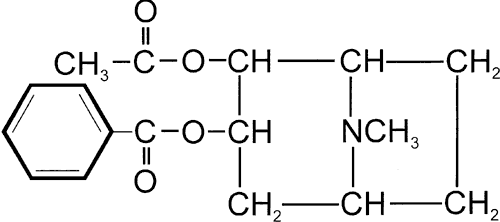
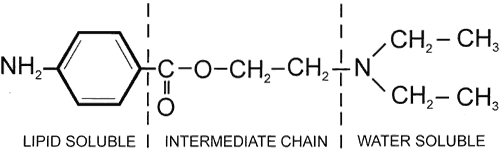
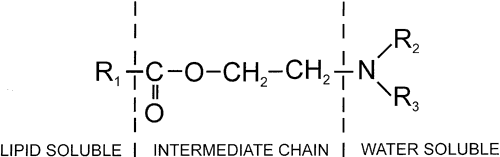
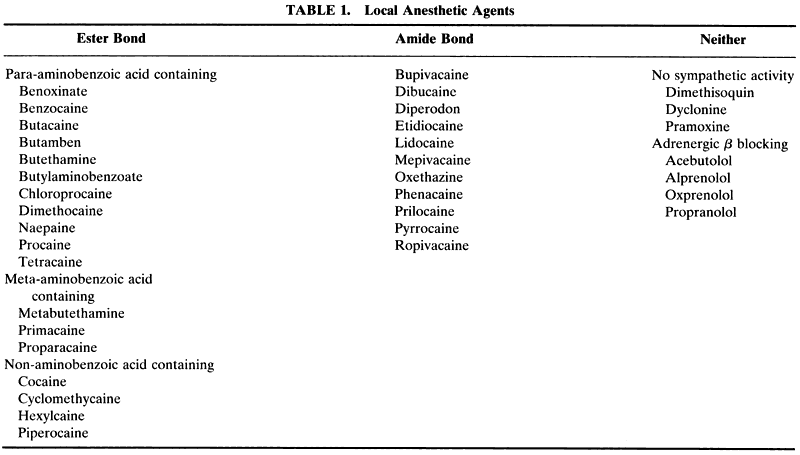
1. de Jong RH: Local anesthetics: from cocaine to xylocaine. In: Local anesthetics. St. Louis, Mosby-Year Book, 1994:4–5. 2. Ogden DC, Siegelbaum SA, Colquhoun D: Blockade of acetylcholine-activated ion channels, by an uncharged local
anesthetic. Nature 289:596–598, 1981. 3. Staiman A, Seeman P: Conduction-blocking concentrations of anesthetics increase with nerve axon
diameter: studies with alcohol, lidocaine and tetrodotoxin on single
myelinated fibers. J Pharm Exp Therap 201:340–349, 1977. 4. Dubbels R, Schloot W: Studies on the metabolism of benoxinate by human pseudocholinesterase. Metabol Pediatr Syst Ophthalmol 7:37–43, 1983. 5. Nelson SD, Garland WA, Breck GD et al: Quantification of lidocaine and several metabolites utilizing chemical-ionization
mass spectrometry and stable isotope labeling. J Pharmaceut Sci 66:1180–1189, 1977. 6. Zoglio MA, Soine TO, Rippie EG: Esters of bicyclic aminoalcohols. V: Duration of corneal anesthesia versus
enzymatic hydrolytic rate of benzoates of 1-2- and 3- methyl -2- hydroxyquinolizidines. J Pharmaceut Sci 60:411–416, 1971. 7. Winnie AP, LaVallee DA, Sosa BP et al: Clinical pharmokinetics of local anaesthetics. Canad Anaesth Soc J 4:252–262, 1977. 8. Backer CL, Tinker JA, Robertson DM et al: Myocardial reinfarction following local anesthesia for ophthalmic surgery. Anesth Analg 59:257–262, 1980. 9. Carruthers JDA, Sanmugasunderan S, Mills K et al: The efficacy of topical corneal anesthesia with 0.5% bupivacaine eyedrops. Can J Ophthalmol 30:264–266, 1995. 10. McDevitt RG: The assessment of beta-adrenoceptor blocking drugs I man. Br J Clin Pharmacol 4:413–425, 1977. 11. Polse KA, Keener RJ, Jauregui MJ: Dose-response effects of corneal anesthetics. Am J Optom Physiol Optics 55:8–14, 1978. 12. Matsumoto S, Hayashi K, Tsuchisaka H et al: Pharmacokinetics of surface
anesthesia in the human cornea Jpn J Ophthalmol 25:235–340, 1981. 13. Weiss JS, Goren MB: The effect of corneal hypesthesia on the duration of proparacaine anesthetic
eyedrops. Am J Ophthalmol 112:326–330, 1991. 14. Gotsis SS, Volonaki OM, Theodossiadis GP: Percutaneous anaesthesia with a lignocaine-prilocaine cream (Emla) for
eyelid skin surgery. Br J Ophthalmol 78:209–210, 1994. 15. Baudouin C, Gastaud P: Influence of topical anesthesia on tonometric values of intraocular pressure. Ophthalmologica 208:309–313, 1994. 16. Carel RS, Korczyn AD, Boyman R: Amethocaine and intraocular pressure. Ophthalmic Res 11:212–215, 1979. 17. Bloom JN, Levene RZ, Thomas G et al: Fluorophotometry and the rate of aqueous flow in man. Arch Ophthalmol 94:435–443, 1976. 18. Clinch TE, Benedetto DA, Felberg NT et al: Schirmer's test—a closer look. Arch Ophthalmol 101:1383–1386, 1983. 19. Shuler JD: Topical anesthesia in a patient with a history of retrobulbar hemorrhage. Arch Ophthalmol 111:733–737, 1993. 20. Crandall AS, Zabriskie NA, Patel BCK et al: A comparison of patient comfort during cataract surgery with topical anesthesia
versus topical anesthesia and intracameral litodaine. Ophthalmolgy 106:60–66, 1999. 21. Boulton JE, Lopatatzidis A, Luck J et al: A randomized controlled trial of intracameral lidocaine during phacoemulsification
under topical anesthesia. Ophthalmology 107:68–71, 2000. 22. Chittenden HB, Meacock WR, Govan JAS: Topical anaesthesia with oxybuprocaine versus sub-Tenon's infiltration
with 2% lignocaine for small incision cataract surgery. Br J Ophthalmol 81:288–290, 1997. 23. Patel BCK, Burns TA, Crandall A et al: A comparison of topical and retrobulbar anesthesia for cataract surgery. Ophthalmology 103:1196–1203, 1996. 24. Duguid IGM, Claque CMP, Thamby-Rajah Y et al: Topical anesthesia for phacoemulsification surgery. Eye 9:456–459, 1995. 25. Jacobi PC, Dietlein TS, Jacobi FK: A comparative study of topical v retrobulbar anesthesia in complicated
cataract surgery. Arch Ophthalmol 118:1037–1043, 2000. 26. Katz J, Feldman MA, Bass EB et al: Injectable versus topical anesthesia
for cataract surgery Ophthalmology 107:2054–2060, 2000. 27. Watson DM: Topical amethocaine in strabismus surgery. Anaesthesia 46:368–370, 1991. 28. Verma S, Corbett MC, Marshall J: A prospective randomized, double-masked trial to evaluate the role of topical
anesthetics in controlling pain after photorefractive keratectomy. Ophthalmology 102:1918–1924, 1995. 29. Zavon MR, Fichte CM: Trigeminal neuralgia relieved by ophthalmic anesthetic. JAMA 265:2807, 1991. 30. Spaziante R, Cappabianca P, Saini M et al: Treatment of trigeminal neuralgia by ophthalmic anesthetic. J Neurosurg 77:159–160, 1992. 31. Rowbotham MC, Davies PS, Fields HL: Topical lidocaine gel relieves postherpetic neuralgia. Ann Neurol 37:246–253, 1995. 32. Ramselaar JAM, Boot JP, van Haerington NJ et al: Corneal epithelial permeability after instillation of ophthalmic solutions
containing local anaesthetics and preservatives. Cur Eye Res 7:947–953, 1988. 33. Herse P, Siu A: Short-term effects of proparacaine on human corneal thickness. Acta Ophthalmol 70:740–744, 1992. 34. Stolwijk TR, van Best JA, Boot JP et al: Corneal epithelial barrier function after oxybuprocaine provocation in
diabetics. Invest Ophthalmol Vis Sci 31:436–439, 1990. 35. Marr MG, Wood R: Effects of topical anesthetics on regeneration of corneal epithelium. Am J Ophthalmol 43:606–614, 1957. 36. Risco JM, Miller LC: Ultrastructural alterations in the endothelium in a patient with topical
anesthesic abuse keratopathy. Ophthalmology 99:628–633, 1992. 37. Rosenwasser GOD, Holland S, Pflugfelder SC et al: Topical anesthetic abuse. Ophthalmology 97:967–972, 1990. 38. Henkes HE, Waubke TN: Keratitis from abuse of corneal anaesthetics. Br J Ophthalmol 62:62–65, 1978. 39. Chern KC, MeislerDM, Wilhelmus KR et al: Corneal anesthetic abuse and Candida keratitis. Ophthalmology 103:37–40, 1996. 40. Badenoch PR, Coste DJ: Antimicrobial activity of topical anesthetic preparations. Br J Ophthalmol 66:364–367, 1982. 41. Kleinfeld J, Ellis PP: Effects of topical anesthetics on growth of micro-organisms. Arch Ophthalmol 76:712–715, 1966. 42. Duffner LR, Pfugfelder SC, Mandelbaum S et al: Potential bacterial contamination in fluorescein-anesthetic solutions. Am J Ophthalmol 110:199–202, 1990. 43. Van Lint A: IL faut toujours paralyser les paupieres dans l'operation de la cataracte. Bull Soc Belge Ophthalmol 43:23–28, 1921. 44. Khurana AK, Sachdeva RK, Gombar KK: Evaluation of subconjunctival anaesthesia vs peribulbar anaesthesia in
cataract surgery. Acta Ophthalmologica 72:727–730, 1994. 45. Fujishima H, Yagi Y, Yang H-Y: Direct sub-Tenon's ocular anesthesia for strabismus surgery. Ophthalmologica 209:208–211, 1995. 46. Fry RA, Henderson J: Local anaesthesia for eye surgery. The peri-ocular technique. Anaesthseia 45:14–17, 1989. 47. Ropo A, Nikki P, Ruusuvaara P et al: Comparison of retrobulbar and periocular injections of lignocaine by computerised
tomography. Br J Ophthalmol 75:417–420, 1991. 48. Carroll FD, deRoetth Jr A: The effect of retrobulbar injections of procaine on the optic nerve. Trans Amer Acad Ophthalmol Oto 59:356–363, 1955. 49. Ropo A, Ruusuvaara P, Setala K: Visual evoked potentials after retrobulbar or periocular anaesthesia. Br J Ophthalmol. 76:541–544, 1992. 50. Talks SJ, Chong NHV, Gibson JM et al: Visual acuity and pupillary reactions after peribulbar anesthesia. Br J Ophthalmol 78:41–43, 1994. 51. Sanchez - Capuchino A, Meadows D, Morgan L et al: Local anaesthesia for eye surgery without a facial nerve block. Anaesthesia 48:428–431, 1993. 52. Ripart J, Metge L, Prat-Pradal D et al: Medial canthus single-injection episcleral (sub-Tenon anesthesia): computedtomography
imaging. Anesth Analg 87:42–45, 1998. 53. Hosten N, Bornfeld N, Lemke AJ et al: MR of the eye with retrobulbar anesthesia. Am J Neuroradiol 18:1788–1790, 1997. 54. Anders N, Heuermann T, Ruther K et al: Clinical and electrophysiological results after intracameral lidocaine 1% anesthesia. Ophthalmology 106:1863–1868, 1999. 55. Iradier MT, Fernandez C, Bohorquez P et al: Intraocular lidocaine in phacoemulsification. An endothelium and blood-aqueous
barrier permeability study. Ophthalmology 107:896–901, 2000. 56. Gillow T, Scotcher SM, Deutsch J et al: Efficacy of supplementary intracameral lidocaine in routine phacoemulsification
under topical anesthesia. Ophthalmology 106:2173–2177, 1999. 57. Malecaze FA, Deneuville SF, Julia BJ et al: Pain relief with intracameral mepiracaine during phacoemulsification. Br J Ophthalmol 84:171–174, 2000. 58. United States Pharmacopeia Convention Inc:
Most frequently reported drugs and their prevalent problems.
Quality Review
55:2,
1996. 59. Gillart T, Bazin JE, Montetagaud M et al: The effects of volume and speed of injection in peribulbar anaesthesia. Anaesthesia. 53:486–510, 1998. 60. Nakano NI: Temperature-dependent aqueous solubilities of lidocaine, mepivacaine and
bupivacaine. J Pharmaceut Sci 68:667–668, 1979. 61. Ursell PG, Spalton DJ: The effect of solution temperature on the pain of peribulbar anaesthesia. Ophthalmology 103:839–841, 1996. 62. Bell RWD, Butt ZA: Warming lidocaine reduces the pain of injection during peribulbar local
anaesthesia for cataract surgery. Br J Ophthalmol 79:1015–1017, 1995. 63. Krause M, Weindler J, Ruprecht KW: Does warming of anesthetic solutions improve analgesia and akinesia in
retrobulbar anesthesia? Ophthalmology 104:429–432, 1997. 64. Mindel JS: Value of hyaluronidase in ocular surgical akinesia. Am J Ophthalmol 85:643–646, 1978. 65. Kallio H, Paloheimo M, Maunuksela EL: Hyaluronidase as an adjuvant inbupivacaine-lidocaine mixture for retrobulbar/peribulbar
block. Anesth Analg 91:934–937, 2000. 66. Sarvela J, Nikki P: Hyaluronidase improves regional ophthalmic anaesthesia
with etidocaine, Can J Anaesth 39:920–924, 1992. 67. Crawford M, Kerr WJ: The effects of hyaluronidase on peribulbar block. Anaesthesia 49:907–908, 1994. 68. Rowley SA, Hale JE, Finlay RD: Sub-Tenon's local anaesthesia: the effect of hyaluronidase. Br J Ophthalmol 84:435–436, 2000. 69. Nicoll JMV, Treuren B, Acharya PA et al: Retrobulbar anesthesia: the role of hyaluronidase. Anesth Analg 65:1324–1328, 1986. 70. Minning Jr CA: Hyaluronidase allergy simulating expulsive choroidal hemorrhage. Arch Ophthalmol 112:585, 1994. 71. Robinson J, Fernando R, Sun Wai WYS et al: Clinical stability of bupivacaine, lidocaine and epinephrine in pH-adjusted
solutions. Anaesthesia 55:853–858, 2000. 72. Lewis P, Hamilton RC, Loken RG: Comparison of plain with pH-adjusted bupivacaine with hyaluronidase for
peribulbar block. Can J Anaesth 39:555–558, 1992. 73. Zahl K, Jordan A, McGroarty J et al: Peribulbar anesthesia effect of bicarbonate on mixtures of lidocaine, bupivacaine
and hyaluronidase with or without epinephrine. Ophthalmology 98:239–242, 1991. 74. Roberts JE, MacLeod BA, Hollands RH: Improved peribulbar anaesthesia with alkalinization and hyaluronidase. Can J Anaesth 40:835–838, 1993. 75. Eccarius SG, Gordon ME, Parelman JJ: Bicarbonate-buffered lidocaine-epinephrine-hyaluronidase for eyelid anesthesia. Ophthalmology 97:1499–1501, 1990. 76. McKay W, Morris R, Mushlin P: Sodium bicarbonate attenuates pain on skin infiltration with lidocaine; with
or without epinephrine. Anesth Analg 66:572–574, 1987. 77. Minasian MC, Ionides AC, Fernando R et al: Pain perception with pH buffered peribulbar anaesthesia: a pilot study. Br J Ophthalmol 84:1041–1044, 2000. 78. Krohn J, Houding G, Seland JH et al: Retrobulbar anesthesia with an without adrenaline in extracapsular cataract
surgery. Acta Ophthalmol Scand 73:56–60, 1995. 79. Bjornstrom L, Hansen A, Otland N et al: Peribulbar anaesthesia—a
clinical evaluation of two different anaesthetic mixtures Acta Ophthalmologica 72:712–714, 1994. 80. Chin GN, Almquist HT: Bupivacaine and lidocaine retrobulbar anesthesia—a double-blind clinical
study. Ophthalmology 90:369–372, 1983. 81. Donlon JV, Moss J: Plasma catecholamine levels during local anesthesia for cataract operations. Anesthesiology 51:471–473, 1979. 82. Dimsdale JE, Moss J: Plasma catecholamines in stress and exercise. JAMA 243:340–342, 1980; Correction 243:2485, 1980. 83. Michels RG, Maumenee AE: Retrobulbar alcohol injection in seeing eyes. Trans Am Acad Ophthalmol Otolaryngol 77:164–167, 1973. 84. Cordes FC: Optic atrophy following alcohol injections. Am J Ophthalmol 12:120, 1929. 85. Payer FL Occlusion of the central retinal artery following retrobulbar
injection of alcohol. Oftalmologica 8:65–69, 1964. 86. Mathur SP: Ciliary block with benzocaine. Am J Ophthalmol 47:867–869, 1959. 87. Fiore D, Lupidi G, Santoni G: Retrobulbar injection of chlorpromazine in absolute glaucoma. J Fr Ophthalmol 3:397–399, 1980. 88. Waltz KL, Williams GA, Goldey SH et al: Alternative to retrobulbar ethanol for blind painful eyes. Ophthalmology 99(suppl): 153, 1992. 89. Foster AH, Carlson BM: Myotonicity of local anesthetics and regeneration of the damaged muscle
fibers. Anesth Analg 58:727–736, 1980. 90. Johnson ME, Jones GH: Effects of Marcaine, a myotoxic drug, on macromolecular synthesis in mice. Biochem Pharmacol 27:1753–1757, 1978. 91. Steer JH, Mastaglia FL: Protein degradation in bupivacaine-treated muscles: The role of extracellular
calcium.J Neurolog Sci 75:343–351, 1986. 92. Porter JD, Edney DP, McMahon EJ et al: Extraocular myotoxicity of the retrobulbar anesthetic bupivacaine hydrochloride. Invest OphthalmolVis Sci 29:163–174, 1988. 93. Okland S, Komorowski TE, Carlson BM: Ultrastructure of Mepicavaine-induced damage and regeneration in rat extraocular
muscle. Invest Ophthalmol Vis Sci 30:1643–1651, 1989. 94. Mather C, Hayden Smith J, Bloom PA: The efficacy of 0.75% bupivacaine with pH adjustment and hyaluronidase
for peribulbar blockade: the incidence of prolonged ptosis. Eur J Ophthalmol 4:13–18, 1994. 95. Rainin EA, Carlson BM: Postoperative diplopia and ptosis. A clinical hypothesis based on the myotoxicity
of local anesthetics. Arch Ophthalmol 103:1337–1339, 1985. 96. Hamed LM, Mancuso A: Inferior rectus muscle contracture syndrome after retrobulbar anesthesia. Ophthalmology 98:1506–1512, 1991. 97. Hunter D: Author's reply. Ophthalmolgy 103:546, 1996. 98. Mazdit J, Boico O, Samii K: Myocardial uptake of bupivacaine: II. Pharmacokinetics and pharmacodynamics
of bupivacaine enantiomers in the isolated perfused rabbit heart. Anesth Analg 77:477–482, 1993. 99. Mc Lure HA, Rubin AP: Comparison of 0.75% levobupivacaine with 0.75% racemic bupivacaine for
peribulbar anaesthesia. Anaesthesia 53:1160–1164, 1998. 100. Mc Lure HA, Rubin AP, Wescott M et al: A comparison of 1% ropivacaine with a mixture of 0.75% bupivacaine and 2% lignocaine
for peribulbar anaesthesia. Anaesthesia 54:1178–1182, 1999. 101. Nicholson G, Sutton B, Hall GM: Comparison of 1% ropivacaine with 0.75% bupivacaine and 2% lidocaine for
peribulbar anaesthesia. Br J Anaesth 84:89–91, 2000. 102. Huha T, Ala-Kokko TI, Salomaki T et al: Clinical efficacy and pharmacokinetics of 1% ropivacaine and 0.75% bupivacaine
in peribulbar anaesthesia for cataract surgery. Anaesthesia 54:137–141, 1999. 103. Atkinson WS: Local anesthesia in ophthalmology. Trans Amer Ophthalmol Soc 2:399–451, 1934. 104. Gifford H: A study of the effect of retrobulbar anesthesia on ocular tension and the
vitreous pressure. Amer J Ophthalmol 32:1359–1368, 1949. 105. Gjotterberg M, Ingemnsson S-O: Effect on intraocular pressure of retrobulbarinjection of Xylocaine with
and without adrenaline. Acta Ophthalmologica 55:709–716, 1977. 106. O'Donoghue E, Batterbury M, Lavy T: Effect on intraocular pressure of local anaesthesia in eyes undergoing
intra-ocular surgery. Br J Ophthalmol 78:605–607, 1994. 107. Horven I: Ophthalmic artery pressure during retrobulbar anaesthesia. Acta Ophthalmologica 56:574–586, 1978. 108. Klein ML, Jampol LM, Condon PI et al: Central retinal artery occlusion without retrobulbar hemorrhage after retrobulbar
anesthesia. Amer J Ophthalmol 93:573–577, 1982. 109. Sullivan KL, Brown GC, Forman AR: Retrobulbar anesthesia and retinal vascular obstruction. Ophthalmology 90:373–377, 1983. 110. Rosenblatt RM, May DR, Barsoumian K: Cardiopulmonary arrest after retrobulbar
block. Am J Ophthalmol 90:425:-427, 1980. 111. Nicoll JMV, Acharya A, Ahlen K et al: Central nervous system complications after 6000 retrobulbar blocks. Anesth Analg 66:1298–1302, 1987. 112. Meyers EF, Ramirez RC, Boniuk I: Grand mal seizures after retrobulbar block. Arch Ophthalmol 96:847, 1978. 113. Javitt JC, Addiego R, Friedberg HL et al: Brain stem anesthesia after retrobulbar block. Ophthalmology 94:718–724, 1987. 114. Cohen SM, Sousa FJ, Kelly NE: Respiratory arrest and new retinal hemorrhages after retrobulbar anesthesia. Am J Ophthalmol 113:209–211, 1992. 115. Greenblatt DJ, Bolognini V, Koch-Weser J et al: Pharmacokinetic approach to the clinical use of lidocaine intravenously. JAMA 236:273–277, 1976. 116. Warner LO, Rogers GL, Marteno JD et al: Intravenous lidocaine reduces the
incidence of vomiting in children after surgery to correct strabismus. Anesthesiology 68:618–621, 988. 117. Lerman J, Kiskis AA: Lidocaine attenuates the intraocular pressure response to rapid intubation
in children. Can Anaesth J 32:339–345, 1985. 118. Wittpenn JR, Rapoza P, Sternberg P et al: Respiratory arrest following retrobulbar anesthesia. Ophthalmoloy 93:867–870, 1986. 119. Schimek F, Friess D, Fahle M: Plasma concentrations of lidocaine after proximal and peripheral facial
nerve blocks. German J Ophthalmol 4:315–319, 1995. 120. Gao F, Budd AJ: Venous levels of lignocaine and bupivacaine after peribulbar block. Anaesthesia 51:1109–1112, 1996. 121. Barr J, Kirkpatrick N, Dick A: Effects of adrenaline and hyaluronidase on plasma concentrations of lidocaine
and bupivacaine after peribulbar anaesthesia. Br J Anaesthesia 75:692–697, 1995. 122. Lerman J, Strong A, LeDez KM et al: Effects of age on the serum concentration of a1-acid glycoprotein and the binding of lidocaine in pediatric patients. Clin Pharmacol Ther 46:219–225, 1989. 123. Barry M, Keeling PWN, Weir D et al: Seventy of cirrhosis and the relationship of a1-acid glycoprotein concentration to plasma protein binding of lidocaine. Clin Pharmacol Ther 47:366–370, 1990. 124. Olson ML, McEvoy GK: Methemoglobinemia induced by local anesthetics. Am J Hosp Pharm 38:89–93, 1981. 125. Goggin M, Crowley K, O'Malley K et al: Serum concentrations of prilocaine following retrobulbar block. Br J Anaesth 64:107–109, 1990. 126. Eltzschig H, Rohrback M, Schroeder TH: Methaemoglobinemia after peribulbar blockade: an unusual complication in
ophthalmic surgery. Br J Ophthalmol 84:442, 2000. 127. Barker MR, McAllen MK: Hypersensitivity to local anaesthetics: a direct challenge test with lignocaine
for definitive diagnosis. BMJ 284:1229–1230, 1982. 128. Adriani J: Etiology and management of adverse reactions to local anesthetics. Int Anesthesiol Clin 10:127–151, 1972. 129. Incaudo G Schatz M, Patterson R et al: Administration of local anesthetic to patients with a history of prior
adverse reactions. J Allergy Clin Immunol 61:339–345, 1978. |