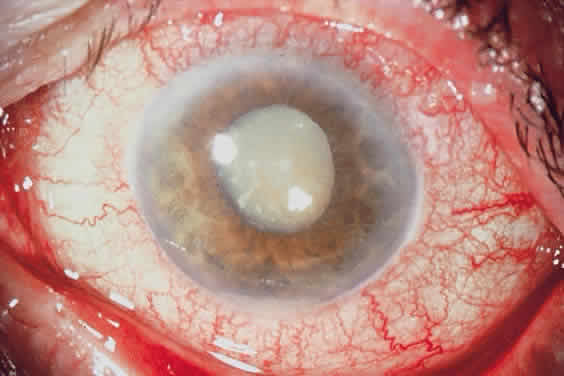
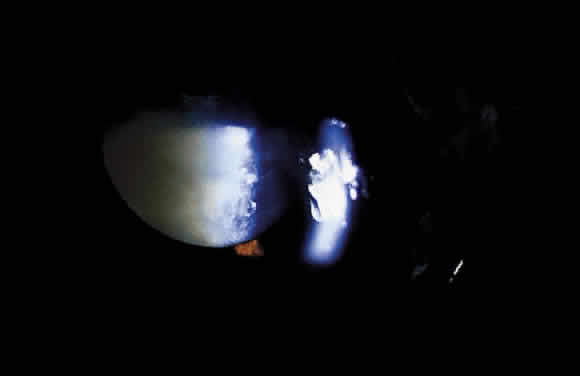
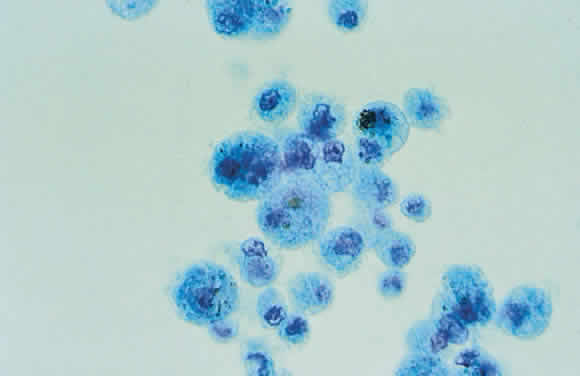
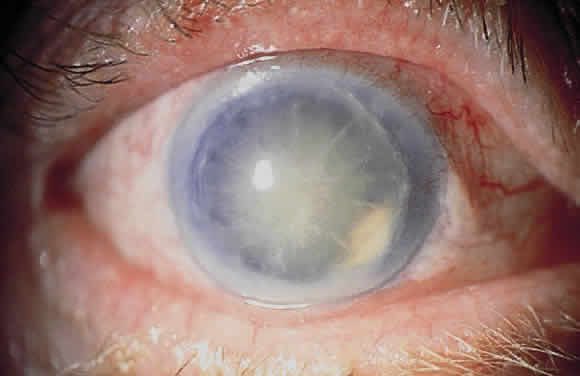
1. Khalil MK, Lorenzetti DW: Lens-induced inflammations. Can J Ophthalmol 21:96, 1986 2. McMahon MS, Weiss JS, Riedel KG, Albert DM: Clinically unsuspected phacoanaphylaxis after extracapsular cataract extraction
with intraocular lens implantiation. Br J Ophthalmol 69:836, 1985 3. Smith ME, Zimmerman LE: Contusive angle recession in phacolytic glaucoma. Arch Ophthalmol 74:799, 1965 4. Rodman HI: Chronic open-angle glaucoma associated with traumatic dislocation of the
lens. Arch Ophthalmol 69: 445, 1963 5. Flocks M, Littwin CS, Zimmerman LE: Phacolytic glaucoma. Arch Ophthalmol 54:37, 1955 6. Dhar GL, Bagotra S, Bhalla A: Lens-induced glaucoma: A clinical study. Indian J Ophthalmol 32:456, 1984 7. Bartholomew RS, Rebello PF: Calcium oxalate crystals in the aqueous. Am J Ophthalmol 88:1026, 1979 8. Brooks AMV, Grant G, Gillies WE: Comparison of specular microscopy and examination of aspirate in phacolytic
glaucoma. Ophthalmology 97:85, 1990 9. Thomas R, Braganza A, George T, Mermoud A: Vitreous opacities in phacolytic glaucoma. Ophthalmic Surg Lasers 27:839, 1996 10. Friberg TR: Retinal perivasculitis in phacolytic glaucoma. Am J Ophthalmol 91:761, 1981 11. Epstein DL: Lens-induced open-angle glaucoma. In Ritch R, Shields MB (eds): The
Secondary Glaucomas, p 121. St. Louis, CV Mosby, 1982 12. Goldberg MF: Cytological diagnosis of phacolytic glaucoma utilizing Millipore filtration
of the aqueous. Br J Ophthalmol 51:847, 1967 13. Lane SS, Kopietz LA, Lindquist TD, Leavenworth N: Treatment of phacolytic glaucoma with extracapsular cataract extraction. Ophthalmology 95:749, 1988 14. Hubbersty FS, Gourlay JS: Secondary glaucoma due to spontaneous rupture of the lens capsule. Br J Ophthalmol 37: 432, 1953 15. Epstein DL, Jedziniak JA, Grant WM: Obstruction of aqueous outflow by lens particles and heavy-molecular-weight
soluble lens proteins. Invest Ophthalmol Vis Sci 17:272, 1978 16. Epstein DL, Jedziniak JA, Grant WM: Identification of heavy-molecular-weight soluble protein in aqueous humor
in human phacolytic glaucoma. Invest Ophthalmol Vis Sci 17:398, 1978 17. Mansour AM: Anterior capsulorhexis in hypermature cataracts (letter). J Cataract Refract Surg 19:116, 1993 18. Mandal AK: Endocapsular surgery and capsular bag fixation of intraocular lenses in
phacolytic glaucoma. J Cataract Refract Surg 22:288, 1996 19. McKibbin M, Gupta A, Atkins AD: Cataract extraction and intraocular lens implantation in eyes with phacomorphic
or phacolytic glaucoma. J Cataract Refract Surg 22:633, 1996 20. Epstein DL: Diagnosis and management of lens-induced glaucoma. Ophthalmology 89:227, 1982 21. Forstot SL, Price PK, Hovland KR, Keyser RB: Phacolytic glaucoma after
extracapsular cataract extraction. Glaucoma 1983;5:206, 1983 22. Fastenberg DM, Schwartz PL, Shakin JL, Golub BM: Management of dislocated nuclear fragments after phacoemulsification. Am J Ophthalmol 112:535, 1991 23. Blodi BA, Flynn HW, Blodi CF et al: Retained nuclei after cataract surgery. Ophthalmology 99:41, 1992 24. Lambrou FH, Stewart MW: Management of dislocated lens fragments during phacoemulsification. Ophthalmology 99: 1260, 1992 25. Gilliland GD, Hutton WL, Fuller DG: Retained intravitreal lens fragments after cataract surgery. Ophthalmology 99: 1263, 1992 26. Kim JE, Flynn HW, Smiddy WE et al: Retained lens fragments after phacoemulsification. Ophthalmology 101:1827, 1994 27. Terasaki H, Miyake Y, Miyake K: Visual outcome after management of a posteriorly dislocated lens nucleus
during phacoemulsification. J Cataract Refract Surg 23:1399, 1997 28. Topping TM: Discussion [of Management of dislocated lens fragments during phacoemulsification]. Ophthalmology 99:1268, 1992 29. Marak GE: Phacoanaphylactic endophthalmitis. Surv Ophthalmol 36:325, 1992 30. Lam S, Tessler HH: Hypersensitivity uveitis. In Tasman W, Jaeger EA (eds): Duane's
Clinical Ophthalmology, Vol 4 p 2. Philadelphia, Lippincott-Raven, 1994 31. Friedlander MH, Allansmith MR: Ocular manifestations of immune disease. In
Garner A, Klintworth GK (eds): Pathobiology of Ocular Disease, p 174. New
York, Marcel Dekker, 1982 32. Riise P: Endophthalmitis phacoanaphylactica. Am J Ophthalmol 60:911, 1965 33. Smith RE, Weiner P: Unusual presentation of phacoanaphylaxis following phacoemulsification. Ophthalmic Surg 7:65, 1976 34. Abrahams IW: Diagnosis and surgical management of phacoanaphylactic uveitis following
extracapsular cataract extraction with intraocular lens implantation. Am Intraocular Implant Soc J 11:444, 1985 35. Thach AB, Marak GE, McLean IW, Green WR: Phacoanaphylactic endophthalmitis: A clinicopathologic review. Int Ophthalmol 15:271, 1991 36. Croxatto JO, Lombardi A, Malbran ES: Inflamed eye in Marfan's syndrome with posteriorly luxated lens. Ophthalmologica 193:23, 1986 37. Ishikawa Y, Kawata K, Ishikawa Y: Three cases of endophthalmitis phacoanaphylactica in the fellow eye after
extracapsular lens extraction. Folia Ophthalmol Jpn 28:1260, 1977 38. Muller H: Phacolytic glaucoma and phacogenic ophthalmia. Trans Ophthalmol Soc UK 83:689, 1963 39. deVeer JA: Bilateral endophthalmitis phacoanaphylactica. Arch Ophthalmol 49:607, 1953 40. Hogan MJ, Zimmerman LE (eds): Ophthalmic Pathology 2nd ed, p 153. Philadelphia, WB
Saunders, 1962 41. Apple DJ, Mamalis N, Steinmetz RL et al: Phacoanaphylactic endophthalmitis associated with extracapsular cataract
extraction and posterior chamber intraocular lens. Arch Ophthalmol 102:1528, 1984 42. Fox GM, Joondeph BC, Flynn HW et al: Delayed-onset pseudophakic endophthalmitis. Am J Ophthalmol 111:163, 1991 43. Rao NA, Nerenberg AV, Forster DJ: Torulopsis candida (Candida famata) endophthalmitis simulating Propionibacterium acnes syndrome. Arch Ophthalmol 109:1718, 1991 44. Winward KE, Pflugfelder SC, Flynn HW et al: Postoperative Propionibacterium endophthalmitis. Treatment strategies and long-term results. Ophthalmology 100:447, 1993 45. Aaberg TM, Rubsamen PE, Joondeph BC, Flynn HW: Chronic postoperative gram-negative endophthalmitis. Retina 17:260, 1997 46. Smith RE: Inflammation after cataract surgery. Am J Ophthalmol 102:788, 1986 47. Meisler DM, Zakov ZN, Bruner WE et al: Endophthalmitis associated with sequestered intraocular Propionibacterium acnes (letter). Am J Ophthalmol 104:428, 1987 48. Markowitz SN, Morin JD: The ratio of lens thickness to axial length for biometric standardization
in angle-closure glaucoma. Am J Ophthalmol 99:400, 1985 49. Appleton B, Lowrey A: Phacogenic glaucoma. Am J Ophthalmol 47:682, 1959 50. Al-Torbak A, Karcioglu ZE, Abboud E, Netland PA: Phacomorphic glaucoma associated with choroidal melanoma. Ophthalmic Surg Lasers 29:510, 1998 51. Tomey KF, Al-Rajhi AA: Neodymium:YAG laser iridotomy in the initial management of phacomorphic
glaucoma. Ophthalmology 99:660, 1992 52. Civerchia LL, Balent A: Intraocular lens implantation in acute angle closure glaucoma associated
with cataract. Am Intraocular Implant Soc J 11:171, 1985 53. Rao SK, Padmanabhan P: Capsulorhexis in eyes with phacomorphic glaucoma. J Cataract Refract Surg 24:882, 1998 54. Singh OS, Simmons RJ, Brockhurst RJ, Trempe CL: Nanophthalmos: a perspective on identification and therapy. Ophthalmology 89:1006, 1982 55. Ryan EA, Zwaan J, Chylak LT: Nanophthalmos with uveal effusion: Clinical and embryologic considerations. Ophthalmology 89:1013, 1982 56. Jin JC, Anderson DR: Laser and unsutured sclerotomy in nanophthalmos. Am J Ophthalmol 109:575, 1990 57. Brockhurst RJ: Cataract surgery in nanophthalmic eyes. Arch Ophthalmol 108:965, 1990 58. Kocak I, Altintas AGK, Yalvac IS et al: Treatment of glaucoma in young nanophthalmic patients. Int Ophthalmol 20:107, 1997 59. Akduman L, Adelberg DA, Del Priore LV: Nanophthalmic uveal effusion managed with scleral windows and topical mitomycin-C. Ophthalmic Surg Lasers 28:325, 1997 60. Nelson LB, Maumenee IH: Ectopia lentis. Surv Ophthalmol 27:143, 1982 61. Fuchs J, Rosenberg T: Congenital ectopia lentis. A Danish national study. Acta Ophthalmol Scand 76:20, 1998 62. Jarrett WH: Dislocation of the lens: A study of 166 hospitalized cases. Arch Ophthalmol 78:289, 1967 63. Peyman GA, Reichand M, Goldberg MF, Ritacca D: Management of subluxated and dislocated lenses with the vitrophage. Br J Ophthalmol 63:771, 1979 64. Demeler U, Sautter H: Surgery in (sub-) luxated lenses in adults. Dev Ophthalmol 11:162, 1985 65. Cross HE, Jensen AD: Ocular manifestations in the Marfan syndrome and homocystinuria. Am J Ophthalmol 75:405, 1973 66. Maumenee IH: The eye in the Marfan syndrome. Trans Am Ophthalmol Soc 79:684, 1981 67. Cruysberg JR, Boers GH, Trijbels JM, Deutman AF: Delay in diagnosis of homocystinuria: A retrospective study of consecutive
patients. Br Med J 313:1037, 1996 68. Willi M, Kut L, Cotlier E: Pupillary-block glaucoma in the Marchesani syndrome
Arch Ophthalmol 90:504, 1973 69. Jensen AD, Cross HE, Paton D: Ocular complications in the Weill-Marchesani syndrome. Am J Ophthalmol 77:261, 1974 70. Johnson GJ, Bosanquet RC: Spherophakia in a Newfoundland family: 8 years experience. Can J Ophthalmol 18:159, 1983 71. Ritch R: Glaucoma secondary to lens intumescence and dislocation. In Ritch
R, Shields MB: The Secondary Glaucomas, p 131. St. Louis, CV Mosby, 1982 72. Schlote T, Volker M, Thanos S, Thiel HJ: Glaucoma in Marfan's syndrome: Position dependent measurement of intraocular
pressure as a diagnostic criterion. Klin Monatsbl Augenheilkd 207:386, 1995 73. Ritch R, Wand M: Treatment of the Weill-Marchesani syndrome. Ann Ophthalmol 13:665, 1981 74. Wright KW, Chrousos GA: Weill-Marchesani syndrome with bilateral angle-closure glaucoma. J Pediatr Ophthalmol Strabismus 22:129, 1985 75. Salehpour O, Lavy T, Leonard J, Taylor D: The surgical management of nontraumatic ectopic lenses. J Pediatr Ophthalmol Strabismus 33:8, 1996 76. Koenig SB, Mieler WF: Management of ectopia lentis in a family with Marfan syndrome. Arch Ophthalmol 114:1058, 1996 77. Halpert M, Ben Ezra D: Surgery of the hereditary subluxated lens in children. Ophthalmology 103:681, 1996 78. Omulecki W, Nawrocki J, Palenga-Pydyn D, Sempinska-Szewczyk J: Pars plana vitrectomy, lensectomy or extraction in transscleral intraocular
lens fixation for the management of dislocated lenses in a family
with Marfan's syndrome. Ophthalmic Surg Lasers 29:375, 1998 |