INDICATIONS
Surgery is indicated for the treatment of infantile esotropia when the size of the deviation is stable, amblyopia has been treated, and an accommodative component of the esodeviation has been eliminated.
Every effort to correct amblyopia should be made prior to surgery; however, Lam and associates7 showed that amblyopia therapy could be successfully continued after surgery is done and that realignment itself can help reverse amblyopia in some cases. Parents should be advised that occlusion might be necessary postoperatively. Occlusion therapy for amblyopia does not seem to result in measureable angle variation in patients with infantile esotropia.8 An accommodative component is said to be present in 15% of patients with infantile esotropia,9 and may be treated with glasses when the hyperopic refractive error is greater than 2.25 diopters. When the refractive error is less than 2.25 diopters, echothiophate iodide (phospholine iodide 0.125%, Wyeth, Philadelphia, PA) instilled in the eye each morning may reduce the size of the deviation by reducing the amount of accommodative convergence. However, its use for this is decreasing in popularity by pediatric ophthalmologists.
The measured deviation should be stable over two office visits. The Pediatric Eye Disease Investigator Group10–12 showed that esotropia with an angle of less than 40 prism diopters and with onset in early infancy frequently resolves; esotropia with a deviation greater than or equal to 40 prism diopters presenting after 10 weeks of age has a low likelihood of resolution. Although surgery technically can be performed safely in patients who are as young as 3 to 4 months of age, many surgeons prefer to wait until the patient is 6 months of age to permit stability of the ocular deviation.
Ing3 reported that patients with infantile esotropia who underwent surgical alignment before the age of 24 months showed improved binocular function compared with those who achieved alignment after the age of 2. Birch and associates13 showed that although surgical correction of infantile esotropia during the first year of life is not associated with a higher prevalence of steroacuity, it is associated with better stereoacuity in children who achieve stereopsis after surgery. Early surgical alignment is thought to be associated with better stereopsis because the duration of misalignment is shortened.14,15
CONTRAINDICATIONS
Surgery should not be performed to correct infantile esotropia when the patient has a life-limiting condition or a medical condition that precludes the safe administration of anesthesia. Surgery should be delayed when the accommodative component has not been treated or when measurements are unstable.
PROCEDURE
Surgical techniques used to correct infantile esotropia include bilateral medial rectus recession,16 unilateral recession of the medial rectus combined with resection of the lateral rectus,17 and bilateral medial rectus recession combined with resection of one or both lateral rectus muscles3,18,19
Recession of both medial rectus muscles is the most commonly performed operation to correct infantile esotropia. However, when one eye is preferred, some surgeons treat esotropia with a recession of the medial rectus muscle combined with a resection of the lateral rectus muscle on the nonpreferred eye.17 Because of concern that large recessions of the medial rectus muscle (greater than 7 mm) will limit adduction and that smaller recessions will produce undercorrection in large angle infantile esotropia, some surgeons combine bilateral medial rectus recessions with resection of one or both lateral rectus muscles when the angle of esotropia is 70 prism diopters or more.18,19 Other surgeons feel that in children younger than 1 year of age, esotropia up to 70 prism diopters can safely be corrected with surgery on both medial rectus muscles.20 Table 1 shows a guide to the quantity of surgery we perform.
The operation should be designed so that it corrects the entire deviation and places the eyes in parallel alignment at distance and near. Measurements are made with the alternate prism and cover test when possible; in infants, the Krimsky test is used.
The amount of correction achieved by recessing the medial rectus muscle can be augmented by recessing the conjunctiva and anterior Tenon's tissue with the use of techniques advocated by Knapp21 and Helveston and colleagues.22 This en bloc surgical technique helps to reduce the restrictive component caused by contracted conjunctiva and Tenon's capsule when a deviation has been present for a long time
The medial rectus muscle can be approached through either a cul-de-sac (fornix) incision23 or an incision directly along the limbus.24 Long-term cosmetic differences between these two surgical approaches are insignificant, but patients seem to be more comfortable in the earlier postoperative period with the fornix-based incision, and the amount of swelling and scarring visible when the eyelids are open is less. The choice of conjunctival incision is best left to the individual surgeon.
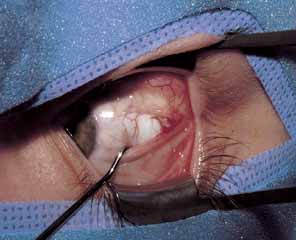
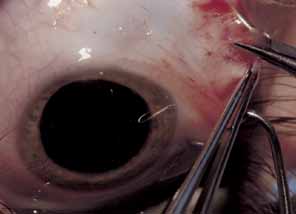
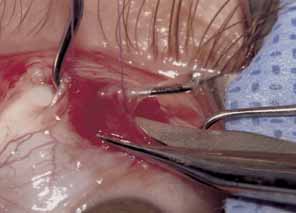
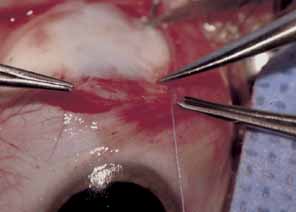
The muscle is then exposed; Tenon's capsule and the intramuscular septum are then dissected free (Figs. 1, 2, and 3). For a rectus muscle recession, a double-armed absorbable suture with a half circle needle is passed transversely through the muscle tendon, approximately 1 mm posterior to its insertion (Fig. 4 and 5). This suture is secured in place at both the superior and inferior poles of the muscle with locking bites. The muscle is disinserted from the globe.
|
|
|
|
|
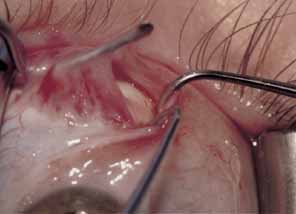
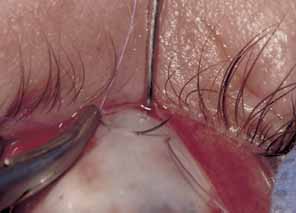
A Castroviejo caliper is used to measure a point on the sclera posterior to the insertion. When measurements are made from the original insertion, the posterior ridge of the original insertion is used (Fig. 6). This landmark is convenient and distinct. The effect of recession may vary if measurements are not always made from the same location. The sclera is marked directly posterior to the upper and lower poles of the original insertion to avoid any infraplacement of supraplacement effect. Some authors prefer to use a curved Scott ruler to achieve these measurements; others measure the distance for recession from the limbus.25 Each method is acceptable, and if used consistently will yield reproducible results.
|
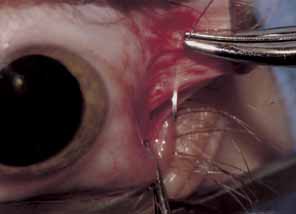
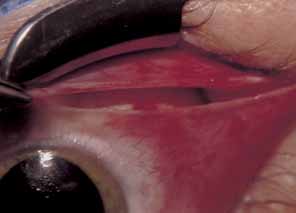
The previously placed intramuscular suture is passed within the sclera where marked, reattaching the muscle to the globe (Fig. 7). The needles are passed in a crossed-swords fashion so that their tips cross. This maneuver creates one site where both needles exit the sclera, and facilitates tying the knot.
The conjunctiva is pulled over the muscle and closed according the surgeon's preference (Fig. 8). Some prefer to leave the small cul-de-sac incision unsutured; others close this incision with interrupted sutures (see Fig. 9) Differences in postoperative discomfort or complications between these two approaches are insignificant.
|
Placement of scleral sutures may be difficult when large recessions are performed to correct congenital esotropia. Difficulty results from the poor exposure caused by the relatively small size of the globe and the confined space of the orbit and its surrounding tissues. In this situation, the hang-back recession is favored by some surgeons.26,27 Sutures are placed through the original insertion, and a series of knots is tied. The muscle is allowed to slide back a measured amount to its new insertion. Studies have shown this approach to be as effective as traditional recession techniques.26
Esotropia up to 28 prism diopters can be corrected with recession of just one medial rectus muscle.28 The advantages of unilateral surgery include the opportunity to use local anesthesia in adults, shortened operating and anesthesia time, and exposure of just one eye to the risks of strabismus surgery. Table 3 provides a guide to the quantity of surgery we perform.
Some surgeons prefer to combine bilateral medial rectus recession with resection of the lateral rectus muscle tendon. The lateral rectus muscle is isolated; Tenon's tissue and the intramuscular septum are dissected away for approximately 3 to 4 mm more than the desired resection. The amount of muscle tendon to be resected is measured from the insertion with a caliper. A double-armed absorbable suture is placed through the center of the muscle; it is passed transversely to the margin, where a locking bite is taken. A second suture is placed across the remaining muscle half and locked at the margin. A hemostat is then placed across the entire muscle, just anterior to the sutures. The muscle is disinserted from the globe, leaving a small stump. Muscle anterior to the hemostat is excised. The previously placed sutures are passed through the original insertion, each in a crossed swords fashion, so that the needle tips cross. The muscle is then pulled up and tied in place. Conjunctiva is pulled over the muscle and closed according to the surgeon's preference.
EXPECTED RESULTS
Ideally, children who are surgically treated for infantile esotropia should have no deviation of the visual axis at distance and near. However, surgery generally is considered successful when the postoperative deviation is within 10 prism diopters of orthotropia. Caputo and co-workers29 reported better long-term results with postoperative measurements of up to 10 prism diopters of esotropia. In their study group, all of the patients who had consecutive exotropia were wither orthophoric or exotropic during the immediate postoperative period.
Ing and associates30 reported at 54% success rate with bimedial recession in 50 patients who had esotropia of 50 prism diopters or more; however, this report was published in 1966, when maximal recession of only 5 mm was done.
More recent series advocate larger amounts of medial rectus recession Hess and Calhoun31 were early proponents of recessing the medial rectus muscles more than the traditional 5 mm. They reported a success rate of 84% in patients who underwent 6-mm recession for esotropia that averaged 57 prism diopters, and a success rate of 60% in patients who underwent 7-mm recession for an average preoperative deviation of 78 prism diopters. Prieto-Diaz32 and Nelson and co-workers33 reported similar success with 6- and 7-mm recessions of the medial rectus muscles in patients followed for 3 years or more.
In an earlier study, von Noorden and colleagues reported a success rate of 42% with a recess–resect procedure performed on the nonfixating eye. The 58% undercorrection rate in this study probably can be attributed to inadequate amounts of medial rectus recession; this study was reported in 1972, when 5-mm recession was considered maximal. Long-term controlled studies are not available to compare the effect of the recess-resect procedure with symmetric medial rectus recession for the correction of infantile esotropia.
Foster and associates34 reported a success rate of 79% with bilateral medial rectus recession and resection of one or both lateral rectus muscles for correction of congenital esotropia. Lee and Dye18 reported a success rate of 61% in their series with this same approach. Scott and associates19 compared recession of the medial recti with medial rectus recession combined with resection of one or both lateral rectus muscles. Good surgical results were found in 37.3% of patients who underwent bimedial recession compared with 64.5% of those who underwent surgery on three or four muscles. However, maximal recession in the bimedial recession group was 5.5 mm.
Successful alignment early in life does not ensure long-term stability. Frequent follow-up visits are recommended; these visits should continue at least through the first 5 years of life. Hiles and co-workers35 performed a retrospective study of a group of 54 consecutive patients who had infantile esotropia and underwent surgical alignment before the age of 1 year. These patients were examined annually until age 10. The patients fell into three distinct groups over the observation period; those who retained stable ocular alignment after the first operation (39%), those who had good alignment that remained stable for a period and then decompensated (31%) and those who had unstable alignment throughout the study period (30%). Secondary operations were required in 69% of patients, and a third operation required in 11%. Further surgery was needed for esotropia in 38%, for exotropia in 12%, and for inferior oblique overaction either alone or in combination with horizontal surgery in 44%. This study was conducted at a time when effective surgery to correct dissociated vertical deviation was not available. In addition to these other procedures, additional operations to correct DVD may be necessary.
A review of our experience with infantile esotropia supports these observations. We retrospectively examined the records of 105 patients who underwent bimedial recession before the age of 18 months and were available for 5-year follow up. Secondary surgery was required in 36% of patients, and a third procedure needed in 12%. Surgery for esotropia was needed in 24%, exotropia in 10%, and inferior oblique surgery either alone or in combination with horizontal surgery in 30%.
Bifixation rarely develops in patients who have infantile esotropia, regardless of age at surgical alignment.36 Clinical evidence suggests that alignment before 2 years of age is associated with development of some degree of binocular vision.2,3,37 Hiles and co-workers35 used the Worth four-dot test and found a fusion response at distance in 19% and near in 63% of patients. Some degree of stereoacuity was obtained in 29% of patients. Our results were similar: 7% showed a fusion response at distance, 58% showed a fusion response at near, and 32% exhibited some degree of stereoacuity.
COMPLICATIONS AND THEIR MANAGEMENT
Unsatisfactory Alignment
Unsatisfactory alignment is the most common complication of surgery for congenital esotropia. Once satisfactory alignment has been obtained, it may not be lasting; decreased vision, lack of fusion and contraction of scar tissue may be destabilizing factors.
Alignment of the eyes should be assessed 6 weeks to 2 months after surgery. If the size of a residual deviation is unsatisfactory, additional surgery should be scheduled. If the alignment is good and the patient does not need glasses or treatment for amblyopia, periodic visits 9 months to 1 year apart are scheduled. These follow-up visits are indicated because of the frequent development of ancillary findings in patients with infantile esotropia; DVD, inferior oblique overaction, amblyopia, and further horizontal misalignment.
Amblyopia is a common postoperative finding in infantile esotropia. Ing reported an incidence of 46%.3 Hiles and co-workers35 found that 17% of their patients exhibited amblyopia at some point over the 10 years after surgery; 32% of the patients in our study had visual asymmetry. When needed, amblyopia is treated with occlusion therapy and optical correction. Maintenance or part-time patching may be necessary until the patient reaches 8 or 9 years of age. Occlusion and correction of amblyopia frequently will assist in correction of small overcorrections or undercorrections.
UNDERCORRECTIONS
Undercorrection of 10 prism diopters of more may respond to treatment of hyperopia when a refractive error of +1.50 or more is present. A trial of 6 weeks is indicated.
Residual esotropia of 15 prism diopters or more 6 weeks postoperatively without amblyopia or an accommodative component is an indication for additional surgery. This surgery should be performed as soon as reasonably possible; surgery generally is performed on the unoperated horizontal muscles. Bilateral medial rectus recession is followed by unilateral or bilateral lateral rectus resection,38 depending on the size of the residual esotropia. When medial rectus restrictive factors or obvious errors in magnitude of the original surgery are noted, medial rectus rerecession or marginal myotomy combined with lateral rectus resection can be performed. If a monocular recession–resection was performed because the original procedure and alignment is unsatisfactory, a similar procedure is performed on the fellow eye to correct alignment.
OVERCORRECTION
Overcorrection almost always requires reoperation. Small overcorrections may be improved by correction of astigmatic or myopic refractive errors. Despite initial satisfactory alignment, exotropia can develop over the long term.29 Exotropia of 15 prism diopters or more 6 weeks postoperatively is an indication for further surgery. The size of the deviation will dictate whether recession of one or both lateral rectus muscles should be performed. Advancement or resection of the medial rectus muscle may be performed when limited ductions or underaction are noted postoperatively.
Associated Conditions
Accommodative esotropia, dissociated vertical deviation, inferior oblique muscle overaction, an abnormal head position, and latent nystagmus are conditions frequently associated with infantile esotropia. Even after initial surgical correction of the esotropia, alignment may be unstable.
Parents and surgeons should be prepared for the possible need for additional surgical procedures. Often, further surgery is required for correction of residual esotropia, recurrent esotropia, consecutive exotropia, inferior oblique overaction or DVD. Reoperation rates as high as 69% have been reported in children whose initial correction was performed before 1 year of age.35 Regular monitoring, the frequency of which is established by the visual acuity and the alignment of the eyes, is essential during the first several years of life. Surgical correction of inferior oblique overaction and dissociated vertical deviation is discussed in other chapters.