DIPLOPIA
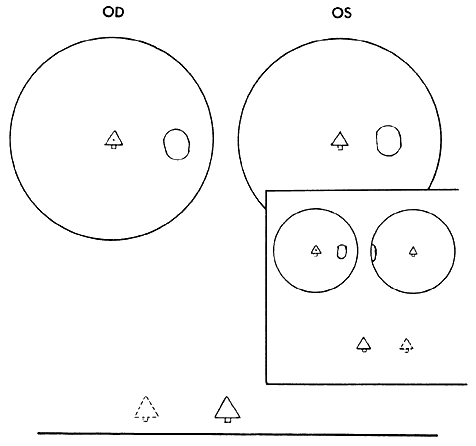
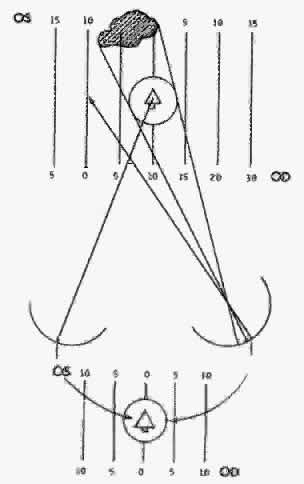
Diplopia results from the simultaneous perception of the images of the object of regard projecting onto noncorresponding retinal areas of the strabismic eyes. The simultaneous perception of the two similar images under these circumstances yields the impression that the object of regard is simultaneously located at two points in space (Fig. 1).
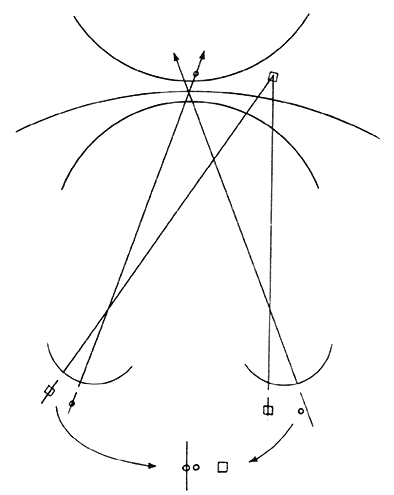
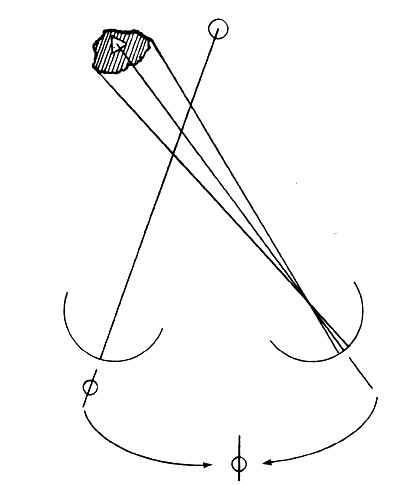
The size of the retinal area in one eye that corresponds to a retinal point in the other eye rapidly expands for retinal loci increasingly peripheral to the fovea. This determines the degree of retinal image disparity that either the macular or extramacular fusion reflex find tolerable. It accounts for Panum's visual space. Thereby, the depth of Panum's visual space is very limited for the macular fusion reflex to function within compared with the ever-expanding depth of fusional space within which the extramacular fusion reflex operates. This explains how an esotropic deviation may be sufficiently small to allow a spatial target peripheral to the object of regard (yet within Panum's fusional space) to project the peripheral target's images onto corresponding retinal areas while the object of regard projects its images onto noncorresponding macular areas (Fig. 2).
According to Ogle,3 foveal horizontal image disparity greater than 20 minutes of arc (2/3 of a prism diopter) is not fusible. From my clinical investigation it appears that extramacular image disparity is fusible up to approximately 480 minutes of arc4,5 (8 prism diopters). The vast differences in tolerance of horizontal image disparity between macular and extramacular fusion suggest they are under the control of separate binocular vision reflexes. This reasoning is strengthened by the frequency of clinical cases that are devoid of a macular fusion reflex but have a normal extramacular fusion reflex.
The purpose of the macular fusion reflex is to produce and maintain bifixation. If the reflex is absent or intermittently shut down for any reason, the patient monofixates. Monofixation is manifest by a three-degree facultative macular scotoma present only in the nonfixating eye during binocular viewing while the intact normal extramacular fusion reflex remains within 0 to 8 prism diopters of horizontal eye alignment.
Such a large range of tolerance for horizontally disparate retinal images to be fused in the monofixator is easily demonstrated using stereopsis tests. Although the stereoacuity is poor in the monofixator compared with the bifixator,6 it does exist as long as the horizontal deviation of the eyes does not exceed 8 prism diopters. Along with the intact stereopsis, the fusional vergence amplitudes also remain normal in the monofixator as long as the horizontal deviation of the eyes does not exceed 8 prism diopters. In my experience, normal fusional vergence amplitudes and stereopsis perception occur only in normal retinal correspondence (NRC) but not in the adapted abnormal retinal correspondence (ARC).
Stereopsis can occur only if the horizontally disparate images in the monofixator's peripheral vision are from targets within Panum's fusional space. Although the center of the Panum's area is scotomatized, the peripheral Panum's area exists in small angles of deviation. To maintain this small horizontal deviation of 0 to 8 diopters (D) requires a good functioning fusional vergence reflex that can be generated by NRC but not by ARC.
VISUAL CONFUSION
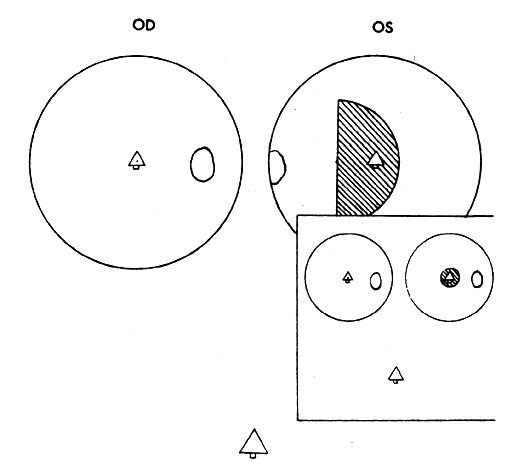
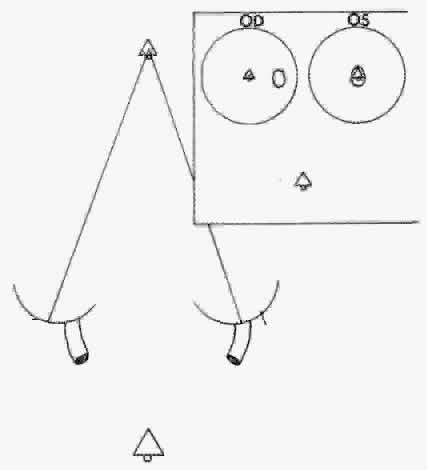
Visual confusion occurs in strabismus as a result of the projection onto corresponding retinal areas of simultaneously perceived dissimilar images of two different spatial objects that normally project onto noncorresponding retinal areas. Consequently, these different objects in space that create dissimilar images are perceived as located at the same place in space. Visual confusion does not exist for all portions of the visual field. The object of regard visually perceived by the fixating eye cannot simultaneously perceive a superimposed different image projecting on the fovea of the deviating eye. From the physiologic fact of binocular vision, as stated in Chapter 5, Fig. 13, only similar images projecting on the foveas are perceived simultaneously. No binocular function results from dissimilar images projecting onto these retinal areas. Consequently, from the moment the strabismic eyes deviate the patient is spared the visual confusion of a different spatial object's image conflicting with the object of regard. The three-degree macular scotoma in the esotropic left eye (Fig. 3) can be plotted using a binocular perimetric technique. This is a physiologic scotoma, present in all strabismic patients immediately at the moment the alignment of the eyes exceeds the threshold of 2/3 of a prism diopter. Therefore sustaining the bifixation reflex requires a very refined ocular alignment control along with an elegant level of similarity of the foveal images. The combination of these two inputs provide the essential stimuli for bifixation. The moment one or both are lacking, the bifixation reflex shuts down and a macular scotoma appears in the nonfixating eye. It is not a pathologic scotoma that evolves gradually to resolve the annoying visual symptom caused by strabismus. It is not an adaptation of the macular fusion reflex that gradually developed to eliminate visual confusion. In short, it is not a suppression scotoma.
RETINAL IMAGE SIMILARITY VERSUS DISSIMILARITY
In addition to image disparity, another stimulus that pertains to the alignment of the eyes with certain thresholds required for fusion is image dissimilarity. The threshold for image dissimilarity acceptable for fusion is different for macular fusion compared with extramacular fusion. Dissimilar image parameters are size, shape, clarity of focus, illumination brilliance, and color. Each has its threshold of dissimilarity that eliminates fusion. Anisometropia, media disturbances within the ocular tissues, optical appliances, and so forth are sources of image dissimilarity that affect fusion.
Anisometropia is probably the maximal cause of image dissimilarity affecting the macular and extramacular binocular vision reflexes. Weakley7 has shown that the macular binocular vision reflex threshold for being shut down by anisometropia is approximately 1.5 D spherical equivalent (SE), but the extramacular binocular vision reflex continues to function until a much greater quantity of SE anisometropia is induced. The test for this is use of a vectographic method for measuring the stereoacuity. The macular level of stereoacuity disappears at approximately 1.5 to 2.0 D SE, but gross stereoacuity continues. The three-degree macular scotoma instantly appears in the more unfocused eye and bifixation is replaced by monofixation. Consequently, two different stimuli are essential for the macular fusion reflex to function.
Function of the macular binocular fusion reflex requires simultaneous input from the two essential stimuli. First, the alignment of the eyes must not exceed 2/3 of a prism diopter (retinal image disparity greater than 20 seconds of arc), and second, anisometropia must be no greater than approximately 2 D SE7 (retinal image dissimilarity). The moment either of the two essential stimuli are absent macular binocular vision (bifixation) ceases to function and a macular scotoma appears in the nonfixating eye. However, these levels of retinal image disparity and dissimilarity are subthreshold for affecting the continued function of the extramacular binocular vision reflex (within 8 prism diopters of the eye deviation and unknown image dissimilarity). In the meantime the extramacular fusion reflex continues to function because its thresholds for fusing disparate images and/or dissimilar images are greater than those of the macular fusion reflex. The fusional vergence amplitude and stereoperception of the extramacular fusion reflex continue as long as the ocular deviation is 8 prism diopters or less and the image dissimilarity is approximately 4 D SE or less of induced anisometropia.*
*Note: In self-experimentation with the stereo fly test held at 16 in., my 40-second stereoacuity continued when incremental-plus-spherical lens power was placed before one eye until + 2 D of induced anisometropia was reached. The stereoacuity precipitously diminished to the 70- to 100-second range, which restored to 40 seconds on removing the + 2 D add and diminished to the 70- to 100-second range the instant the + 2 D add was replaced. With incremental addition of more + D add the 70- to 100-second stereoacuity range persisted until reaching the + 4 D level of induced anisometropia, at which time precipitously all stereoperception disappeared. The 3000-second stereotest fly went flat and appeared as a picture of a fly; when the + 4 D add is reduced to a + 3 D add, the stereoperception of the fly instantly returned. My interpretation of the results are that + 2 D-induced anisometropia is just beyond threshold of image dissimilarity tolerated by the macular binocular vision reflex, but + 4 D of induced anisometropia is required to exceed the threshold of image dissimilarity tolerated by the extramacular binocular vision reflex.