space, such as occurs in retinal detachment. But at the optic disk and the ora serrata, the retina and retinal pigment epithelium are firmly bound together, thus limiting the spread of subretinal fluid in retinal detachment. This contrasts with the potential suprachoroidal space between the choroid and sclera, which extends to the scleral spur. Choroidal detachments thus extend beyond the ora serrata, under the pars plana and pars plicata. The epithelial layers of the inner surface of the ciliary body and the posterior surface of the iris represent anterior extensions of the retina and retinal pigment epithelium. The inner surface of the retina is apposed to the vitreous.
Figure 1-12). In adults the ora serrata is about 6.5 mm behind Schwalbe's line on the temporal side and 5.7 mm behind it nasally. The outer surface of the sensory retina is apposed to the retinal pigment epithelium and thus related to Bruch's membrane, the choroid, and the sclera. In most areas, the retina and retinal pigment epithelium are easily separated to form the subretinal space, such as occurs in retinal detachment. But at the optic disk and the ora serrata, the retina and retinal pigment epithelium are firmly bound together, thus limiting the spread of subretinal fluid in retinal detachment. This contrasts with the potential suprachoroidal space between the choroid and sclera, which extends to the scleral spur. Choroidal detachments thus extend beyond the ora serrata, under the pars plana and pars plicata. The epithelial layers of the inner surface of the ciliary body and the posterior surface of the iris represent anterior extensions of the retina and retinal pigment epithelium. The inner surface of the retina is apposed to the vitreous.
The layers of the retina, starting from its inner aspect, are as follows: (1) internal limiting membrane; (2) nerve fiber layer, containing the ganglion cell axons passing to the optic nerve; (3) ganglion cell layer; (4) inner plexiform layer, containing the connections of the ganglion cells with the amacrine and bipolar cells; (5) inner nuclear layer of bipolar, amacrine, and horizontal cell bodies; (6) outer plexiform layer, containing the connections of the bipolar and horizontal cells with the photoreceptors; (7) outer nuclear layer of photoreceptor cell nuclei; (8) external limiting membrane; (9) photoreceptor layer of rod and cone inner and outer segments; and (10) retinal pigment epithelium (Figure 1-17). The inner layer of Bruch's membrane is actually the basement membrane of the retinal pigment epithelium.
The retina is 0.1 mm thick at the ora serrata and 0.56 mm thick at the posterior pole. In the center of the posterior retina is the macula. This can be defined clinically as the area of yellowish pigmentation resulting from the presence of luteal pigment (xanthophyll), which is 3 mm in diameter. An alternative histologic definition is that part of the retina in which the ganglion cell layer is more than one cell thick. Clinically, this corresponds to the area bounded by the temporal retinal vascular arcades. In the center of the macula, 4 mm lateral to the optic disk, is the fovea, clinically obvious as a depression that creates a particular reflection when viewed ophthalmoscopically. It corresponds to the retinal avascular zone of fluorescein angiography. Histologically, the fovea is characterized by thinning of the outer nuclear layer and absence of the other parenchymal layers as a result of the oblique course of the photoreceptor cell axons (Henle fiber layer) and the centrifugal displacement of the retinal layers that are closer to the inner retinal surface. The foveola is the most central portion of the fovea, in which the photoreceptors are all cones, and the thinnest part of the retina (0.25 mm). All these histologic features provide for fine visual discrimination. The normally empty extracellular space of the retina is potentially greatest at the macula, and diseases that lead to accumulation of extracellular material cause considerable thickening of this area.
The retina receives its blood supply from two sources: the choriocapillaris immediately outside Bruch's membrane, which supplies the outer third of the retina, including the outer plexiform and outer nuclear layers, the photoreceptors, and the retinal pigment epithelium; and branches of the central retinal artery, which supply the inner two-thirds (![]() Figure 1-4). The fovea is supplied entirely by the choriocapillaris and is susceptible to irreparable damage when the retina is detached. The retinal blood vessels have a nonfenestrated endothelium, which forms the inner blood-retinal barrier. The endothelium of choroidal vessels is fenestrated. The outer blood-retinal barrier lies at the level of the retinal pigment epithelium.
Figure 1-4). The fovea is supplied entirely by the choriocapillaris and is susceptible to irreparable damage when the retina is detached. The retinal blood vessels have a nonfenestrated endothelium, which forms the inner blood-retinal barrier. The endothelium of choroidal vessels is fenestrated. The outer blood-retinal barrier lies at the level of the retinal pigment epithelium.
THE VITREOUS
The vitreous is a clear, avascular, gelatinous body that comprises two-thirds of the volume and weight of the eye. It fills the space bounded by the lens, retina, and optic disk (![]() Figure 1-7). The outer surface of the vitreous-the hyaloid membrane-is normally in contact with the following structures: the posterior lens capsule, the zonular fibers, the pars plana epithelium, the retina, and the optic nerve head. The base of the vitreous maintains a firm attachment throughout life to the pars plana epithelium and the retina immediately behind the ora serrata. The attachment to the lens capsule and the optic nerve head is firm in early life but soon disappears.
Figure 1-7). The outer surface of the vitreous-the hyaloid membrane-is normally in contact with the following structures: the posterior lens capsule, the zonular fibers, the pars plana epithelium, the retina, and the optic nerve head. The base of the vitreous maintains a firm attachment throughout life to the pars plana epithelium and the retina immediately behind the ora serrata. The attachment to the lens capsule and the optic nerve head is firm in early life but soon disappears.
The vitreous is about 99% water. The remain- ing 1% includes two components, collagen and hyaluronic acid, which give the vitreous a gel-like form and consistency because of their ability to bind large volumes of water.
THE EXTERNAL ANATOMIC LANDMARKS
Accurate localization of the position of internal structures with reference to the external surface of the globe is important in many surgical procedures. The distance of structures from the limbus as measured externally is less than their actual length. Externally, the ora serrata is situated approximately 5.5 mm from the limbus on the medial side and 7 mm on the temporal side of the globe. This corresponds to the level of insertion of the rectus muscles. Injections into the vitreous cavity through the pars plana should be given 4-5 mm from the limbus in the phakic eye. In the aphakic eye, it is possible to inject 0.5-1 mm more anteriorly. The pars plicata, which is the target for cyclodestructive procedures in the treatment of intractable glaucoma, occupies the 2-3 mm directly posterior to the limbus.
THE EXTRAOCULAR MUSCLES
Six extraocular muscles control the movement of each eye: four rectus and two oblique muscles.
Rectus Muscles
The four rectus muscles originate at a common ring tendon (annulus of Zinn) surrounding the optic nerve at the posterior apex of the orbit (![]() Figure 1-3). They are named according to their insertion into the sclera on the medial, lateral, inferior, and superior surfaces of the eye. The principal action of the respective muscles is thus to adduct, abduct, depress, and elevate the globe (see Chapter 12). The muscles are about 40 mm long, becoming tendinous 4-9 mm from the point of insertion, where they are about 10 mm wide. The approximate distances of the points of insertion from the corneal limbus are as follows: medial rectus, 5.5 mm: inferior rectus, 6.75 mm; lateral rectus, 7 mm; and superior rectus, 7.5 mm (Figure 1-18). With the eye in the primary position, the vertical rectus muscles make an angle of about 23 degrees with the optic axis.
Figure 1-3). They are named according to their insertion into the sclera on the medial, lateral, inferior, and superior surfaces of the eye. The principal action of the respective muscles is thus to adduct, abduct, depress, and elevate the globe (see Chapter 12). The muscles are about 40 mm long, becoming tendinous 4-9 mm from the point of insertion, where they are about 10 mm wide. The approximate distances of the points of insertion from the corneal limbus are as follows: medial rectus, 5.5 mm: inferior rectus, 6.75 mm; lateral rectus, 7 mm; and superior rectus, 7.5 mm (Figure 1-18). With the eye in the primary position, the vertical rectus muscles make an angle of about 23 degrees with the optic axis.
Oblique Muscles
The two oblique muscles control primarily torsional movement and, to a lesser extent, upward and downward movement of the globe (see Chapter 12).
The superior oblique is the longest and thinnest of the ocular muscles. It originates above and medial to the optic foramen and partially overlaps the origin of the levator palpebrae superioris muscle. The superior oblique has a thin, fusiform belly (40 mm long) and passes anteriorly in the form of a tendon to its trochlea, or pulley. It is then reflected backward and downward to attach in a fan shape to the sclera beneath the superior rectus. The trochlea is a cartilaginous structure attached to the frontal bone 3 mm behind the orbital rim. The superior oblique tendon is enclosed in a synovial sheath as it passes through the trochlea.
The inferior oblique muscle originates from the nasal side of the orbital wall just behind the inferior orbital rim and lateral to the nasolacrimal duct. It passes beneath the inferior rectus and then under the lateral rectus muscle to insert onto the sclera with a short tendon. The insertion is into the posterotemporal segment of the globe and just over the macular area. The muscle is 37 mm long.
In the primary position, the muscle plane of the superior and inferior oblique muscles forms an angle of 51-54 degrees with the optic axis.
Fascia
All the extraocular muscles are ensheathed by fascia. Near the points of insertion of these muscles the fascia is continuous with Tenon's capsule, and fascial condensations to adjacent orbital structures serve as check ligaments (Figures 1-19 and 1-20).
Nerve Supply
The oculomotor nerve (III) innervates the medial, inferior, and superior rectus muscles and the inferior oblique muscle. The abducens nerve (VI) innervates the lateral rectus muscle; the trochlear nerve (IV) innervates the superior oblique muscle.
Blood Supply
The blood supply to the extraocular muscles is derived from the muscular branches of the ophthalmic artery. The lateral rectus and inferior oblique muscles are also supplied by branches from the lacrimal artery and the infraorbital artery, respectively.
THE OCULAR ADNEXA
1. EYEBROWS
The eyebrows are folds of thickened skin covered with hair. The skin fold is supported by underlying muscle fibers. The glabella is the hairless prominence between the eyebrows.
2. EYELIDS
The upper and lower eyelids (palpebrae) are modified folds of skin that can close to protect the anterior eyeball (Figure 1-21). Blinking helps spread the tear film, which protects the cornea and conjunctiva from dehydration. The upper lid ends at the eyebrows; the lower lid merges into the cheek.
The eyelids consist of five principal planes of tissues. From superficial to deep, they are the skin layer, a layer of striated muscle (orbicularis oculi), areolar tissue, fibrous tissue (tarsal plates), and a layer of mucous membrane (palpebral conjunctiva) (![]() Figure 1-22).
Figure 1-22).
Structures of the Eyelids
A. Skin Layer:
The skin of the eyelids differs from skin on most other areas of the body in that it is thin, loose, and elastic and possesses few hair follicles and no subcutaneous fat.
B. Orbicularis Oculi Muscle:
The function of the orbicularis oculi muscle is to close the lids. Its muscle fibers surround the palpebral fissure in concentric fashion and spread for a short distance around the orbital margin. Some fibers run onto the cheek and the forehead. The portion of the muscle that is in the lids is known as its pretarsal portion; the portion over the orbital septum is the preseptal portion. The segment outside the lid is called the orbital portion. The orbicularis oculi is supplied by the facial nerve.
C. Areolar Tissue:
The submuscular areolar tissue that lies deep to the orbicularis oculi muscle communicates with the subaponeurotic layer of the scalp.
D. Tarsal Plates:
The main supporting structure of the eyelids is a dense fibrous tissue layer that-along with a small amount of elastic tissue-is called the tarsal plate. The lateral and medial angles and extensions of the tarsal plates are attached to the orbital margin by the lateral and medial palpebral ligaments. The upper and lower tarsal plates are also attached by a condensed, thin fascia to the upper and lower orbital margins. This thin fascia forms the orbital septum.
E. Palpebral Conjunctiva:
The lids are lined posteriorly by a layer of mucous membrane, the palpebral conjunctiva, which adheres firmly to the tarsal plates. A surgical incision through the gray line of the lid margin (see below) splits the lid into an anterior lamella of skin and orbicularis muscle and a posterior lamella of tarsal plate and palpebral conjunctiva.
Lid Margins
The free lid margin is 25-30 mm long and about 2 mm wide. It is divided by the gray line (mucocutaneous junction) into anterior and posterior margins.
A. Anterior Margin:
1. Eyelashes-
The eyelashes project from the margins of the eyelids and are arranged irregularly. The upper lashes are longer and more numerous than the lower lashes and turn upward; the lower lashes turn downward.
2. Glands of Zeis-
These are small modified sebaceous glands that open into the hair follicles at the base of the eyelashes.
3. Glands of Moll-
These are modified sweat glands that open in a row near the base of the eyelashes.
B. Posterior Margin:
The posterior lid margin is in close contact with the globe, and along this margin are the small orifices of modified sebaceous glands (meibomian, or tarsal, glands).
C. Lacrimal Punctum:
At the medial end of the posterior margin of the lid, a small elevation with a central small opening can be seen on the upper and lower lids. The puncta serve to carry the tears down through the corresponding canaliculus to the lacrimal sac.
Palpebral Fissure
The palpebral fissure is the elliptic space between the two open lids. The fissure terminates at the medial and lateral canthi. The lateral canthus is about 0.5 cm from the lateral orbital rim and forms an acute angle. The medial canthus is more elliptic than the lateral canthus and surrounds the lacrimal lake (Figure 1-21).
Two structures are identified in the lacrimal lake: the lacrimal caruncle, a yellowish elevation of modified skin containing large modified sweat glands and sebaceous glands that open into follicles which contain fine hair (Figure 1-9); and the plica semilunaris, a vestigial remnant of the third eyelid of lower animal species.
In Orientals, a skin fold known as epicanthus passes from the medial termination of the upper lid to the medial termination of the lower lid, hiding the caruncle. Epicanthus may be present normally in young infants of all races and disappears with development of the nasal bridge but persists throughout life in Orientals.
Orbital Septum
The orbital septum is the fascia behind that portion of the orbicularis muscle that lies between the orbital rim and the tarsus and serves as a barrier between the lid and the orbit.
The orbital septum is pierced by the lacrimal vessels and nerves, the supratrochlear artery and nerve, the supraorbital vessels and nerves, the infratrochlear nerve (Figure 1-23), the anastomosis between the angular and ophthalmic veins, and the levator palpebrae superioris muscle.
The superior orbital septum blends with the tendon of the levator palpebrae superioris and the superior tarsus; the inferior orbital septum blends with the inferior tarsus.
Lid Retractors
The lid retractors are responsible for opening the eyelids. They are formed by a musculofascial complex, with both striated and smooth muscle components, known as the levator complex in the upper lid and the capsulopalpebral fascia in the lower lid.
In the upper lid, the striated muscle portion is the levator palpebrae superioris, which arises from the apex of the orbit and passes forward to divide into an aponeurosis and a deeper portion that contains the smooth muscle fibers of Müller's (superior tarsal) muscle (![]() Figure 1-22). The aponeurosis elevates the anterior lamella of the lid, inserting into the posterior surface of the orbicularis oculi and through this into the overlying skin to form the upper lid skin crease. Müller's muscle inserts into the upper border of the tarsal plate and the superior fornix of the conjunctiva, thus elevating the posterior lamella.
Figure 1-22). The aponeurosis elevates the anterior lamella of the lid, inserting into the posterior surface of the orbicularis oculi and through this into the overlying skin to form the upper lid skin crease. Müller's muscle inserts into the upper border of the tarsal plate and the superior fornix of the conjunctiva, thus elevating the posterior lamella.
In the lower lid, the main retractor is the inferior rectus muscle, from which fibrous tissue extends to enclose the inferior oblique muscle and insert into the lower border of the tarsal plate and the orbicularis oculi. Associated with this aponeurosis are the smooth muscle fibers of the inferior tarsal muscle.
The smooth muscle components of the lid retractors are innervated by sympathetic nerves. The levator and inferior rectus muscles are supplied by the third cranial (oculomotor) nerve. Ptosis is thus a feature of both Horner's syndrome and third nerve palsy.
Levator Palpebrae Superioris Muscle
The levator palpebrae muscle arises with a short tendon from the undersurface of the lesser wing of the sphenoid above and ahead of the optic foramen. The tendon blends with the underlying origin of the superior rectus muscle. The levator belly passes forward, forms an aponeurosis, and spreads like a fan. The muscle, including its smooth muscle component (Müller's muscle), and its aponeurosis form an important part of the upper lid retractor (see above). The palpebral segment of the orbicularis oculi muscle acts as its antagonist.
The two extremities of the levator aponeurosis are called its medial and lateral horns. The medial horn is thin and is attached below the frontolacrimal suture and into the medial palpebral ligament. The lateral horn passes between the orbital and palpebral portions of the lacrimal gland and inserts into the orbital tubercle and the lateral palpebral ligament.
The sheath of the levator palpebrae superioris is attached to the superior rectus muscle inferiorly. The superior surface, at the junction of the muscle belly and the aponeurosis, forms a thickened band that is attached medially to the trochlea and laterally to the lateral orbital wall, the band forming the check ligaments of the muscle. The band is also known as Whitnall's ligament.
The levator is supplied by the superior branch of the oculomotor nerve (III). Blood supply to the levator palpebrae superioris is derived from the lateral muscular branch of the ophthalmic artery.
Sensory Nerve Supply
The sensory nerve supply to the eyelids is derived from the first and second divisions of the trigeminal nerve (V). The small lacrimal, supraorbital, supratrochlear, infratrochlear, and external nasal nerves are branches of the ophthalmic division of the fifth nerve. The infraorbital, zygomaticofacial, and zygomaticotemporal nerves are branches of the maxillary (second) division of the trigeminal nerve.
Blood Supply & Lymphatics
The blood supply to the lids is derived from the lacrimal and ophthalmic arteries by their lateral and medial palpebral branches. Anastomoses between the lateral and medial palpebral arteries form the tarsal arcades that lie in the submuscular areolar tissue.
Venous drainage from the lids empties into the ophthalmic vein and the veins that drain the forehead and temple. The veins are arranged in pre- and posttarsal plexuses (![]() Figure 1-6).
Figure 1-6).
Lymphatics from the lateral segment of the lids run into the preauricular and parotid nodes. Lymphatics draining the medial side of the lids empty into the submandibular lymph nodes.
3. THE LACRIMAL APPARATUS
The lacrimal complex consists of the lacrimal gland, the accessory lacrimal glands, the canaliculi, the lacrimal sac, and the nasolacrimal duct (![]() Figure 1-24).
Figure 1-24).
The lacrimal gland consists of the following structures:
The almond-shaped orbital portion, located in the lacrimal fossa in the anterior upper temporal segment of the orbit, is separated from the palpebral portion by the lateral horn of the levator palpebrae muscle. To reach this portion of the gland surgically, one must incise the skin, the orbicularis oculi muscle, and the orbital septum.
The smaller palpebral portion is located just above the temporal segment of the superior conjunctival fornix. Lacrimal secretory ducts, which open by approximately ten fine orifices, connect the orbital and palpebral portions of the lacrimal gland to the superior conjunctival fornix. Removal of the palpebral portion of the gland cuts off all of the connecting ducts and thus prevents secretion by the entire gland.
The accessory lacrimal glands (glands of Krause and Wolfring) are located in the substantia propria of the palpebral conjunctiva.
Tears drain from the lacrimal lake via the upper and lower puncta and canaliculi to the lacrimal sac, which lies in the lacrimal fossa. The nasolacrimal duct continues downward from the sac and opens into the inferior meatus of the nasal cavity, lateral to the inferior turbinate. Tears are directed into the puncta by capillary attraction and gravity and by the blinking action of the eyelids. The combined forces of capillary attraction in the canaliculi, gravity, and the pumping action of Horner's muscle, which is an extension of the orbicularis oculi muscle to a point behind the lacrimal sac, all tend to continue the flow of tears down the nasolacrimal duct into the nose.
Blood Supply & Lymphatics
The blood supply of the lacrimal gland is derived from the lacrimal artery. The vein that drains the gland joins the ophthalmic vein. The lymphatic drainage joins with the conjunctival lymphatics to drain into the preauricular lymph nodes.
Nerve Supply
The nerve supply to the lacrimal gland is by (1) the lacrimal nerve (sensory), a branch of the trigeminal first division; (2) the great superficial petrosal nerve (secretory), which comes from the superior salivary nucleus; and (3) sympathetic nerves accompanying the lacrimal artery and the lacrimal nerve.
Related Structures
The medial palpebral ligament connects the upper and lower tarsal plates to the frontal process at the inner canthus anterior to the lacrimal sac. The portion of the lacrimal sac below the ligament is covered by a few fibers of the orbicularis oculi muscle. These fibers offer little resistance to swelling and distention of the lacrimal sac. The area below the medial palpebral ligament becomes swollen in acute dacryocystitis, and fistulas commonly open in the area.
The angular vein and artery lie just deep to the skin, 8 mm to the nasal side of the inner canthus. Skin incisions made in surgical procedures on the lacrimal sac should always be placed 2-3 mm to the nasal side of the inner canthus to avoid these vessels.
THE OPTIC NERVE
The trunk of the optic nerve consists of about 1 million axons that arise from the ganglion cells of the retina (nerve fiber layer). The optic nerve emerges from the posterior surface of the globe through the posterior scleral foramen, a short, circular opening in the sclera about 1 mm below and 3 mm nasal to the posterior pole of the eye (![]() Figure 1-8). The nerve fibers become myelinated on leaving the eye, increasing the diameter from 1.5 mm (within the sclera) to 3 mm (within the orbit). The orbital segment of the nerve is 25-30 mm long; it travels within the optic muscle cone, via the bony optic canal, and thus gains access to the cranial cavity. The intracanalicular portion measures 4-9 mm. After a 10 mm intracranial course, the nerve joins the opposite optic nerve to form the optic chiasm.
Figure 1-8). The nerve fibers become myelinated on leaving the eye, increasing the diameter from 1.5 mm (within the sclera) to 3 mm (within the orbit). The orbital segment of the nerve is 25-30 mm long; it travels within the optic muscle cone, via the bony optic canal, and thus gains access to the cranial cavity. The intracanalicular portion measures 4-9 mm. After a 10 mm intracranial course, the nerve joins the opposite optic nerve to form the optic chiasm.
Eighty percent of the optic nerve consists of visual fibers that synapse in the lateral geniculate body on neurons whose axons terminate in the primary visual cortex of the occipital lobes. Twenty percent of the fibers are pupillary and bypass the geniculate body en route to the pretectal area. Since the ganglion cells of the retina and their axons are part of the central nervous system, they will not regenerate if severed.
Sheaths of the Optic Nerve (Figure 1-25)
The fibrous wrappings that ensheathe the optic nerve are continuous with the meninges. The pia mater is loosely attached about the nerve near the chiasm and only for a short distance within the cranium, but it is closely attached around most of the intracanalicular and all of the intraorbital portions. The pia consists of some fibrous tissue with numerous small blood vessels (![]() Figure 1-26). It divides the nerve fibers into bundles by sending numerous septa into the nerve substance. The pia continues to the sclera, with a few fibers running into the choroid and lamina cribrosa.
Figure 1-26). It divides the nerve fibers into bundles by sending numerous septa into the nerve substance. The pia continues to the sclera, with a few fibers running into the choroid and lamina cribrosa.
The arachnoid comes in contact with the optic nerve at the intracranial end of the optic canal and accompanies the nerve to the globe, where it ends in the sclera and overlying dura. This sheath is a diaphanous connective tissue membrane with many septate connections with the pia mater, which it closely resembles. It is more intimately associated with pia than with dura.
The dura mater lining the inner surface of the cranial vault comes in contact with the optic nerve as it leaves the optic canal. As the nerve enters the orbit from the optic canal, the dura splits, one layer (the periorbita) lining the orbital cavity and the other forming the outer dural covering of the optic nerve. The dura becomes continuous with the outer two-thirds of the sclera. The dura consists of tough, fibrous, relatively avascular tissue lined by endothelium on the inner surface.
The subdural space is between the dura and the arachnoid; the subarachnoid space is between the pia and the arachnoid. Both are more potential than actual spaces under normal conditions but are direct continuations of their corresponding intracranial spaces. Subarachnoid or subdural fluid under sufficient pressure will fill these potential spaces about the optic nerve. The meningeal layers are adherent to each other and to the optic nerve and the surrounding bone within the optic foramen, making the optic nerve resistant to traction from either end.
Blood Supply (![]() Figure 1-26)
Figure 1-26)
The surface layer of the optic disk receives blood from branches of the retinal arterioles. The rest of the nerve in front of the lamina cribrosa is supplied by branches from the peripapillary choroidal vessels. In the region of the lamina cribrosa, the arterial supply is from the short posterior ciliary arteries. The retrolaminar optic nerve receives some blood from branches of the central retinal artery. The remainder of the intraorbital nerve, as well as the intracanalicular and intracranial portions, are supplied by a pial network of vessels derived from the various branches of the ophthalmic artery and other branches of the internal carotids.
THE OPTIC CHIASM
The optic chiasm is variably situated near the top of the diaphragm of the sella turcica, most often posteriorly, projecting 1 cm above it and at a 45-degree angle upward from the optic nerves as they emerge from the optic canals (![]() Figure 1-27). The lamina terminalis forms the anterior wall of the third ventricle. The internal carotid arteries lie just laterally, adjacent to the cavernous sinuses. The chiasm is made up of the junction of the two optic nerves and provides for crossing of the nasal fibers to the opposite optic tract and passage of temporal fibers to the ipsilateral optic tract. The macular fibers are arranged similarly to the rest of the fibers except that their decussation is farther posteriorly and superiorly. The chiasm receives many small blood vessels from the neighboring circle of Willis.
Figure 1-27). The lamina terminalis forms the anterior wall of the third ventricle. The internal carotid arteries lie just laterally, adjacent to the cavernous sinuses. The chiasm is made up of the junction of the two optic nerves and provides for crossing of the nasal fibers to the opposite optic tract and passage of temporal fibers to the ipsilateral optic tract. The macular fibers are arranged similarly to the rest of the fibers except that their decussation is farther posteriorly and superiorly. The chiasm receives many small blood vessels from the neighboring circle of Willis.
THE RETROCHIASMATIC VISUAL PATHWAYS
Each optic tract begins at the posterolateral angle of the chiasm and sweeps around the upper part of the cerebral peduncle to end in the lateral geniculate nucleus. Afferent pupillary fibers leave the tract just anterior to the nucleus and pass via the brachium of the superior colliculus to the midbrain. (The pupillary pathway is diagrammed in ![]() Figure 14-2.) Afferent visual fibers terminate on cells in the lateral geniculate nucleus that give rise to the geniculocalcarine tract. This tract traverses the posterior limb of the internal capsule and then fans out into a broad bundle called the optic radiation. The fibers in this bundle curve backward around the anterior aspect of the temporal horn of the lateral ventricle and then medially to reach the calcarine cortex of the occipital lobe, where they terminate. The most inferior fibers, which carry projections from the superior aspect of the contralateral half of the visual field, course anteriorly into the temporal lobe in a configuration known as Meyer's loop. Lesions of the temporal lobe that extend 5 cm back from the anterior tip involve these fibers and can produce superior quandrantanopic field defects.
Figure 14-2.) Afferent visual fibers terminate on cells in the lateral geniculate nucleus that give rise to the geniculocalcarine tract. This tract traverses the posterior limb of the internal capsule and then fans out into a broad bundle called the optic radiation. The fibers in this bundle curve backward around the anterior aspect of the temporal horn of the lateral ventricle and then medially to reach the calcarine cortex of the occipital lobe, where they terminate. The most inferior fibers, which carry projections from the superior aspect of the contralateral half of the visual field, course anteriorly into the temporal lobe in a configuration known as Meyer's loop. Lesions of the temporal lobe that extend 5 cm back from the anterior tip involve these fibers and can produce superior quandrantanopic field defects.
The primary visual cortex (area V1) occupies the upper and lower lips and the depths of the calcarine fissure on the medial aspect of the occipital lobe. Each lobe receives input from the two ipsilateral half-retinas, representing the contralateral half of the binocular visual field. Projection of the visual field onto the visual cortex occurs in a precise and orderly retinotopic pattern. The macula is represented at the medial posterior pole, and the peripheral parts of the retina project to the most anterior part of the calcarine cortex. On either side of area V1 lies area V2, and then area V3. V2 appears to function in a manner very similar to V1. Area V4, situated on the medial surface of the cerebral hemisphere but more anterior and inferior than V1 in the region of the fusiform gyrus, seems to be primarily concerned with color processing. Motion detection localizes to an area at the junction of the occipital and temporal lobes, lateral to area V1 and known as area V5 or MT.
THE OCULOMOTOR NERVE (III)
The oculomotor nerve leaves the brainstem between the cerebral peduncles and passes near the posterior communicating artery of the circle of Willis. Lateral to the pituitary gland, it is closely approximated to the optic tract, and here it pierces the dura to course in the lateral wall of the cavernous sinus. As the nerve leaves the cavernous sinus, it divides into superior and inferior divisions. The superior division enters the orbit within the annulus of Zinn at its highest point and adjacent to the trochlear nerve (![]() Figure 1-3). The inferior division enters the annulus of Zinn low and passes below the optic nerve to supply the medial and inferior rectus muscles. A large branch from the inferior division extends forward to supply the inferior oblique. A small twig from the proximal end of the nerve to the inferior oblique carries parasympathetic fibers to the ciliary ganglion.
Figure 1-3). The inferior division enters the annulus of Zinn low and passes below the optic nerve to supply the medial and inferior rectus muscles. A large branch from the inferior division extends forward to supply the inferior oblique. A small twig from the proximal end of the nerve to the inferior oblique carries parasympathetic fibers to the ciliary ganglion.
THE TROCHLEAR NERVE (IV)
Although the thinnest of the cranial nerves, the trochlear nerve (![]() Figure 1-3) has the longest intracranial course, and it is also the only nerve to originate on the dorsal surface of the brain stem. The fibers decussate before they emerge from the brainstem just below the inferior colliculi, where they are subject to injury from the tentorium. The nerve pierces the dura behind the sella turcica and travels within the lateral walls of the cavernous sinus to enter the superior orbital fissure medial to the frontal nerve. From this point it travels within the periorbita of the roof over the levator muscle to the upper surface of the superior oblique muscle.
Figure 1-3) has the longest intracranial course, and it is also the only nerve to originate on the dorsal surface of the brain stem. The fibers decussate before they emerge from the brainstem just below the inferior colliculi, where they are subject to injury from the tentorium. The nerve pierces the dura behind the sella turcica and travels within the lateral walls of the cavernous sinus to enter the superior orbital fissure medial to the frontal nerve. From this point it travels within the periorbita of the roof over the levator muscle to the upper surface of the superior oblique muscle.
THE TRIGEMINAL NERVE (V) (![]() Figure 1-3)
Figure 1-3)
The trigeminal nerve originates from the pons, and its sensory roots form the trigeminal ganglion. The first (ophthalmic) of the three divisions passes through the lateral wall of the cavernous sinus and divides into the lacrimal, frontal, and nasociliary nerve. The lacrimal nerve passes through the upper lateral aspect of the superior orbital fissure, outside the annulus of Zinn, and continuing its lateral course in the orbit to terminate in the lacrimal gland, providing its sensory innervation. Slightly medial to the lacrimal nerve within the superior orbital fissure is the frontal nerve, which is the largest of the first division of branches of the trigeminal nerve. It also crosses over the annulus of Zinn and follows a course over the levator to the medial aspect of the orbit, where it divides into the supraorbital and supratrochlear nerves. These provide sensation to the brow and forehead. The nasociliary nerve is the sensory nerve of the eye. After entering through the medial portion of the annulus of Zinn, it lies between the superior rectus and the optic nerve. Branches to the ciliary ganglion and those forming the ciliary nerves provide sensory supply to the cornea, iris, and ciliary body. The terminal branches are the infratrochlear nerve, which supplies the medial portion of the conjunctiva and eyelids, and the anterior ethmoidal nerve, which provides sensation to the tip of the nose. Thus, the skin on the tip of the nose may be affected with vesicular lesions prior to the onset of herpes zoster ophthalmicus.
The second (maxillary) division of the trigeminal nerve passes through the foramen rotundum and enters the orbit through the inferior orbital fissure. It passes through the infraorbital canal, becoming the infraorbital nerve, and exits via the infraorbital foramen, supplying sensation to the lower lid and adjacent cheek. It is frequently damaged in fractures of the orbital floor.
THE ABDUCENS NERVE (VI)
The abducens nerve (![]() Figure 1-3) originates between the pons and medulla and pursues an extended course up the clivus to the posterior clinoid, penetrates the dura, and passes within the cavernous sinus. (All other nerves course through the lateral wall of the cavernous sinus.) After passing through the superior orbital fissure within the annulus of Zinn, the nerve continues laterally to innervate the lateral rectus muscle.
Figure 1-3) originates between the pons and medulla and pursues an extended course up the clivus to the posterior clinoid, penetrates the dura, and passes within the cavernous sinus. (All other nerves course through the lateral wall of the cavernous sinus.) After passing through the superior orbital fissure within the annulus of Zinn, the nerve continues laterally to innervate the lateral rectus muscle.
NEXT
Page: 1 | 2 | 3 | 4
10.1036/1535-8860.ch1