FLUORESCEIN Generations of clinicians have been taught about the clinical taboo of
instilling fluorescein in the presence of a hydrogel lens. Indeed, it
is a considerable disservice to discolor an aphakic or cosmetic hydrogel
lens with fluorescein. However, with therapeutic lens use, and especially
with disposable lenses, the need for fluorescein staining of the
cornea outweighs any concern for lens spoilage and temporary discoloration. In evaluating the corneal epithelium, it is often helpful to instill a
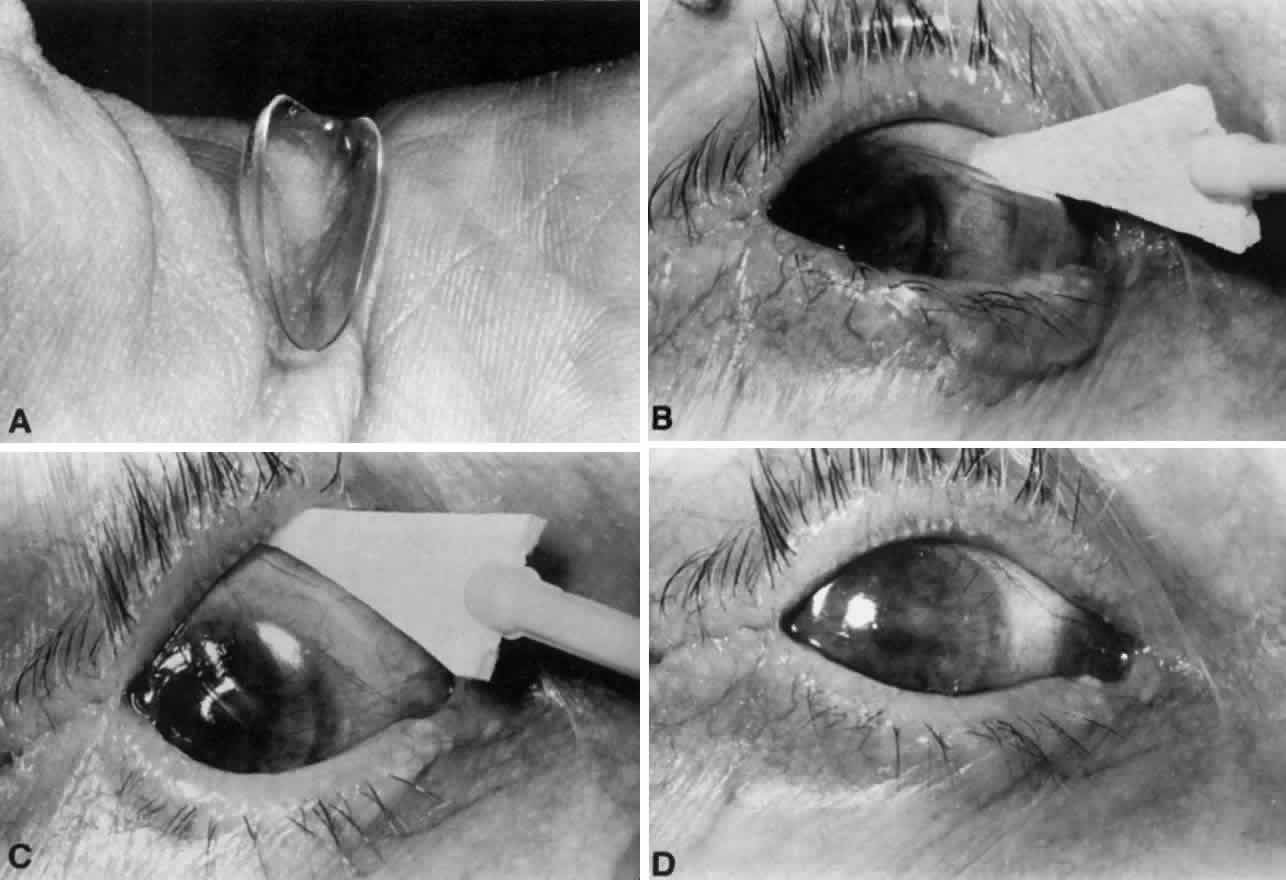
fluorescein-containing topical anesthetic solution, place the patient
at the slit lamp, slide the therapeutic lens temporally with a sterile
cotton-tipped applicator, and ask the patient to blink once or twice. The
corneal surface can then be evaluated with cobalt blue illumination
before the contact lens is blinked or moved back into position with
a cotton-tipped applicator. TONOMETRY Intraocular pressure must be monitored periodically during the use of a
therapeutic lens. This can be accomplished with minimal ocular trauma
by sliding the lens as described above before using an applanation tonometer. Alternatively, and with somewhat less trauma to the cornea, less
difficulty for the examiner, and reasonable accuracy, the intraocular
pressure can be measured with a Tono-Pen or Pneumatonometer with the
contact lens in situ.8,9 TOPICAL MEDICATIONS Topical medications administered for infectious keratitis, inflammation, or
glaucoma may often accompany the use of a therapeutic lens. There
is considerable uncertainty as to the precise amount of drug delivered
to the cornea and anterior segment with a hydrogel lens in place. This
issue has considerable clinical importance, which McCarey addressed
by using a mathematical model of gentamicin delivery. This model demonstrated
that although diffusion through the lens was increased by the
presence of a higher water content, the more important pathway for drug
delivery was around the edge of the lens.10 PRESERVATIVES There is concern about the safety of using topical medications containing
preservatives in the presence of a therapeutic lens. Lemp11 found that keratoconjunctivitis sicca patients treated with bandage lenses
and frequent applications of preserved artificial tear solutions
showed no accumulation of the preservative benzalkonium chloride in the
contact lens after several weeks of treatment, nor any evidence of corneal
epithelial toxicity. This is consistent with clinical experience
using topical antibiotics and corticosteroids in the presence of therapeutic
lenses without notable lens deterioration or epithelial toxicity. As
always, especially with an already compromised ocular surface, preserved
topical medications should be kept at the minimum level compatible
with the desired therapeutic response. PROPHYLACTIC TOPICAL ANTIBIOTIC There is no definite consensus on the precise indications for topical antibiotic
prophylaxis when using a therapeutic hydrogel lens, or on the
antibiotic of choice. It would seem reasonable to use a topical antibiotic
in the presence of measurable corneal epithelial defects or penetrating
corneal lesions, but antibiotic prophylaxis may not be necessary
when dealing with an intact epithelial surface or minimal punctate
epitheliopathy. Indeed, potential unwanted consequences of topical antibiotics
include toxicity, colonization by nonsusceptible organisms, and
the theoretic emergence of resistant species. No specific agent exists that uniquely qualifies for topical antibacterial
prophylaxis. The aminoglycoside agents are lacking in the gram-positive
spectrum. The newer quinoline medications have a somewhat broader
spectrum than the aminoglycosides but are more effective against the
virulent gram-negative rods than against streptococci and other gram-positive
cocci. The present ophthalmic formulation of ciprofloxacin is
not compatible with hydrogel lenses and leads to dense deposits on the
lens surface. The newer quinoline formulations are more compatible with
therapeutic lenses and may be the agents of choice when the risk-to-benefit
ratio favors antibiotic use in the presence of a bandage contact
lens. An alternative agent for broad-spectrum antibacterial prophylaxis
might be a polymyxin-sulfa combination drop. AVOIDING BANDAGE LENSES As beneficial as therapeutic lenses may be, their use does entail additional
patient expense, as well as potentially serious complications demanding
greater physician responsibility. If at all possible, the use
of therapeutic hydrogel lenses should be avoided in patients who are unreliable, have
severe keratoconjunctivitis sicca, or have markedly diminished
corneal sensation. In a significant number of cases, the use
of bandage lenses may be avoided by substituting other more appropriate
forms of treatment. Many persistent epithelial defects may be persuaded to heal by merely decreasing
or discontinuing unnecessary topical medications. A common example
is the need to decrease rather than increase topical antiviral
medication after prolonged treatment of herpes simplex keratitis. Systemic
medications may be substituted for topical medications when surface
toxicity is suspected (e.g., oral acyclovir may be substituted for topical trifluridine when antiviral
coverage is needed but surface toxicity is present). Ophthalmic ointments, frequently
formulated without preservatives, may be substituted
for equivalent preserved aqueous solutions. It is desirable, if possible, to identify and treat the responsible pathophysiologic
factor rather than obtain instant and temporary improvement
from the continued use of the bandage lens. Thus, punctal occlusion, often
using removable plugs, may minimize the effect of dry eyes on
an ocular surface problem. Defective eyelid closure and defective eyelid
position are frequent causes of corneal epitheliopathy for which oculoplastic
repair is more appropriate than prolonged bandage contact
lens wear. Therapeutic hydrogel lenses are more helpful in the short to medium term
than in the long term. This relates to both the patient and clinician
resources necessary for continued use, as well as the cumulative increased
probability of lens-associated complications over time. Thus, self-limited
conditions such as a postoperative “rough surface” are
much more appropriate for bandage lens therapy than a chronic
ocular surface condition without a definite end point. ADDITIONAL MEASURES I have found that the loss of therapeutic lenses occurs frequently at night
during sleep. For that reason, we instruct our patients to wear an
eye shield at bedtime, which seems to increase lens retention. It is not uncommon for therapeutic lens wear to be indicated in the presence
of marked blepharophimosis or lateral tarsorrhaphy. In such cases, insertion
may be difficult or impossible through the narrowed interpalpebral
fissure with the use of customary insertion techniques. Sliding
the contact lens superiorly with a sterile applicator in an anesthetized
eye will usually overcome this problem (see Fig. 1). |