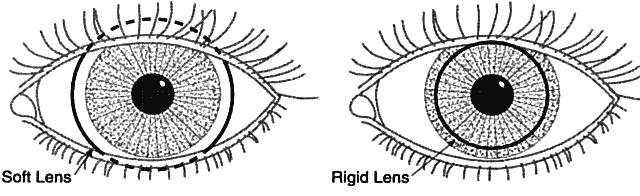
| Loose lens is identified by (1) excess movement with eye rotation or blink; (2) poor
centration; (3) variable vision, especially after a blink; and (4) discomfort. The
treatment for a loose lens is a steeper base
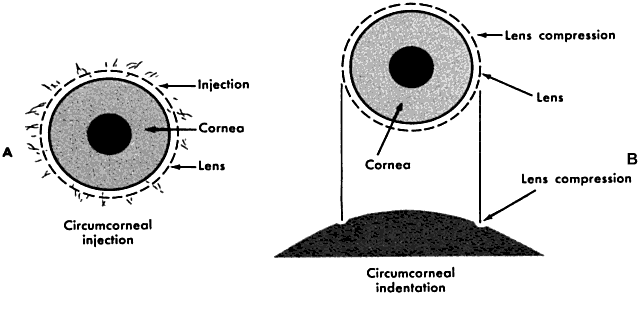
curve or larger lens diameter. Tight lens is identified by (1) fluctuating vision that clears after a
blink; (2) circumcorneal compression; (3) minimal movement after a blink; (4) circumcorneal
indentation; and (5) difficulty removing the lens. The
treatment for a tight lens is a flatter base curve in a lathe-cut
lens; with spin-cast lenses, switch to thinner or a smaller-diameter
series. When fitting soft lenses it is best to fit from the flatter side. Soft
lenses may tighten with time as a result of either dehydration or protein
accumulation on their surfaces. Spin-cast lenses have an aspheric posterior surface. Therefore, K readings
are not as useful as with lathe-cut lenses. A better indication of
lens selection can be based on the measurement of the horizontal visible
iris diameter. The fit of the lens should be assessed 20 minutes after
insertion because the base curve can steepen after 5 to 10 minutes
of wear. LENS CLASSIFICATION SYSTEMS Soft contact lenses are classified in several ways. The most common classification
is based on water content. Lenses are said to be of low water content when they contain 37.5% to 45% water; these
lenses are usually suitable for daily wear. Lenses that
are of medium water content contain 46% to 58% water; they are suitable
for either daily or extended wear. Lenses with a high water content
consist of 59% to 79% water and are primarily used for extended wear. Soft lens materials have been classified by the US Food and Drug Administration (FDA) into
four groups based on water content and ionicity. The
purpose of the FDA classification system is to evaluate the safety
and efficacy of the various disinfection systems and solutions with different
preservatives. Another classification system is based on the process used to manufacture the lens. The first soft contact lenses were produced
by spin casting. Bausch & Lomb adopted and perfected the technology and still produces
many lenses this way. In spin casting, the liquid lens material is
dropped into a small mold that spins at various controlled rates of speed. The
shape of the mold and the rate of spin determine the final characteristics
of the lens. Lathe cutting, developed later, involves cutting solid polymer rods to form lenses. The
control available in lathe cutting makes possible the production of
complex lens types, and lathe cut lenses are available in a range of
designs from single-cut to toric and lenticular configurations. A method
of cast molding, is now used by some manufacturers because it provides
lenses with smooth surfaces, free of the marks that may be produced
in the lathe-cutting process. Finally, lenses are classified by their design (e.g., spheric, toric) or function (e.g., daily wear, extended wear). This classification describes lenses in such
terms as daily wear spheric, extended wear spheric, daily or extended
wear toric, bifocal, or therapeutic. Of course, different lens classification
schemes can be combined; thus it is possible, for example, to
describe a particular lens as being a low water content, lathe cut, daily
wear spheric lens. In the following sections some of the major
categories are examined. Daily Wear Spheric Soft Contact Lenses Daily wear spheric soft contact lenses can correct up to 1.25 D of astigmatism. They
are manufactured in a wide range of diameters to allow the
fitter to optimize alignment and centration and to accommodate varying
lid apertures and sensitivities. The center thicknesses available
range from 0.4 to 1 mm. (Center thickness is important to both patients
and fitters. Thinner lenses are more comfortable and allow for greater
oxygen transmission; thicker lenses provide better correction of astigmatism
and are easier for the patient to handle.) Spin-cast, lathe-cut, and
cast-molded lenses are available to meet the fitting characteristics
required. Daily wear spheric lenses are available in tints that enhance eye color
and facilitate handling. Daily wear spheric soft lenses are ideal for
patients who wear their lenses on a limited basis, such as for sports
or social activities. Extended Wear Spheric Soft Contact Lenses Extended wear spheric lenses are available in low-, medium-, and high-water-content
models. All other things being equal, the higher the water
content the greater the oxygen permeability of the lens. Most of the
extended wear soft contact lenses fitted today are of relatively high
water content. As with daily wear lenses, extended wear lenses are available
in tints and may be spin cast, molded, or lathe cut. In general, extended
wear soft contact lenses of spheric design will not correct
more than 0.5 D of astigmatism. Single vision spheric soft contact lenses may be the correction mode of
choice for presbyopic patients who want extended wear, because there
are no extended wear bifocal soft contact lenses on the market yet. Presbyopes
who will not tolerate eyeglasses for reading but who still want
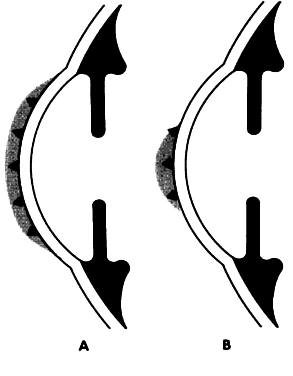
extended wear may be best served by a monovision fit. Bifocal Soft Contact Lenses When discussing bifocal lenses, visual design available as lens design
must be considered. Two bifocal visual designs are available: alternating vision and simultaneous vision. In alternating vision, light that reaches the patient's retina passes
through either the near vision or the distance vision portion of
the lens, depending on the direction of the patient's gaze. In distance
gaze, the patient views through the upper distance portion of the
lens. In near gaze, the lens translates (moves) up so that the viewing
is through the lower part (near segment) of the lens (Fig. 9). In simultaneous vision designs, light reaches the retina from both the
distance and near vision portions of the lens at all times. Some of
the light entering the eye is focused for near and some is focused for
distance, and the two foci are formed simultaneously. The brain then
selects the image of regard. Aspheric and diffractive bifocals, while
technically simultaneous vision designs, are so different from standard
simultaneous vision lenses that they are often treated separately. 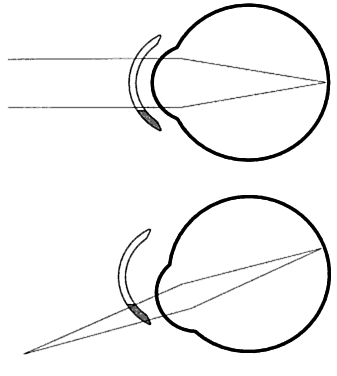 Fig. 9. Segmented contact lens showing positioning for viewing distant and near
objects. (Adapted from Stein HA, Slatt BJ, Stein RM: The Ophthalmic Assistant, 5th
ed. St. Louis, CV Mosby, 1988) Fig. 9. Segmented contact lens showing positioning for viewing distant and near
objects. (Adapted from Stein HA, Slatt BJ, Stein RM: The Ophthalmic Assistant, 5th
ed. St. Louis, CV Mosby, 1988)
|
A number of bifocal lens designs are available. Segmented lenses have two visual regions: the distance vision portion is in the top of
the lens, and the near vision portion is in the crescent-shaped segment
on the inferior portion of the lens (see Fig. 9). The key to success with segmented bifocal lenses is positioning. The
lens cannot be allowed to rotate, yet it must translate up and down in
order to position the segments. One way to prevent lens rotation is
to add weight to the inferior portion of the lens through insertion of
a wedge shape (prism ballasting) or thickening the lower edge (periballasting-ballasting). Truncation (Fig. 10) involves cutting off the lower portion of the lens; this creates “corners” that
hold tightly to the sclera. The flat edge also
helps maintain orientation by aligning with the lower lid. 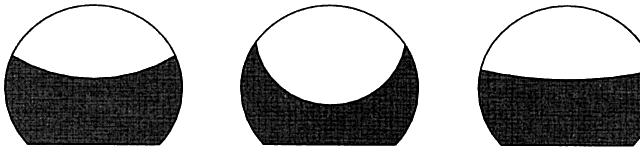 Fig. 10. Truncated contact lenses. The lenses pictured are segmented bifocals that
provide alternating vision. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 84. Thorofare, NJ, Slack, 1996) Fig. 10. Truncated contact lenses. The lenses pictured are segmented bifocals that
provide alternating vision. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 84. Thorofare, NJ, Slack, 1996)
|
The advantage of translating (segmented) bifocals is the quality of sight
they provide when properly fitted, giving good vision for both distance
and near. The main problem with segmented lenses is fitting them
to move properly on the eye. If the lens does not translate, it will not
function as a bifocal. Quick movement is required to prevent blurring. Tight
lids are needed to make the lens move properly. Obtaining a
good fit requires skill and patience. In annular (or concentric) lenses, the different portions of the lens are configured
in concentric circles or rings (annular means “ring shaped”). That
is, if the distance correction is in the center of the lens, the
near vision part of the lens forms a concentric ring around the
center
(Fig. 11). Alternatively, the distance correction may be located in the concentric
ring and the near correction located in the center. Unlike segmented
lenses, annular lenses are not affected by lens rotation problems. 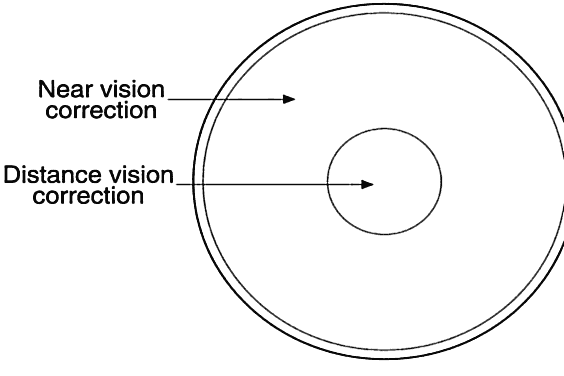 Fig. 11. Annular bifocal lens. In some annular designs, the near vision correction
is in the center of the lens; in other designs, distance correction
is in the center. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 85. Thorofare, NJ, Slack
Inc, 1996) Fig. 11. Annular bifocal lens. In some annular designs, the near vision correction
is in the center of the lens; in other designs, distance correction
is in the center. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 85. Thorofare, NJ, Slack
Inc, 1996)
|
The advantage of the annular design is that the lens does not have to translate, as
both foci are formed simultaneously. (Because lens orientation
and position are less critical than with segmented designs, annular
lenses are easier to fit.) A disadvantage of the design is that it
is dependent on pupil size. For example, in the center-near design, the
pupil will constrict in bright sunlight, so that only the center portion
of the lens is used; this effectively limits the lens to near vision. Another
disadvantage is that visual contrast is reduced, becasue
only a portion of light reaching the retina is in focus for each image. Finally, many
patients have difficulty with annular lenses because
they are unable to adjust to simultaneous vision. Multifocal (or aspheric) lenses have a gradually flattening (anterior or posterior) curve
from the center to the edge of the lens. The result is a progressively
increasing add power from the center to the periphery (Fig. 12). In effect, the multifocal lens is a concentric design in which there
is no sharp zone of transition between the near and distance portion
of the lens. 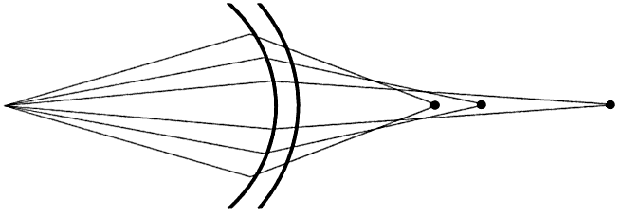 Fig. 12. Multifocal lens with distance correction in the center and near correction
at the periphery. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1990) Fig. 12. Multifocal lens with distance correction in the center and near correction
at the periphery. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1990)
|
Bifocal designs are either anterior or posterior, depending on manufacturer
and type. Anterior designs have the power manufactured into the front surface of the lens; posterior designs have the power manufactured into the back surface. An alternative for the correction of presbyopia is monovision. Monovision techniques employ either one or two single-vision lenses to
correct near and distance vision separately. That is, one eye is corrected
for near vision, and the fellow eye (with or without correction) is
used for distance. Because there are no lenses designed specifically
for monovision, standard single vision lenses are used. With bifocal soft contact lenses, the in-office verification of lens power
and lens design is often difficult. In slit-lamp examination, multifocal
lenses appear as spheric single-vision lenses. A few bifocal lenses
have features that allow them to be distinguished. Toric Soft Contact Lenses There are a wide variety of toric soft contact lenses on the market, and
fitters must familiarize themselves with what is available on a lens-by-lens
basis. In the following paragraphs, specific lenses are not described
because there are excellent manufacturers' fitting guides available; however, a
word of caution is offered: before choosing a specific
lens for fitting, the fitter must make sure that the lens is available
in parameters to match the patient's refractive error. Some “off-the-shelf” toric lenses are available, but only in
a limited range of cylinders and axes. Manufacturers make and stock
lenses in the most commonly requested power ranges, and these are available
immediately on request. Off-the-shelf lenses are available in powers
from -6 D to + 4 D, with cylinder powers of -0.75 D, -1 D, -1.25 D, -1.75 D, and -2 D. (Lenses with -3 D of cylinder are available, but
usually in a more limited axis range, generally 20° on either
side of the 90° or 180° meridians.) Limited fitting sets are
available. A diagnostic lens of the selected toric design must be evaluated
on the patient's eye for fit and position of axis. For custom work, one must also use a fitting set. The fitting lenses are
spheric designs, but with the diameters and orientation systems of the
toric lenses the patient will eventually wear. Toric lenses that can
be fit in this manner are available in powers from -20 D to + 20 D, with
axes of rotation available in 5° increments. A few manufacturers
provide made-to-order toric lenses with even greater powers and
cylinders and with axes of rotation in 1° increments. There are several design alternatives used to maintain the orientation
of toric lenses. Double slab off, the creation of thin zones on the inferior and superior parts of the lens, allowing
the eyelids to hold the lens in position (Fig. 13). As with bifocal soft contact lenses, prism ballasting
(Fig. 14), periballasting, and truncation are also used to maintain orientation. 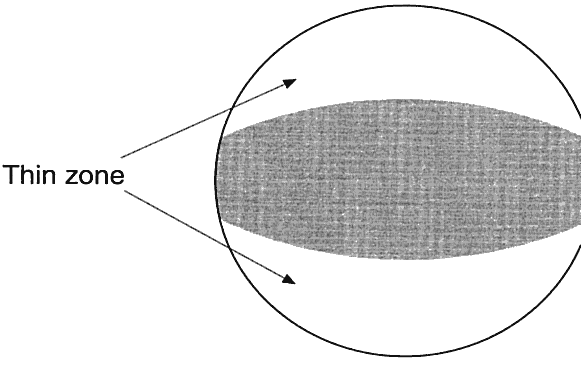 Fig. 13. Double slab-off lens with thin zones inferiorly and superiorly that cause
the lens to rotate to the proper position and then remain in place. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 88. Thorofare, NJ, Slack, 1996) Fig. 13. Double slab-off lens with thin zones inferiorly and superiorly that cause
the lens to rotate to the proper position and then remain in place. (Stein HA, Freeman MI, Stein RM, Maund LD (eds): Basics of Soft Contact
Lenses. In: Contact lenses: Fundamentals and Clinical Use, p 88. Thorofare, NJ, Slack, 1996)
|
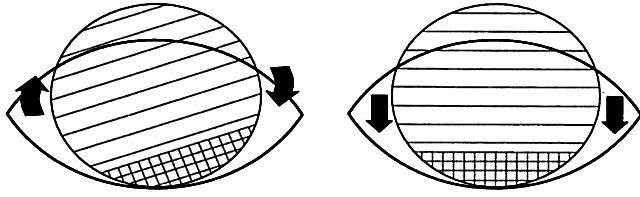 Fig. 14. Lens orientation with prism ballasting. The weighted portion of the lens
rotates to the inferior position. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1994) Fig. 14. Lens orientation with prism ballasting. The weighted portion of the lens
rotates to the inferior position. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1994)
|
Toric lenses have orientation (or scrib) marks near the lens edge: some at the 3- and 9-o'clock positions, and others
at the 6-o'clock position. The manufacturer is sometimes identified by
these marks (or by laser marks on the lens). Therapeutic Soft Contact Lenses Therapeutic soft contact lenses have many uses and are made from a variety
of materials. In most cases, this kind of lens is used as a short-term
corneal “bandage” to protect the eye during treatment
for nonhealing corneal ulcers or recurrent erosions. Occasionally, therapeutic
lenses are used to cover small corneal lacerations that do not
require suturing. In this case, extended wear lenses must be used. Therapeutic lenses are generally of low to medium water content; however, lenses
of higher water content, although not designed specifically
as therapeutic lenses, are sometimes worn by patients with bullous keratopathy. Tinted
lenses (usually of low to medium water content) can be
used to improve cosmesis when the cornea is scarred. The use of therapeutic contact lenses is contraindicated in patients with
corneal exposure and dryness, because in these situations therapeutic
lenses may simply aggravate the problem. It is mandatory that therapeutic-lens
patients be carefully selected and monitored. Aphakic Soft Contact Lenses Soft contact lenses, particularly extended wear lenses, can be a great
help for aphakic patients who do not achieve correction with intraocular
lenses. Aphakic soft contact lenses are available in spin-cast and
lathe-cut designs and with medium and high water contents. The lathe-cut
lenticular designs have the best stability on the eye and provide the
sharpest and most stable vision (Fig. 15).  Fig. 15. Regular (left) and aphakic (right) soft lenses. The lens at right is a lenticular design. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1990) Fig. 15. Regular (left) and aphakic (right) soft lenses. The lens at right is a lenticular design. (Adapted from Stein HA, Slatt BJ, Stein RM: Fitting Guide for Rigid and
Soft Contact Lenses, 3rd ed. St. Louis, CV Mosby, 1990)
|
As with therapeutic soft contact lens patients, aphakic patients must be
carefully fitted and followed. Aphakic patients (who tend to be elderly) have
an above-average incidence of microbial corneal ulcers associated
with contact lens wear. With careful patient selection and fitting, however, it
is possible to rehabilitate an aphakic patient quickly
and without further surgery. Soft Contact Lens Candidates: Patient Selection Soft lenses are the contact lenses of choice for the following classes
of patients: - Rigid lens dropouts: These are patients who are unable or unwilling to tolerate a rigid lens
adaptation and wearing schedule, as well as those with rigid lens overwear
reactions or rigid lens—induced irritation.
- Intermittent lens wearers: These are patients such as actors or athletes who may want to wear contact
lenses on an occasional basis only.
- Workers in occupations where losing a lens must be minimized: Rigid lenses are much more likely to fall off the eye than soft lenses.
- Aphakic patients not fitted with intraocular lenses.
- Elderly persons: These patients may have difficulty with various aspects of rigid lens
wear.
- Patients requiring relatively small amounts of correction (i.e., correction less than 1.50 D).
Extended Wear Soft Contact Lenses Extended wear soft contact lenses for the correction of myopia were first
approved by the FDA in 1980. Since then, their popularity has increased, but
so has the awareness of problems associated with extended wear. The
fitting of extended wear lenses is not difficult; however, it
requires a basic understanding of the physiologic needs of the cornea
and the potential complications of extended wear. The cornea adapts in different ways to different types of contact lenses. For
example, the movement of non—gas-permeable PMMA contact lenses
that occurs on blinking promotes tear exchange beneath the lens, bringing
dissolved oxygen to the corneal surface. (PMMA lenses are also
small in diameter, which minimizes the area affected.) Despite tear
exchange under the lens, overwear of PMMA lenses will produce corneal
edema. Rigid gas-permeable and hydrogel lenses attempt to solve the problem
by allowing gases to pass directly through the lens to the cornea. Movement
of these lenses helps to bring nutrients to and remove waste
products from the corneal epithelium. At night, when the eyes are closed for sleep, the cornea gets the oxygen
it needs by diffusion from capillary blood vessels in the palpebral
conjunctiva. However, the oxygen available from the conjunctival capillaries
is limited and just barely sufficient to meet corneal needs. Gas-permeable (hard
and soft) contact lenses that allow an adequate supply
of oxygen to the cornea when the eyes are open may not provide an adequate
oxygen supply when the eyes are closed, and the amount of oxygen
available is greatly reduced. The major challenge of extended wear
is creating lenses that allow sufficient gas transfer during the closed-eye
state. Other challenges of extended wear include producing a lens (1) with adequate
movement to promote tear exchange and removal of debris; and (2) that
resists spoilage from deposit formation. What allows some hydrogel contact lenses to be worn on an extended wear
basis is primarily their high degree of oxygen transmissibility (i.e., their ability to let oxygen pass through to the cornea). Enough oxygen
must pass through the lens to allow normal or near-normal corneal metabolic
activity, even with the eyes closed during sleep. In addition, adequate
lens movement is required to remove metabolic waste products
from the corneal surface. Most daily wear soft contact lenses do not supply enough oxygen to the
cornea to allow them to be used for extended wear. However, modifications
of the lens polymer or the lens design can improve the lens' oxygen
transmission characteristics. Specifically, oxygen transmission through
a soft contact lens may be increased by: - Increasing the water content of the lens: As lens hydration increases, so does the lens' permeability to oxygen.
- Markedly thinning the lens: As lens thickness increases, oxygen transmission decreases. Conversely, thinning
the lens creates an increase in oxygen transmission.
- Thinning the lens and moderately increasing the water content (i.e., a combination of the above techniques).
Based on the means used to enhance oxygen transmission, hydrogel soft contact
lenses for extended wear can be divided into three types: - Very high water content: Lenses with 70% to 79% hydration (e.g., Permalens, Permaflex)
- Ultra thin, low water content: Lenses with 38% to 45% hydration (e.g., Soflens O3/O4, CSI)
- Thin, medium water content: Lenses with 46% to 58% hydration (e.g., Acuvue, NewVues, Softcon, Durasoft 3)
In short, the higher the water content and the thinner the lens, the greater
the gas exchange through the polymer. One must consider that lens
thickness is usually measured in the center of the lens. In a high minus
power lens (thin in the center and thick at the edge) the edge thickness
may be such that oxygen transmission is considerably decreased, causing
corneal edema at the periphery. Conversely, central corneal
edema may occur if insufficient oxygen is reaching the cornea through
the thick center of a high plus lens. (This is a consideration in fitting
the high plus power lenses needed by aphakic patients.) LENS SELECTION. Each of the various extended wear soft contact lens types has certain
advantages and drawbacks. Lenses with a high water content tend to be
more comfortable, particularly when the eye is slightly dry; however, these
lenses are also more easily damaged than those with a low water
content, are more prone to deposit formation, and may offer less stable
vision. Ultra-thin lenses with a low water content present handling
problems and are susceptible to damage during care procedures. Thin (not ultra-thin) lenses with a medium (46% to 58%) water content offer
something of a compromise: more durability and more stable vision
than high-water-content lenses, as well as a lower tendency toward deposit
formation. Medium-water-content lenses are durable enough to be
used for daily wear. PATIENT SELECTION. Success in fitting extended wear soft contact lenses is a function of
the proper screening of prospective lens wearers. General health, anterior
segment health, tear function, hygienic habits, motivation, visual
requirements, manual dexterity, and financial commitment represent a
partial list of patient selection criteria. Patients considering extended
wear must understand and accept the fact that this mode of lens wear
carries considerable added risk of complications. Because extended wear is associated with a greater risk of infection and
other serious ocular conditions, extended wear patients must be seen
by a practitioner more frequently than daily wear patients. Therefore, before
fitting a patient with extended wear lenses, the practitioner
must assess the patient's willingness to keep follow-up appointments. Also
of great importance is the patient's willingness and ability
to remove the lens at the earliest sign of redness or ocular irritation. Before
the fitting procedure is performed, the patient must
be informed that failure to follow the practitioner's instructions
may lead to a serious compromise of ocular health. Realistic patient expectations with regard to lens wear are also important. The
patient must understand that extended wear does not mean the
ability to leave the lens in place on the eye for unlimited periods of
time. Rather, the eyes must be given regular “rest periods,” during
which they can resume their normal metabolic activity. Cost is an additional factor. Compared with most daily wear lenses, the
contact lenses used for extended wear are more prone to deposit formation
and to damage during handling. These factors shorten lens life and
increase cost to the patient. The greater frequency of practitioner
visits that is necessary with extended wear also increases the cost. While
the extended wear patient may spend less on lens care solutions than
the daily wear patient, the greater number of office visits and the
greater frequency of lens replacement combine to make extended wear
more costly than daily wear. In summary, the extended wear soft contact lens patient must: - Understand the risk
- Accept the added expense
- Be willing to comply with lens care regimens
- Have regular follow-up visits
- Be willing to remove the contact lens at the first sign of redness or ocular
irritation
The ideal candidate for extended wear is an experienced daily wear lens
patient who is already adept at insertion and removal techniques. If
the patient is a first-time lens wearer, it is recommended that there
be a daily wear adaptation period of at least 6 weeks. This promotes familiarity
with insertion, removal, and care procedures and decreases
the patient's fear of lens manipulation. Available data suggest that extended wear lenses should be worn for a maximum
of 6 days and removed on the seventh day to permit reestablishment
of normal aerobic metabolism. Each patient must be evaluated on an
individual basis to determine the appropriate wearing schedule. The lens
care regimen should be compatible with the lens type being dispensed (e.g., heat disinfection may be damaging to high-water-content lenses). The extended wear contact lens should remain off the eye for at least overnight
before reinsertion. Planned lens replacement every 3 months is
recommended as a form of “preventive maintenance” that tends
to decrease complications associated with extended wear lenses. Disposable
extended wear lenses are available for weekly replacement. The critical need for hand washing and proper lens hygiene should be emphasized. In
addition, one must communicate to the patient the need for
immediate lens removal in the event of ocular redness or pain. The large
number of reported infections (particularly those involving Pseudomonas aeruginosa) among extended wear patients compel the practitioner to counsel the patient
in strict lens care procedures. PROBLEMS. Recent studies suggest that no currently available extended wear soft
contact lens provides adequate oxygen transmission for continued normal
aerobic corneal epithelial metabolism. As a result, one sometimes sees
in extended wear soft contact lens patients (1) microcysts and (2) changes
in the appearance of the endothelium (morphometric changes). The
long-term significance of these effects is unknown, and further research
into the effects of soft contact lens extended wear is warranted. Any
of the following findings is cause for immediate concern: - Acute hypoxic episodes
- Microcysts of the epithelium
- Giant papillary conjunctivitis
- Neovascularization
Any of the these findings should immediately lead the practitioner to discontinue
the patient's extended wear routine and change the patient
to daily wear, which is something that can be done without refitting
or dispensing new lenses. IS THERE REALLY EXTRA RISK IN REGULAR EXTENDED WEAR? The answer to this question is a clear-cut “yes.” In a study
of the safety of extended wear sponsored by the Contact Lens Institute, that
was published in the New England Journal of Medicine in 1989, Poggio and coworkers found the following rates of ulcerative
keratitis in their very large sample group: Extended wear: 20.9 per 10,000
Daily wear: 4.1 per 10,000
Thus the rate of this serious complication was five times greater with
extended wear. This doesn't mean that extended wear is not an acceptable mode of lens
use. It does mean that great care must be taken in choosing suitable candidates
for extended wear, and then educating patients to minimize its
dangers. Patients who choose extended wear should do so knowing the
added risk. APHAKIC EXTENDED WEAR SOFT CONTACT LENSES. Aphakic patients without intraocular lenses require strong plus power
lenses for visual functioning. Although aphakic eyeglasses are available, the
quality of the vision these provide is limited and often unsatisfactory
for tasks that require good peripheral vision, such as driving. A
number of extended wear soft contact lenses are available in high
plus powers for aphakic patients. Because the aphakic population is largely elderly, particular care must
be given to patient selection for extended wear. Elderly patients may
not have the manual dexterity to remove, clean, disinfect, and reinsert
their lenses. Before fitting such patients, it must be ascertained
that if the patient cannot perform care routines alone, there is someone
else at hand to render assistance. Even more important, removal of
extended wear contact lenses at the very first sign of trouble can prevent (or
greatly minimize) subsequent problems. If the patient cannot
do this (and assistance is not readily available), the risk of serious
ocular complications is greatly increased. Diminished tear function is more likely to be present in an elderly patient, as
are disorders of the corneal epithelium. Aphakic patients should
be evaluated for these conditions before being fitted with extended
wear contact lenses. Flexible Wear Because of the questions that have been raised about extended wear of contact
lenses, some practitioners have decided to stop recommending extended
wear to their patients and will prescribe only daily wear lenses. A
middle ground exists, however, between extended wear and daily wear. This
is the concept of flexible wear, which refers to the combination of daily wear with occasional or intermittent
extended wear. An example of a flexible wear schedule is 5 days
of daily wear followed by 1 day of extended wear. The lenses are cleaned
and disinfected every time they are removed. Contact lenses to be worn on a flexible wear schedule must be approved
for extended wear. However, the more fragile extended wear lenses should
be avoided in flexible wear because of the increased handling they
will receive. Rigid gaspermeable extended wear lenses or thin, medium-water-content
soft lenses (as opposed to ultra-thin or high-water-content
soft lenses) are potentially suited to flexible wear. It must be emphasized that contact lenses that are not approved for extended
wear must be used only on a daily wear basis. Extended wear lenses
can be used for daily wear, with the caveat that because of their fragility, some
extended wear lenses may not stand up to the increased
handling in daily wear. Disposable/Frequent Replacement Lenses A new era is at hand with the development of disposable extended wear contact
lenses. Lenses such as the Vistakon Acuvue, Bausch & Lomb SeeQuence, CIBA
Vision New Vues, Ocular Sciences/American Hydron Biomedics, and
Wesley-Jessen Fresh Look have been positioned in the market as
lenses that can be worn on an extended wear schedule for 1 week and
then thrown away. (Alternatively, these lenses may be worn on a daily
wear basis for 2 weeks and then discarded.) Recently true daily-wear disposable
lenses such as Vistakon's 1-Day Acuvue and Bausch & Lomb's
New Day have been introduced. These lenses are designed to
be worn for 1 day. They are discarded at the end of the day. It is important
that routine screening procedures be applied to candidates for
disposable lenses. Ideally, these patients should be monitored as daily
wear patients for up to 6 weeks and then monitored for potential extended
wear over a 6-month period. Advantages are as follows: - Much less needs to be spent on lens care solutions when the lenses are
used in an extended wear situation.
- There is minimal deposit formation. (In most cases, the lenses will be
discarded before significant deposits can form.)
- A sterile lens is always inserted (when used exclusively in extended wear).
- There is better patient compliance. (It is hoped that elimination of lens
care routines will end the dangerous short cuts that some patients
take out of ignorance or in order to save time or money.)
It must be stressed that any time a disposable lens is removed from the
eye with the intention of reinserting it, the lens must be disinfected. Some
practitioners recommend that disposable lenses be worn on a daily
wear basis; in this case, the lenses are removed and disinfected at
the end of each day. Disadvantages include the following: - The disposable system is more expensive than traditional extended wear (which
in turn costs more than traditional daily wear).
- Because of cost and convenience factors, patients may decide to wear lenses
beyond the recommended time period.
Finally, the disposable lenses in current use are not greatly different
from normal extended wear soft lenses. Although problems with solutions
and deposits may be helped by disposing of the lens after a relatively
brief wearing period, the basic physiologic questions about long-term
extended wear remain unanswered and will not be solved simply by disposability. Frequent/Planned Replacement Contact Lenses A growing proportion of soft lens patients are wearing their lenses on
a frequent-replacement basis. The aim of frequent replacement is to achieve
better vision with fewer contact lens—related complications. In
addition, frequent replacement allows practitioners increased patient
contact and better control of patient lens wear. (The terms planned replacement and programmed replacement are used interchangeably with frequent replacement.) Frequent replacement seeks to minimize the problems related to prolonged
wear of a soft contact lens. Many soft lens patients use the same pair
of contact lenses for a year or more. Among the problems related to
the long-term use of the same lenses are the following: - Changes in fit (e.g., lenses may tighten)
- Decreased visual acuity
- Increased lens awareness or discomfort
- Increased risk of infection
- Red eye
- Increased risk of corneal abrasion or insult from lens or lens edge
- Decreased oxygen permeability of lens and increased risk of corneal edema
Most of these problems are related to the formation of deposits on soft
lenses. Regular use of surfactant and enzymatic cleaners helps a great
deal, but does not fully eliminate deposits. In addition, deposit-related
complications may begin well before decreased visual acuity or discomfort
make the patient aware of the deposits. A major risk is the
development of ulcerative keratitis caused by P. aeruginosa. Deposits on soft lenses may act as a focus for the growth of this organism. Although deposits begin to form almost immediately on new soft contact
lenses, frequent replacement allows for the discarding of soft contact
lenses before deposit problems manifest. At the least, frequent replacement
should serve to reduce deposit-related complications. Frequent
replacement can be thought of as preventative care. Incorporating the frequent-replacement concept into contact lens fitting
requires the practitioner to make several choices. Decisions to be made
include whether to use frequent replacement with all or just a portion
of one's patients. Frequent-replacement lenses give the practitioner
some options: the practitioner can dispense at one time a full
year's supply of lenses or, alternatively, the patient can pick
up the lenses quarterly. Where frequent-replacement lenses are being considered, patient evaluation
is performed the way one would with any soft lens candidate. A complete
history and examination are required. If the patient is currently
a contact lens wearer, the lenses should be examined for deposits. Tear
film evaluation should be performed; this should include a break-up
test and Schirmer tests. Blink rate should be at least 12 per minute. Frequent-replacement lenses can be divided into two categories. The first
is disposable lenses, which are approved for continuous wear for up
to 6 days. Disposable lenses when used in this way require no lens care. This
assumes that a lens that is removed from the eye will be discarded
and a new, sterile lens inserted. The second category is programmed replacement of daily wear or flexible
wear lenses. This type of frequent-replacement lens must be cleaned with
a surfactant and disinfected every time the lens is to be reinserted
into the eye. (Enzyme cleaning may not be necessary for patients who
are not heavy depositors.) Programmed replacement may be the method
of choice for patients who tend to tear or lose lenses as well as those
who are not candidates for extended wear. Programmed replacement is
also often less expensive than disposable wear. One month is commonly
used as the replacement interval for programmed replacement lenses, but
intervals from 2 weeks to 6 months are also used. The importance of patient compliance with the lens replacement schedule
must be stressed when prescribing frequent-replacement lenses. Incentives
such as including lens cleaning and disinfection products with dispensed
cases may help increase compliance. Some practitioners have patients
sign an agreement that outlines the responsibilities of the both
patient and practitioner. The practitioner may reserve the right to
terminate the agreement in the case of patient noncompliance. |