GENERAL INDICATIONS
A muscle transposition procedure may be indicated when the function of one or more of the rectus muscles is absent or severely deficient as a result of third or sixth cranial nerve (CN-3, CN-6) palsy, lost muscle, type I Duane syndrome, supranuclear defects (as occur in double-elevator palsy), or in the rare case of congenital absence of one or more extraocular muscles.
BACKGROUND
Hummelsheim is credited with describing the first rectus muscle transposition procedure for treatment of paralytic strabismus in 1908.1 His procedure for treating the esodeviation that results from CN-6 palsy involved splitting the vertical recti and reattaching the temporal halves of each vertical rectus muscle adjacent to the insertion of the lateral rectus muscle. Several modifications to this procedure have been described, including those of O'Connor (1921),2 Berens and Girard (1950),3 Wiener and Scheie (1952),4 Hildreth (1953),5 Costenbader (1958),6 Schillinger (1959),7 Jensen (1964),8 Uribe (1968),9 and Knapp (1969).10 These modifications were reviewed by Helveston in 1971.11 More recently, alternatives have been suggested by Carlson and Jampolsky (1979),12 Kushner (1979),13 Rosenbaum and associates (1989),14 and Foster (1997).15 Even with these modifications, the original principle remains the same. All extraocular muscle transpositions that involve the rectus muscles shift forces of antagonist muscles to a rectus muscle that lies between them and acts in the opposite plane. For example, vertical recti are transposed nasally or temporally to enhance horizontal function, and horizontal recti are transposed superiorly or inferiorly to enhance vertical function. In this manner, a surgeon is able to alter both active contractile and passive elastic force vectors to augment the action of a deficient rectus muscle, improving alignment, ductions, and field of single binocular vision.
The most widely used procedures for rectus muscle transposition are the full-tendon transfer combined with either chemodenervation or recession of the antagonist of the paretic muscle, the Hummelsheim procedure, and the Jensen procedure. The most common indication for muscle transposition is lateral rectus weakness secondary to acquired CN-6 palsy. Transposition is indicated less often for superior rectus weakness such as occurs in double-elevator palsy and least often for inferior rectus palsy. Transposition is also useful when a rectus muscle has lost attachment to the globe and cannot be found and reattached. This occurrence most commonly is a complication of medial rectus surgery. Superior oblique transposition can be used for medial rectus paralysis when the vertical recti also are compromised, an occurrence most often associated with CN-3 palsy. Techniques for this procedure have been described by Jackson16 and also by Scott.17
CONTRAINDICATIONS
Restriction
Muscle transposition alone is not effective when free movement of the eye is restricted. Some mild restriction, or stiffness, of the antagonist of a paretic muscle invariably occurs after several months. This restriction is due to the effects of the chronic contracture of the antagonist of the paretic muscle. In most cases, it can be relieved by recessing the muscle or treating it with botulinum. When restricted movement is caused by abnormal attachments of the orbital fascia to other orbital fascia or to the globe, the attachments must be freed by surgery before an extraocular muscle transposition is performed if the best result from this surgery is to be obtained.
Vascular Insufficiency
This surgery is relatively contraindicated in older patients who have significant vascular disease because such patients have an increased risk of developing anterior segment ischemia after surgery. This topic is covered more fully in the section of this chapter on complications.
TECHNIQUES
Full-Tendon Transfer
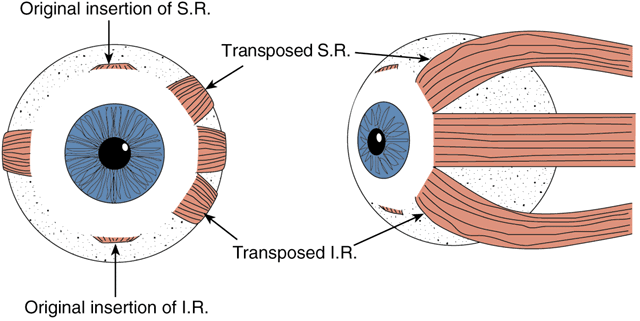
Our preferred technique for muscle transposition is a full-tendon transfer. Any two antagonist rectus muscles can be used for the transfer, but the most common method involves transposing the vertical recti temporally to treat lateral rectus palsy. This procedure can be performed through two fornix incisions, for example, superotemporal and inferotemporal, or through a large, 240-degree limbal peritomy, such as from the 5-o'clock position clockwise around to the 1-o'clock position in the right eye.
The superior rectus muscle is located, engaged on a muscle hook, and dissected from the overlying anterior Tenon's capsule and the intermuscular membrane at least 10 mm posteriorly. Attachments to the underlying superior oblique muscle also are divided. The muscle insertion is secured with a double-armed 6-0 absorbable suture (Vicryl, Biosorb) and disinserted from the globe. The inferior rectus muscle is exposed, secured, and disinserted in a similar fashion. Thorough dissection of the inferior rectus muscle from attachments to Lockwood's ligament is necessary to minimize lid fissure changes postoperatively.
The superior and inferior rectus muscles are then reattached to the globe in juxtaposition to the lateral rectus insertion. We prefer to suture the vertical recti so that the temporal corner of the transposed muscle abuts the adjacent corner of the lateral rectus insertion. The nasal corner of the transposed muscle is attached so that the new insertion of this muscle coincides with the spiral of Tillaux (Fig. 1). The antagonist muscle, in this case the medial rectus, must often be weakened. Our preferred method is by chemodenervation with botulinum A toxin. This method, when successful, obviates the need for recession of a third rectus muscle. A single injection of up to 5 U of botulinum may be given at the time of surgery, with the site of injection in the muscle belly under direct observation. As an alternative, botulinum may be given before or after the transposition procedure with the aid of electromyographic localization.
Some surgeons believe that there is an advantage to pretreating the restricted antagonist with botulinum several days before the transposition so that the drug has a chance to initiate relaxation of the antagonist muscle before the transposition and thereby enhance the effect of surgery. Usually, we inject the antagonist at the time of the transposition; it also can be effective if given after surgery. An initial overcorrection after muscle transposition with botulinum injection is desirable. This overcorrection resolves in several weeks as the effect of the botulinum diminishes. A potential side effect of botulinum treatment is ptosis from diffusion of the botulinum into the levator, which can persist for several weeks or months, but usually resolves.
If the surgeon chooses not to use botulinum, or if restriction persists despite treatment with botulinum, then recession of the antagonist muscle is indicated. In the case of a lateral rectus palsy, a recession of the ipsilateral medial rectus muscle, either with or without an adjustable suture, would be performed.
Anterior segment ischemia is always a potential complication of full-tendon transfer procedures. Saunders and Sandall18 reported two cases of anterior segment ischemia after a full-tendon transfer alone, but in both cases, the two horizontal recti had been operated on several years earlier. It is well known that anterior ciliary artery circulation is not re-established when a rectus muscle has been detached and reattached. Olver and Lee19 pointed out the greater risk of anterior segment ischemia after removal of the vertical recti because these muscles do not have an associated long posterior ciliary artery.
Normally transposed rectus muscle bellies are known to approach their new insertion at an angle approximating 45 degrees.20 This produces a significant gap between the borders of the paretic muscle and the two transposed rectus muscles (see Fig. 1). Magnetic resonance imaging (MRI) studies confirm that, despite significant displacement of the anterior insertion sites, transposition procedures produce relatively small changes in more posterior portions of the rectus muscle. Following vertical rectus muscle transposition to the lateral rectus, the path of the muscle belly posterior to the equator has been found to shift 3 mm or less.21
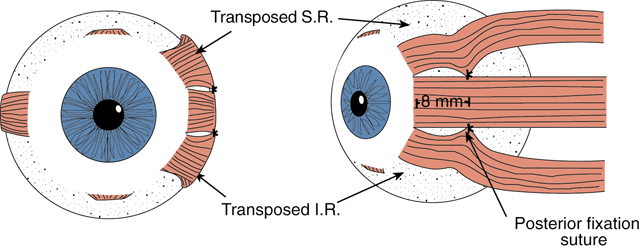
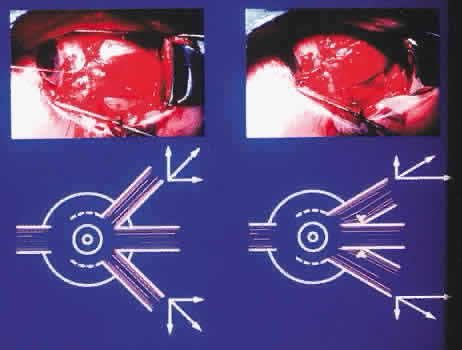
A modification of the traditional full-tendon transfer is Foster's procedure,15 which involves vertical muscle transposition augmented with a lateral fixation suture (Fig. 2). The goal of Foster's procedure is to close the gap between the transposed rectus muscles and the nondisplaced paretic muscle, thereby enhancing the desired force vectors of the transposed muscles (Fig. 3). For a lateral rectus palsy, a full-tendon transfer is performed as described above, with the addition of a 5-0 nonabsorbable, polyester fiber suture placed through the sclera adjacent to the border of the lateral rectus. This suture is placed 16 mm posterior to the limbus or approximately 8 mm posterior to the insertion of the lateral rectus; separate sutures are placed along the upper and lower borders of the muscle. After being anchored in the sclera, the sutures are passed through the adjacent transposed vertical rectus, incorporating 25% of its width. When the suture is pulled tight and tied, the gap between the muscles is closed as the borders are drawn into approximation and securely fixed at the equator.
|
|
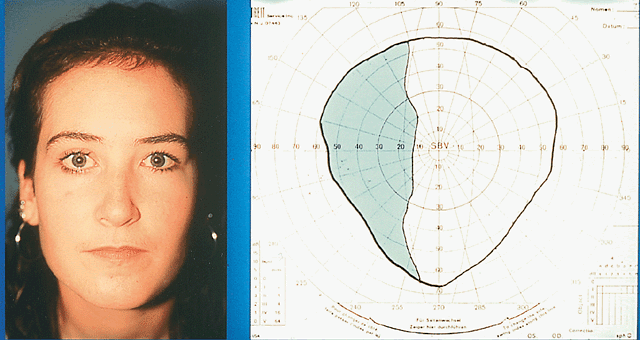
Lateral fixation suture-augmented transposition has been shown to increase the tonic force of the transposed vertical muscles significantly in cases of CN-6 palsy and type I Duane syndrome.15 This frequently results in satisfactory alignment and a larger diplopia-free visual field without requiring ipsilateral medial rectus recession (Fig. 4). Leaving the medial rectus unoperated reduces the risk of anterior segment ischemia or loss of adduction ability.
The Hummelsheim Procedure
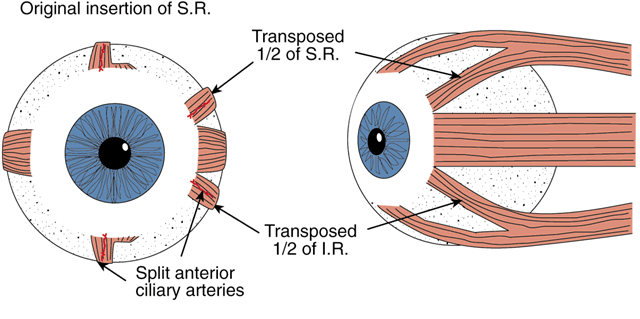
The technique for the Hummelsheim procedure is similar to that already described for the full-tendon transfer, except that the muscle is divided along its long axis, and only half the muscle is transposed. The other half remains attached to the globe with some vasculature presumably intact. This procedure is indicated for cases in which preservation of anterior segment circulation is a special concern.
Once the vertical recti have been identified, engaged on a muscle hook, and freed from surrounding attachments to intermuscular membrane and anterior Tenon's capsule, a small muscle hook is used to split the muscle and tendon, beginning at the insertion and extending approximately 15 mm posteriorly. The anterior ciliary arteries should be identified and the splitting done between the vessels, leaving intact the vessel in the half of the muscle that was not detached. That half of the muscle to be transposed is secured on a double-armed, absorbable 6-0 suture and disinserted. The same splitting procedure is performed on the direct antagonist. In a fashion similar to that used in a full-tendon transfer, the halves of the two muscles are then secured to the sclera, concentric with the limbus, at the edges of the insertion of the palsied or lost muscle (Fig. 5). The ipsilateral antagonist to the lost or palsied muscle should be weakened by chemodenervation or surgical recession, as already described above.
The Jensen Procedure
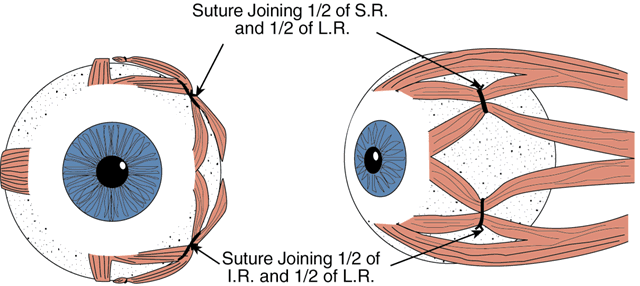
The Jensen procedure is performed by splitting the palsied muscle and joining the midportion of this muscle to the similarly split midportions of the two adjacent rectus muscles. The dissection and splitting are performed as described for the Hummelsheim procedure, but the muscles are not disinserted. Instead, a 5-0 nonabsorbable polyester fiber suture is used to join the muscle bellies to effect a union of the two muscles just anterior to the equator, as shown in Figure 6.
This procedure has little advantage over the full-tendon transfer or Hummelsheim procedure, other than theoretically reducing the risk of anterior segment ischemia. However, anterior segment ischemia has been reported after the Jensen procedure,22,23 so choosing this technique does not ensure avoidance of this complication. Significant scar tissue formation occurs with the Jensen procedure, making subsequent surgical exploration more difficult when compared with other tendon transfer techniques.
As with full- or half-tendon transfers, a weakening of the ipsilateral antagonist muscle is performed by chemodenervation or recession.
Superior Oblique Muscle Transposition
Before the superior oblique transposition is performed, the surgeon must verify that superior oblique muscle function is present. In the patient with complete CN-3 palsy, this function can be determined reliably by noting incyclotorsion of the involved exodeviated eye when the patient attempts to look down.
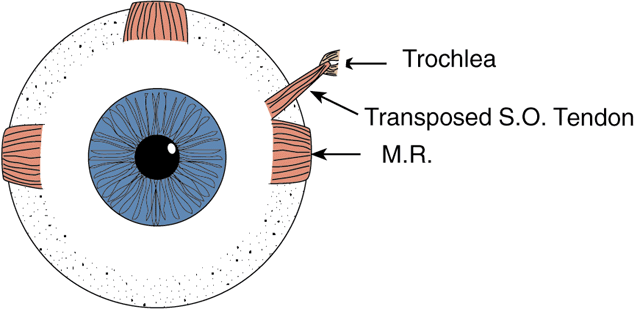
A limbal peritomy is made in the superonasal quadrant sufficient to provide exposure to both medial and superior recti. Traction sutures placed at 11- and 4-o'clock positions in the right eye (1- and 8-o'clock positions in the left eye) can facilitate exposure before the limbal incision is made. Alternatively, exposure to both the superior oblique and medial rectus can be obtained through a superonasal fornix incision. The superior oblique is isolated and dissected free of surrounding fascial attachments along the tendon, following underneath and along the nasal side of the superior rectus. After the superior oblique tendon is engaged on a small muscle hook on the nasal side of the superior rectus muscle, a suture is placed around the tendon, passed beneath the superior rectus, and pulled temporally, thus exposing the superior oblique insertion. The tendon is freed of surrounding attachments from the insertion toward the trochlear cuff. Dissection is limited to the sub-Tenon's space to avoid fat prolapse, which can produce postoperative restrictions.
A double-armed 6-0 absorbable suture is used to secure the superior oblique tendon at its insertion. The tendon is disinserted and brought down toward the superior edge of the medial rectus insertion. The superior oblique tendon will be redundant at this point. It is sutured to the sclera at the superior border of the medial rectus insertion at the point on the tendon that brings the eye to a centered position. It is unwise to shorten the superior oblique tendon excessively because doing so may produce an unwanted hypertropia or esotropia. Conversely, the tendon should not be left so long that the tonic contraction of the superior oblique muscle is not transmitted to the globe. After an appropriate length for the tendon is determined and it is sutured to the sclera, the excess tendon is excised (Fig. 7). A more physiologic, but also more difficult, superior oblique tendon transfer can be done after fracture of the trochlea. The trochlea may be fractured by passing a fine-tipped hemostat, spatula, or other instrument nasally along the tendon into the trochlea and then twisting or prying toward the orbit to force the arch of the trochlea open by fracture. The tendon then can be pulled free from the trochlea so that it passes straight to the new insertion site at the superior border of the medial rectus insertion. In adult eyes, the trochlea resists fracture, and the effort required to fracture it can lead to unintentional severance of the superior oblique tendon, making the transfer procedure impossible. For practical reasons, trochlea fracture should be confined to young patients, if it is done at all. We favor leaving the trochlea intact in most cases.
|
RESULTS
The results obtained from the transposition procedure vary with both the technique and the underlying problem. Specific results are covered in the following discussions of several common indications.
COMPLICATIONS
Anterior Segment Ischemia
Anterior segment ischemia is caused by compromising more than a critical amount of anterior segment circulation during strabismus surgery. This complication has been reported after detachment of only two rectus muscles, conversely, all four rectus muscles have been detached in other patients without producing this clinical picture. The risk of this complication increases whenever a third rectus muscle is detached in the same eye, especially if two of these muscles are vertical recti, because vertical recti do not have an associated posterior ciliary artery. If surgery on a fourth rectus muscle becomes necessary, a procedure that spares the anterior ciliary artery can be used,24 or botulinum injection to the muscle can be considered.