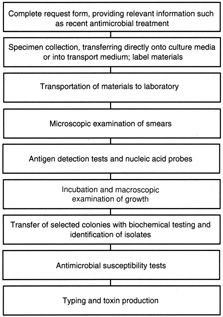
1. Wilhelmus KR, Liesegang TJ, Osato MS, et al. Laboratory Diagnosis of Ocular Infections, Cumitech 13A. Washington, D.C.: American Society for Microbiology, 1994 2. Byrne KA, Burd EM, Tabbara KF, et al: Diagnostic Microbiology and Cytology of the Eye. Boston: Butterworth Heinemann, 1995 3. Becan-McBride K, Ross DL: Essentials for the Small Laboratory and Physician's Office. Chicago: YearBook Medical Publishers, 1988 4. Huang JS, Russack V, Flores-Aguilar M, et al: Evaluation of cytologic specimens obtained during experimental vitreous
biopsy. Retina 13:160, 1993 5. Diamond J, Leeming J, Coombs G, et al: Corneal biopsy with tissue micro-homogenisation for isolation of
organisms in bacterial keratitis. Eye 13:545, 1999. 6. Kompa S, Langefeld S, Kirchhof B, et al: Corneal biopsy in keratitis performed with the microtrephine. Graefes Arch Clin Exp Ophthalmol 237:15, 1999 7. Ficker L, Kirkness C, McCartney A, et al: Microbial keratitis—The false negative. Eye 5:549, 1991 8. Smith RF: Microscopy and Photomicroscopy: A Working Manual. Boca Raton: CRC Press, 1990 9. Taylor HR, Munoz B, Keyvan-Larijani E, et al: Reliability of detection of microfilariae in skin snips in the diagnosis
of onchocerciasis. Am J Trop Med Hyg 41:467, 1989 10. Newell ED: Comparison of the use of skin scarification and skin biopsies to determine
the prevalence and intensity of Onchocerca volvulus infection. Ann Trop Med Parasitol 91:633, 1997 11. Deng XG, Li JC, Zhu L: [Laboratory diagnosis of Acanthamoeba keratitis]. Zhongguo Ji Sheng Chong Xue Yu Ji Sheng Chong Bing Za Zhi 18:301, 2000 12. Belin MW, Baltch AL, Hay PB: Secondary syphilitic uveitis. Am J Ophthalmol 92:210, 1981 13. Gopinathan U, Garg P, Fernandes M, et al: The epidemiological features and laboratory results of fungal keratitis: A 10-year
review at a referral eye care center in South India. Cornea 21:555, 2002 14. Nag DR, Chatterjee S, Khan M: Role of potassium hydroxide mount in rapid diagnosis of fungal corneal
ulcers. J Indian Med Assoc 100:18, 2002 15. Arffa RC, Avni I, Ishibashi Y, et al: Calcofluor and ink-potassium hydroxide preparations for identifying
fungi. Am J Ophthalmol 100:719, 1985 16. Thomas PA, Kuriakose T, Kirupashanker MP, et al: Use of lactophenol cotton blue mounts of corneal scrapings as an aid to
the diagnosis of mycotic keratitis. Diagn Microbiol Infect Dis 14:219, 1991 17. Thomas PA, Kuriakose T: Rapid detection of Acanthamoeba cysts in corneal scrapings by lactophenol cotton blue staining. Arch Ophthalmol 108:168, 1990 18. Glasgow BJ, Foos RY: Ocular Cytopathology, Boston: Butterworth-Heinemann, 1993 19. Chapin KC, Murray PR: Principles of stains and media. In: Murray PR, Baron EJ, Jorgensen JH, Pfaller MA, Yolken RH, eds. Manual of Clinical Microbiology, 8th ed. Washington, D.C.: ASM Press, 2003:257–266 20. Kean BH, Sun T, Ellsworth RM,: eds. Color Atlas: Text of Ophthalmic Parasitology, New York: Igaku-Shoin, 1991 21. Wilhelmus KR, Robinson NM, Tredici LL, et al: DB. Conjunctival cytology of adult chlamydial conjunctivitis. Arch Ophthalmol 104:691, 1986 22. Schachter J, Moncada J, Dawson CR, et al: Nonculture methods for diagnosing chlamydial infection in patients with
trachoma: A clue to the pathogenesis of the disease? J Infect Dis 158:1347, 1988 23. Sheppard JD, Kowalski RP, Meyer MP, et al: Immunodiagnosis of adult chlamydial conjunctivitis. Ophthalmology 95:434, 1988 24. Madhavan HN, Rao SK, Natarajan K, et al: Evaluation of laboratory tests for diagnosis of chlamydial infections in
conjunctival specimens. Indian J Med Res 100:5, 1994 25. Groden LR, Rodnite J, Brinser JH, et al: Acridine orange and Gram stains in infectious keratitis. Cornea 9:122, 1990 26. Sharma S, Kunimoto DY, Gopinathan U, et al. Evaluation of corneal scraping smear examination methods in the diagnosis
of bacterial and fungal keratitis: a survey of eight years of laboratory
experience. Cornea 21:643, 2002 27. Dunlop AA, Wright ED, Howlader SA, et al: Suppurative corneal ulceration in Bangladesh. A study of 142 cases examining
the microbiological diagnosis, clinical and epidemiological features
of bacterial and fungal keratitis. Aust N Z J Ophthalmol 22:105, 1994 28. Kanungo R, Srinivasan R, Rao RS: Acridine orange staining in early diagnosis of mycotic keratitis. Acta Ophthalmol 69:750, 1991 29. Seal DV, Bron AJ, Hay J: Ocular Infection: Investigation and Treatment in Practice, London: Martin Dunitz, 1998 30. Bharathi MJ, Ramakrishnan R, Vasu S, et al: Epidemiological characteristics and laboratory diagnosis of fungal keratitis: A
three-year study. Indian J Ophthalmol 51:315, 2003 31. Barza M, Pavan PR, Doft BH, et al: Evaluation of microbiological diagnostic techniques in postoperative endophthalmitis
in the Endophthalmitis Vitrectomy Study. Arch Ophthalmol 115:1142, 1997 32. Roychoudhury B, Sharma S, Reddy MK, et al: Fluorescent Gram stain in the microbiologic diagnosis of infectious keratitis
and endophthalmitis. Curr Eye Res 16:620, 1997 33. Wilhelmus KR, Peacock J, Coster DJ: The role of the Gram stain in the management of suppurative keratitis. In: Trevor-Roper PD, ed. The Cornea in Health and Disease. The VIth Congress of the European Society
of Ophthalmology. London: Royal Society of Medicine, 1981:399–402 34. Durfee K, Smith JP: Use of gram stain to predict vitreous fluid infection. Am J Med Technol 48:525, 1982 35. Gigliotti F, Williams WT, Hayden FG, et al: Etiology of acute conjunctivitis in children. J Pediatr 98:531, 1981 36. Fitch CP, Rapoza PA, Owens S, et al: Epidemiology and diagnosis of acute conjunctivitis at an inner-city
hospital. Ophthalmology 96:1215, 1989 37. Weiss A, Brinser JH, Nazar-Stewart V: Acute conjunctivitis in childhood. J Pediatr 122:10, 1993 38. Yoneda C, Dawson CR, Daghfous T, et al: Cytology as a guide to the presence of chlamydial inclusions in Giemsa-stained
conjunctival smears in severe endemic trachoma. Br J Ophthalmol 59:116, 1975 39. Streeten BW, Streeten EA: "Blue-body" epithelial cell inclusions in conjunctivitis. Ophthalmology 92:575, 1985 40. Duggan MA, Pomponi C, Kay D, et al: Infantile chlamydial conjunctivitis: a comparison of Papanicolaou, Giemsa
and immunoperoxidase staining methods. Acta Cytol 30:341, 1986 41. Dewan S, Mittal S, D'Souza , et al: Cytological evaluation of conjunctival scrape smears in cases of conjunctivitis. Indian J Pathol Microbiol 35:118, 1992 42. Cvenkel B, Globocnik M: Conjunctival scrapings and impression cytology in chronic conjunctivitis: correlation
with microbiology. Eur J Ophthalmol 7:19, 1997 43. Pirehma M, Suresh K, Sivanandam S, et al: Field's stain—A rapid staining method for Acanthamoeba spp. Parasitol Res 85:791, 1999 44. Daines BS, Vroman DT, Sandoval HP, et al: Rapid diagnosis and treatment of mycobacterial keratitis after laser in
situ keratomileusis. J Cataract Refract Surg 29:1014, 2003 45. Grossniklaus HE, Waring GO, Akor C, et al: Evaluation of hematoxylin and eosin and special stains for the detection
of Acanthamoeba keratitis in penetrating keratoplasties. Am J Ophthalmol 136:520, 2003 46. Gardner LM, Mathers WD, Folberg R: New technique for the cytologic identification of presumed Acanthamoeba from corneal epithelial scrapings. Am J Ophthalmol 127:207, 1999 47. Shah GK, Pfister D, Probst LE, et al: Diagnosis of microsporidial keratitis by confocal microscopy and the chromatrope
stain. Am J Ophthalmol 121:89, 1996 48. Drancourt M, Raoult D, Lepidi H, et al: Culture of Tropheryma whippelii from the vitreous fluid of a patient presenting with unilateral uveitis. Ann Intern Med 139:1046, 2003 49. Forster RK, Wirta MG, Solis M, et al: Methenamine-silver-stained corneal scrapings in keratomycosis. Am J Ophthalmol 82:261, 1976 50. Wear DJ, Malaty RH, Zimmerman LE, et al: Cat scratch disease bacilli in the conjunctiva of patients with Parinaud's
oculoglandular syndrome. Ophthalmology 92:1282, 1985 51. Catalano RA, Webb RM, Smith RS, et al: A modified immunoperoxidase method for rapid diagnosis of herpes simplex
I keratitis. Am J Clin Pathol 86:102, 1986 52. Collum LM, Mullaney J, Hillery M, et al: Two laboratory methods for diagnosis of herpes simplex keratitis. Br J Ophthalmol 71:742, 1987 53. Pflugfelder SC, Huang A, Crouse C: Epstein-Barr virus keratitis after a chemical facial peel. Am J Ophthalmol 110:571, 1990 54. Torres-Gomez J, Robinson NM, Osato MS, et al: Comparison of acridine orange and Gram stains in bacterial keratitis. Am J Ophthalmol 106:735, 1988 55. Hahn TW, O'Brien TP, Sah WJ, et al: Acridine orange staining for rapid diagnosis of Acanthamoeba keratitis. Jpn J Ophthalmol 42:108, 1998 56. Sutphin JE, Robinson NM, Wilhelmus KR, et al: Improved detection of oculomycoses using induced fluorescence with cellufluor. Ophthalmology 93:416, 1986 57. Wilhelmus KR, Osato MS, Font RL, et al: Rapid diagnosis of Acanthamoeba keratitis using calcofluor white. Arch Ophthalmol 104:1309, 1986 58. Chander J, Chakrabarti A, Sharma A, et al: Evaluation of calcofluor staining in the diagnosis of fungal corneal ulcer. Mycoses 36:243, 1993 59. Robin JB, Chan R, Rao NA, et al: Fluorescein-conjugated lectin visualization of fungi and acanthamoebae
in infectious keratitis. Ophthalmology 96:1198, 1989 60. Boerner CF, Lee FK, Wickliffe CL, et al: Electron microscopy for the diagnosis of ocular viral infections. Ophthalmology 88:1377, 1981 61. Van Rij G, Klepper L, Peperkamp E, et al: Immune electron microscopy and a cultural test in the diagnosis of adenovirus
ocular infection. Br J Ophthalmol 66:317, 1982 62. Wolters RW, Jorgensen JH, Calzada E, et al: Limulus lysate assay for early detection of certain Gram-negative corneal
infections. Arch Ophthalmol 97:875, 1979 63. Alfonso EC, Miller D: Rapid detection of gram-negative endotoxin contamination of contact
lens saline solutions. Arch Ophthalmol 110:1763, 1992 64. Levey SB, Katz HR, Abrams DA, et al: The role of cultures in the management of ulcerative keratitis. Cornea 16:383, 1997 65. Waxman E, Chechelnitsky M, Mannis MJ, et al: Single culture media in infectious keratitis. Cornea 18:257, 1999 66. Nachamkin I, Warren NG, Body BA: Eye specimens. In: Dalton HP, Nottebart HC Jr, eds. Interpretive Medical Microbiology. New York: Churchill Livingstone, 1986:771–845 67. Kaye SB, Rao PG, Smith G, et al: Simplifying collection of corneal specimens in cases of suspected bacterial
keratitis. J Clin Microbiol 41:3192, 2003 68. Marangon FB, Miller D, Alfonso EC: Impact of prior therapy on the recovery and frequency of corneal pathogens. Cornea 23:158, 2004 69. Collins C, Lyne P, Grange P, et al: Collins and Lyne's Microbiological Methods, 8th ed. London: Edward Arnold, 2004 70. Larone DH: Medically Important Fungi: a Guide to Identification, 4th ed. Washington, D.C.: ASM Press, 2002 71. Sutton DA, Fothergill AW, Rinaldi MG: Guide to Clinically Significant Fungi, Baltimore: Williams & Wilkins, 1998 72. Jansen B, Hartmann C, Schumacher-Perdreau F, et al: Late onset endophthalmitis associated with intraocular lens: A case of
molecularly proved S. epidermidis aetiology. Br J Ophthalmol 75:440, 1991 73. Matoba AY, Lee BL, Robinson NM, et al: Combination drug testing of Mycobacterium chelonae. Invest Ophthalmol Vis Sci 34:2786, 1993 |