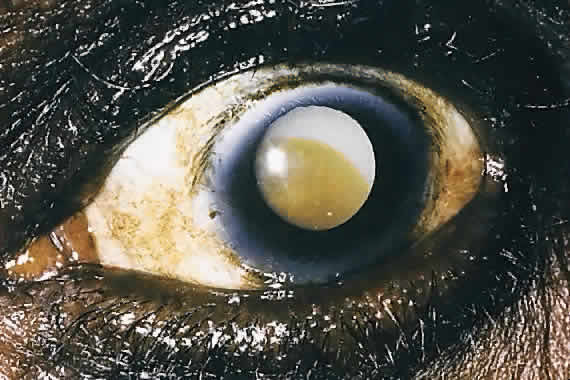
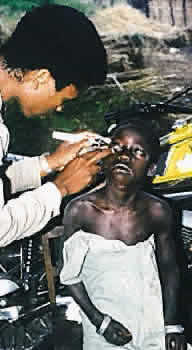
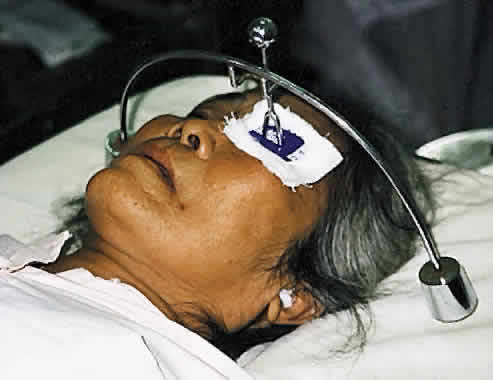
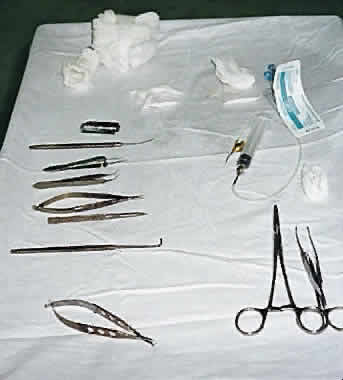
1. Global Initiative for the Elimination of Avoidable Blindness: World Health
Organization Programme for Prevention of Blindness and Deafness. World
Health Organization, Geneva, Switzerland, 1997 2. Staff Appraisal Report: India Cataract Blindness Control Project Report
No. 12246-IN. World Bank, Washington, DC, 1993 3. Thylefors B: Documenta: The WHO programme for the prevention of blindness and cataract
in developing countries. Ophthalmologica 81:339–344, 1992 4. WHO Programme for Prevention of Blindness 1990. PBL 91.1. World Health
Organization, Geneva, Switzerland, 1990 5. Chatterjee A, Milton RC, Thyle S: Prevalence and aetiology of cataract in Punjab. Br J Ophthamol 66:35–42, 1982 6. Lepkowski JM, Zurita B, Brilliant GE et al: Epidemiology of cataract in
South India. 1989 Mimeograph cited in Drummond MF: Economic aspects of
cataract blindness. In Kupfer C, Gillen T (eds): World Blindness and
Its Prevention. Vol 4. International Agency for the Prevention of Blindness, Haywards
Heath, England, 1990 7. Brilliant LB, Pokhrel RP, Grasset NC et al: Epidemiology of blindness in Nepal. Bull WHO 63:375–386, 1985 8. Minassian DC, Mehra V, Johnson GJ: Mortality and cataract findings from a population-based longitudinal study. Bull WHO 70:219–223, 1992 9. Marseille E: Cost effectiveness of cataract surgery in a public health eye care programme
in Nepal. Bull WHO 74:319–324, 1996 10. Ellwein LB, Kupfer C: Strategic issues in preventing cataract blindness in developing countries. Bull WHO 73:681–690, 1995 11. Taylor HR: The epidemiology of age-related cataract. Eye 13:445–448, 1999 12. Klein BEK, Klein R, Ritter LL: Is there evidence of an estrogen effect on aged related lens capacities? The
Beaver Dam Eye Study. Arch Ophthalmol 112:85–91, 1994 13. Heiba IM, Elston RC, Klein BEK et al: Genetic etiology of nuclear cataract: Evidence for a major gene. Am J Med Genet 47:1208–1214, 1993 14. Heiba IM, Elston RC, Klein BEK: Evidence for a major gene for cortical cataract. Invest Ophthalmol Vis Sci 36:227–225, 1995 15. McCarty CA, Mukesh BN, Fu CL, Taylor HR: The Epidemiology of cataract in Australia. Am J Ophthalmol 128:446–465, 1999 16. Taylor HR, West SK, Rosenthal FS et al: Effect of ultraviolet radiation on cataract formation. N Engl J Med 319:1429–1433, 1988 17. World Health Organization: The Effects of Solar UV Radiation on the Eye: Report
of an Informal Consultation. WHO/PBL/EHG/94.1, Geneva: World
Health Organization, 1994 18. Taylor A, Jacques PF, Epstein EM: Relations among aging, antioxidant status and cataract. Am J Clin Nutr 62:1439S–1447S, 1995 19. Vitale S, West SK, Hallfrisch J et al: Plasma antioxidants and risk of cortical and nuclear cataract. Epidemiology 4: 195–203, 1993 20. Mares-Perlman JA, Klein BEK, Klein R et al: Relation between lens opacities and vitamin and mineral supplement use. Ophthalmology 101:315–325, 1994 21. Rouhiainen P, Rouhiainen H, Salonen JT: Association between low plasma vitamin E concentration and the progression
of early cortical lens opacities. Am J Epidemiol 144:496–500, 1996 22. Leske MC, Wu S-Y, Connell AMS et al: The Barbados Eye Study Group: Lens opacities, demographic factors and nutritional
supplements in the Barbados eye study. Int J Epidemiol 26:1314–1322, 1997 23. Minassian DC, Mehra V, Jones BR: Dehydration crises from severe diarrhoea or heatstroke and risk of cataract. Lancet 1:751–753, 1984 24. Minassian DC, Mehra V, Verry J-D: Dehydration crises: Major risk factor
in blinding cataract. Br J Ophthalmol 1989;73:100–105 25. Kahn MU, Kahn MR, Sheikh AK: Dehydrating diarrhoea and cataract in rural Bangladesh. Indian J Med Res 85:311–315, 1987 26. West SK, Valmadrid CT: Epidemiolgy of risk factors for age-related cataract. Surv Ophthalmol 39:323–334, 1995 27. Ederer F, Hiller R, Taylor HR: Senile lens changes and diabetes in two population studies. Am J Ophthalmol 91: 381–395, 1981 28. Hodge WG, Whitcher JP, Satariano W: Risk factors for age-related cataracts. Epidemiol Rev 17:336–345, 1995 29. Cumming RG, Mitchell P, Leeder SR: Use of inhaled corticosteroids and the risk of cataracts. N Engl J Med 337:8–14, 1997 30. Garbe E, Suissa S, LeLorier J: Association of inhaled corticosteroid use with cataract extraction in elderly
patients. JAMA 280:539–543, 1998 31. Solberg Y, Rosner M, Belkin M: The association between cigarette smoking and ocular disease: Public health
and the eye. Surv Ophthalmol 42:535–547, 1998 32. Harding JJ, van Heyningen R: Drugs, including alcohol, that act as risk factors for cataract, and possible
protection against cataract by aspirin-like analgesics and cyclopenthiazide. Br J Ophthalmol 72:809–814, 1988 33. Munoz B, Tajchman U, Bochow TW et al: Alcohol use and risk of posterior subcapsular opacities. Arch Ophthalmol 111:110–112, 1993 34. Brilliant G, Brilliant L: Using social epidemiology to understand who stays blind and who gets operated
on for cataract in a rural setting. Soc Sci Med 21:553–558, 1985 35. Natchiar G, Robin AL, Thulasiraj RD et al: Attacking the backlog of India's curable blind. Arch Ophthalmol 112:987–993, 1994 36. Courtright P, Kanjaloti S, Lewallen S: Barriers to acceptance of cataract surgery among patients presenting to
district hospitals in rural Malawi. Trop Geogr Med 47:15–18, 1995 37. Brilliant GE, Lepkowski JM, Zurita B et al: Social determinants of cataract surgery utilization in South India. Arch Ophthalmol 109:584–589, 1991 38. Reshef DS, Refshef SH: Postoperative cataract surgery satisfaction in a rural Kenyan clinic. J Cataract Refract Surg 23:575–580, 1997 39. Hogeweg M, Sapkota YD, Foster A: Acceptability of aphakic correction: Results from Karnali eye camps in
Nepal. Acta Ophthalmol (Copenhagen) 70:407–412, 1992 40. Faal H, Minasian D, Sowa S et al: National survey of blindness and low vision in The Gambia. Br J Ophthalmol 73:82–87, 1989 41. Tabbara KF, Ross-Degnan D: Blindness in Saudi Arabia. JAMA 255:3378–3384, 1986 42. Gupta AK, Ellwein LB: The pattern of cataract surgery in India: 1992. Indian J Ophthalmol 43:3–8, 1995 43. Limberg H, Kumar R, Bachani D: Monitoring and evaluating cataract intervention in India. Br J Ophthamol 80:951–955, 1996 44. Natchiar GN, Thulasiraj RD, Negrel AD et al: The Madurai intraocular lens study: I. A randomized clinical trial comparing
complications and vision outcomes of intracapsular cataract extraction
and extracapsular cataract extraction with posterior chamber intraocular
lens. Am J Ophthalmol 125:1–13, 1998 45. Venkatesh Prajna N, Chandrakanth KS, Kim R et al: The Madurai intraocular lens study: II. Clinical outcomes. Am J Ophthalmol 125:15–25, 1998 46. Fletcher A, Vijaykumar V, Selvaraj S et al: The Madurai intraocularlens study: III. Visual functioning and quality
of life outcomes. Am J Ophthalmol 125:26–35, 1998 47. Hennig A Evans JR, Pradhan D et al: Randomised controlled trial of anterior-chamber intraocular lenses. Lancet 349: 1129–1133, 1997 48. The South Asian Cataract Management Study Group: I. The first 662 cataract
surgeries: A preliminary report. Br J Ophthalmol 79:1029–1035, 1995 49. Cook CD, Evans JR, Johnson GJ: Is anterior chamber lens implantation after intracapsular cataract extraction
safe in rural black patients in Africa? A pilot study in KwaZulu-Natal, South
Africa. Eye 12:821–825, 1998 50. Braganza A, Cheng AC, Thomas R et al: Surgical magnification for intracapsular cataract surgery in a rural hospital. Indian J Ophthalmol 44:179–182, 1996 51. Limburg H, Vaidyanathan K, Pampattiwar KN: Cataract blindness on the rise? Results of a door-to-door examination in
Mohadi. Indian J Ophthalmol 44:241–244, 1996 52. Killestein J, Hillegers M, van der Windt C et al: Outcome of cataract surgery by a general medical doctor at district level, Zimbabwe: A
retrospective follow-up study. Int Ophthalmol 20:279–283, 1996-1997 53. Chew A: Intracapsular cataract extraction: Experience of a general surgeon in Niger, West
Africa. Aust NZ J Ophthalmol 25:43–46, 1997 54. Connell B, Brian G, Bond MJ: A case-control study of biometry in healthy and cataractous Eritrean eyes. J Ophthalmol Epidemiol 4:151–155, 1997 55. La Nauze J, Thanh TK, Lap TD et al: Intraocular lens power prediction in a Vietnamese population: A study to
establish a limited set of intraocular lens powers that could be used
in a Vietnamese population. J Ophthalmol Epidemiol 6:147–158, 1999 56. Tobin S, Dat N, Binh P et al: Extracapsular cataract surgery in Vietnam: A 1 year follow-up study. Aust NZ J Ophthalmol 26:13–17, 1998 57. Dat N, Binh P, La Nauze J et al: A Two Year Follow Up Study of Extrcapsular
Cataract and Posterior Chamber IOL Surgery. Sydney, Australia: Fred
Hollows Foundation, 2000 (in press) 58. Lertsumitkul S, La Nauze J: Cost-minimization analysis of capsulotomy after ECCE and IOL implantation. Ophthalmic Epidemiol 3:77–83, 1996 59. Venkataswamy G: Massive eye elief in eye camps. Am J Ophthalmol 79:137–138, 1975 60. Report of the National Trachoma and Eye Health Programme, Royal Australia
College of Ophthalmology, Sydney, Australia, 1980, pp 161–170 61. Civerchia L, Ravindran RD, Apoorvananda SW et al: High-volume intraocular lens surgery in a rural eye camp in India. Ophthalmic Surg Lasers 27:200–208, 1996 62. Murthy GV, Sharma P: Cost analysis of eye camps and camp-based cataract surgery. Natl Med J India 7:111–114, 1994 63. Gillies M, Brian G, La Nauze J et al: Modern surgery for global cataract blindness. Arch Ophth 116:90–92, 1998 64. Natchiar G, Robin AL, Thulasiraj RD et al: Attacking the backlog of India's curable blind: The Aravind Eye Hospital
model. Arch Ophth 112:987–993, 1994 |